 Back
BackMolecules and Compounds: Types, Representation, and Quantitative Composition
Study Guide - Smart Notes

Chapter 3: Molecules and Compounds

Introduction to Molecules and Compounds
Molecules and compounds are fundamental chemical entities formed by the combination of atoms. Understanding their types, how they are represented, and how to quantify their composition is essential in general chemistry.
Types of Compounds
Ionic Compounds
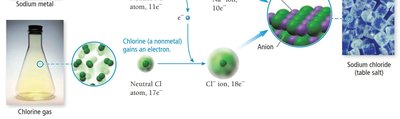
Ionic compounds are formed when metals transfer electrons to nonmetals, resulting in the formation of oppositely charged ions (cations and anions) that are held together by electrostatic attraction known as an ionic bond. The basic unit of an ionic compound is the formula unit, which is the smallest, electrically neutral collection of ions.
Metals lose electrons to form cations.
Nonmetals gain electrons to form anions.
Example: Sodium (Na) reacts with chlorine (Cl) to form sodium chloride (NaCl).

Covalent Compounds
Covalent compounds are formed when two or more nonmetals share electrons, resulting in a covalent bond. The atoms involved share electrons to achieve lower potential energy, and the resulting entity is called a molecule.
Nonmetal + Nonmetal → Covalent compound
Electrons are shared, not transferred.
Example: Water (H2O) is formed by sharing electrons between hydrogen and oxygen.
Representing Compounds: Chemical Formulas and Models
Chemical Formulas
Chemical formulas indicate the elements present in a compound and the relative number of atoms or ions of each element. Subscripts denote the number of atoms; a subscript of 1 is typically omitted. The more metallic element is usually listed first.
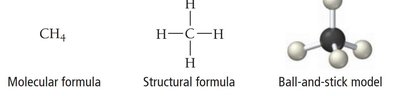
Molecular formula: Actual number of atoms of each element (e.g., H2O2).
Empirical formula: Simplest whole-number ratio of atoms (e.g., HO for H2O2).
Structural formula: Shows how atoms are connected (e.g., H–O–O–H).

Molecular Models
Models help visualize molecular structure:
Ball-and-stick model: Atoms as balls, bonds as sticks, showing geometry.
Space-filling model: (Not covered in detail here.)
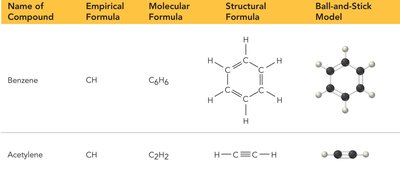
Comparison of Formula Types
Name of Compound | Empirical Formula | Molecular Formula | Structural Formula | Ball-and-Stick Model |
|---|---|---|---|---|
Benzene | CH | C6H6 | Hexagonal ring with alternating double bonds | Ring structure with alternating bonds |
Acetylene | CH | C2H2 | H–C≡C–H | Linear structure |
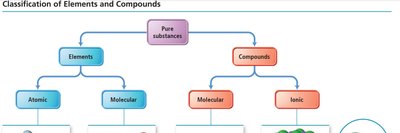
Classification of Elements and Compounds
Pure Substances: Elements vs. Compounds
Pure substances can be classified as elements (atomic or molecular) or compounds (molecular or ionic).
Atomic element: Single atoms (e.g., Ne).
Molecular element: Molecules of the same element (e.g., O2).
Molecular compound: Molecules of different elements (e.g., H2O).
Ionic compound: Lattice of ions (e.g., NaCl).
Practice: Classifying Substances
Classify substances as atomic element, molecular element, molecular compound, or ionic compound based on their composition and bonding.

Quantitative Composition of Compounds
Formula Mass and Molar Mass
The formula mass of a compound is the sum of the atomic masses of all atoms in its chemical formula, expressed in atomic mass units (amu). The molar mass is the mass of one mole of the compound, expressed in grams per mole (g/mol).
Formula mass = (number of atoms of element 1 × atomic mass of element 1) + (number of atoms of element 2 × atomic mass of element 2) + ...
Molar mass (g/mol) numerically equals formula mass (amu).

The Mole Concept: Converting Between Mass and Number of Molecules
The mole is a counting unit used to relate the mass of a substance to the number of particles (atoms, molecules). Avogadro's number (6.022 × 1023) is the number of particles in one mole.
To convert mass to moles:
To convert moles to number of molecules:

Mass Percent Composition
Mass percent composition expresses the percentage by mass of each element in a compound. It is calculated as:
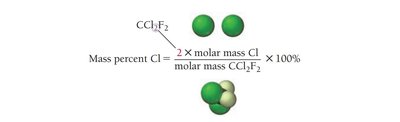
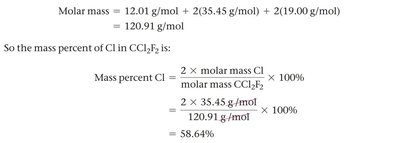
Using Mass Percent as a Conversion Factor
Mass percent can be used to convert between the mass of an element and the mass of a compound containing that element.
Example: If NaCl is 39% sodium by mass, the mass of Na in a given mass of NaCl can be found by multiplying by 0.39.

Determining Chemical Formulas from Experimental Data
Empirical and Molecular Formulas
The empirical formula gives the simplest whole-number ratio of atoms in a compound, while the molecular formula gives the actual number of atoms. Experimental data, such as mass percent composition, can be used to determine these formulas.
Steps to determine empirical formula:
Convert mass percent to grams (assume 100 g sample).
Convert grams to moles for each element.
Divide by the smallest number of moles to get subscripts.
If necessary, multiply subscripts to get whole numbers.

Combustion Analysis
Combustion analysis is a technique used to determine the empirical formula of a compound containing carbon, hydrogen, and sometimes oxygen. The compound is burned in oxygen, and the masses of CO2 and H2O produced are measured to calculate the amounts of C and H in the original sample.
Oxygen content is found by subtracting the masses of C and H from the total mass.

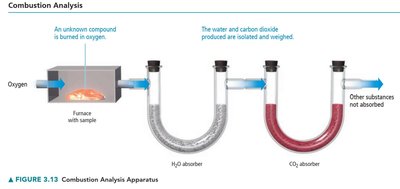
Determining Molecular Formula from Empirical Formula and Molar Mass
The molecular formula is a whole-number multiple of the empirical formula. It is determined by dividing the compound's molar mass by the empirical formula mass.
Molecular formula = (empirical formula) × n

Summary Table: Types of Chemical Formulas
Type | Description | Example (for H2O2) |
|---|---|---|
Molecular Formula | Actual number of atoms of each element | H2O2 |
Empirical Formula | Simplest whole-number ratio | HO |
Structural Formula | Shows connectivity | H–O–O–H |
Key Takeaways:
Compounds can be ionic or covalent, with distinct bonding and properties.
Chemical formulas and models provide different levels of information about molecular structure.
Quantitative composition (formula mass, molar mass, mass percent) is essential for laboratory calculations.
Empirical and molecular formulas can be determined from experimental data, including combustion analysis.
