 Back
BackMolecules and Compounds: Structure, Nomenclature, and Calculations
Study Guide - Smart Notes

Molecules and Compounds
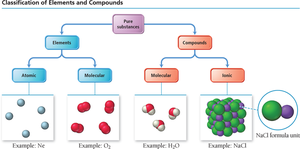
Classification of Matter
Matter can be classified as pure substances, which are further divided into elements and compounds. Elements may exist as single atoms (atomic) or as molecules (molecular), while compounds are formed from two or more elements bonded together and can be molecular or ionic in nature.
Elements: Pure substances that cannot be broken down into simpler substances by chemical means.
Compounds: Substances composed of two or more elements chemically bonded in fixed proportions.
Atomic Elements: Exist as single atoms (e.g., Ne, Na).
Molecular Elements: Exist as molecules, such as diatomic (O2, N2) or polyatomic (P4, S8).
Molecular Compounds: Composed of molecules formed by covalent bonds between nonmetals (e.g., H2O).
Ionic Compounds: Composed of ions held together by ionic bonds, typically between metals and nonmetals (e.g., NaCl).
Chemical Bonds
Atoms combine to form compounds through chemical bonds, which arise from attractions between charged particles (electrons and protons). The two main types of chemical bonds are ionic and covalent.
Ionic Bonds: Formed by the transfer of electrons from a metal to a nonmetal, resulting in cations and anions held together by electrostatic forces.
Covalent Bonds: Formed by the sharing of electrons between two or more nonmetals, resulting in molecules.
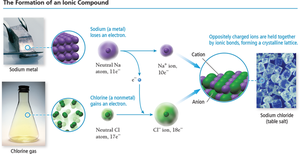
Ionic Bonds and Formation of Ionic Compounds
Ionic compounds are formed when metals transfer electrons to nonmetals, creating oppositely charged ions that attract each other.
Process: Metal loses electrons to form a cation; nonmetal gains electrons to form an anion.
Electrostatic Attraction: The resulting ions are held together by strong electrostatic forces.
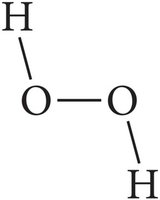
Covalent Bonds and Molecular Compounds
Molecular compounds are formed when nonmetals share electrons, resulting in discrete molecules. The term "molecule" specifically refers to compounds formed by covalent bonds.
Bonding: Electrons are shared between atoms to achieve stability.
Examples: H2O, CO2, CH4
Representing Compounds: Chemical Formulas and Molecular Models
Chemical Formulas
Chemical formulas use element symbols and subscripts to indicate the types and numbers of atoms in a compound.
Empirical Formula: Shows the simplest whole-number ratio of atoms (e.g., HO for hydrogen peroxide).
Molecular Formula: Shows the actual number of atoms in a molecule (e.g., H2O2 for hydrogen peroxide).
Structural Formula: Uses lines to represent covalent bonds and shows how atoms are connected.
Molecular Models
Molecular models provide a visual representation of the structure and shape of molecules. Ball-and-stick models show atoms as balls and bonds as sticks, while space-filling models represent the actual space occupied by atoms.
Ball-and-Stick Model: Highlights molecular geometry and bond angles.
Space-Filling Model: Shows the relative sizes and proximity of atoms in a molecule.
Color Coding: Atoms are often color-coded for clarity (e.g., hydrogen = white, oxygen = red).

Nomenclature of Compounds
Nomenclature: Ionic Compounds
Ionic compounds are named based on the ions they contain. Monoatomic ions are formed from individual atoms, while polyatomic ions consist of multiple atoms.
Monoatomic Cations: Metal atoms lose electrons to form cations (e.g., Na+ = sodium ion).
Monoatomic Anions: Nonmetal atoms gain electrons to form anions, named with the suffix "-ide" (e.g., Cl- = chloride ion).
Stock System: Used for metals that form more than one ion; charge is indicated by Roman numerals (e.g., Fe2+ = iron(II) ion).
Old System: Uses "-ic" for higher charge and "-ous" for lower charge (e.g., Fe3+ = ferric, Fe2+ = ferrous).
Polyatomic Ions: Ions composed of multiple atoms, often containing oxygen (e.g., NO3- = nitrate).
Writing Chemical Formulas: Ionic Compounds
Formulas for ionic compounds must reflect electrical neutrality. The crossover method is commonly used to balance charges.
Crossover Method: The charge of each ion becomes the subscript of the other ion, omitting the sign.
Polyatomic Ions: Use parentheses when more than one polyatomic ion is needed.
Lowest Ratio: Formulas should be reduced to the lowest whole-number ratio.

Nomenclature: Molecular Compounds
Molecular compounds (binary compounds of nonmetals) are named using Greek prefixes to indicate the number of atoms of each element.
First Element: Prefix + element name (mono- is usually omitted).
Second Element: Prefix + element stem + "-ide".
Examples: CO = carbon monoxide, PCl3 = phosphorus trichloride.

Acids and Hydrates
Acids: Nomenclature and Formulas
Acids are compounds that release hydrogen ions in aqueous solution. Their names depend on the anion present.
Binary Acids: Prefix "hydro-" + nonmetal stem + "-ic acid" (e.g., HF = hydrofluoric acid).
Ternary Acids: If the anion ends in "-ate", use "-ic acid"; if "-ite", use "-ous acid" (e.g., HNO3 = nitric acid, HNO2 = nitrous acid).
Formula Writing: Add enough H+ ions to balance the negative charge of the anion.
Hydrates
Hydrates are compounds that contain a specific number of water molecules bound to their structure. The number of water molecules is indicated by Greek prefixes.
Example: FeSO4 · H2O is iron(II) sulfate monohydrate.
Naming: Use prefixes such as mono-, di-, tri-, etc., to indicate the number of water molecules.
Molecular and Formula Weight, Molar Mass, and Calculations
Molecular Weight vs Formula Weight
The molecular weight (MW) is the sum of atomic weights of all atoms in a molecule, used for molecular compounds. Formula weight (FW) is used for ionic compounds and refers to the sum of atomic weights in a formula unit.
Molecular Weight: Used for molecules (e.g., CO2).
Formula Weight: Used for ionic compounds (e.g., MgSO4).
Molar Mass and Mole Calculations
Molar mass is the mass of one mole of a substance, numerically equal to the molecular or formula weight, with units of g/mol. It is used as a conversion factor between grams and moles, and Avogadro’s number (6.022 × 1023) is used to convert between moles and number of particles.
Molar Mass:
Avogadro’s Number: particles/mol
Mass Percent Composition
Mass Percent Composition
Mass percent composition expresses the percentage by mass of each element in a compound. It is calculated by dividing the mass of each element by the total mass of the compound and multiplying by 100%.
Formula:
Application: Used to determine the composition of compounds and for empirical formula calculations.
Empirical and Molecular Formulas
Empirical and Molecular Formulas
The empirical formula shows the simplest whole-number ratio of atoms in a compound, while the molecular formula shows the actual number of atoms. The molecular formula is always a whole-number multiple of the empirical formula.
Empirical Formula: Simplest ratio (e.g., CH for benzene).
Molecular Formula: Actual number of atoms (e.g., C6H6 for benzene).
Relationship:
Calculating Empirical and Molecular Formulas
Empirical formulas are determined from mass or percent composition data. The molecular formula is calculated if the molar mass is known.
Steps:
Convert mass or percent to moles for each element.
Divide by the smallest number of moles to get the simplest ratio.
Multiply to obtain whole numbers if necessary.
Calculate molecular formula using molar mass.
Formula:
Combustion Analysis and Instrumental Methods
Combustion Analysis
Combustion analysis is used to determine the percent composition of carbon, hydrogen, and oxygen in organic compounds. The sample is burned to produce CO2 and H2O, which are measured to calculate the amounts of C and H.
Steps:
Convert mass of CO2 to mass of C.
Convert mass of H2O to mass of H.
Calculate mass percent of C and H.
Determine percent of O by difference.
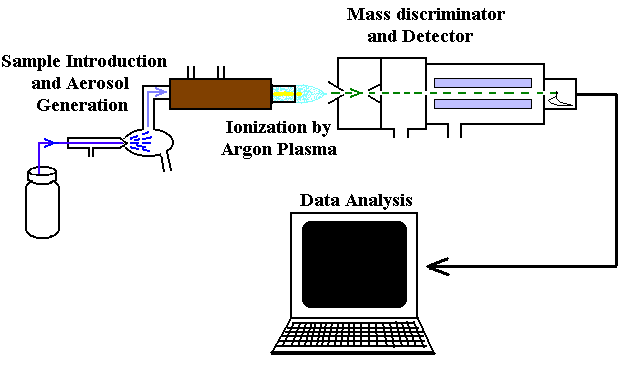
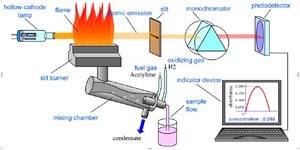
Additional info: Images 7 and 8 illustrate instrumental methods (ICP-MS, AAS, AES) used for elemental analysis in combustion analysis, relevant for determining percent composition in unknown compounds.
