 Back
BackNuclear Chemistry: Structure, Decay, and Applications
Study Guide - Smart Notes

Nuclear Chemistry
Introduction to Nuclear Chemistry
Nuclear chemistry is the study of the structure of atomic nuclei and the changes they undergo. It is essential for understanding radioactivity, nuclear reactions, and their applications in medicine, energy, and industry.
Nucleus: The dense, positively charged center of an atom, containing protons and neutrons.
Radioactivity: The spontaneous emission of particles or energy from unstable atomic nuclei.
Applications: Medical imaging, cancer therapy, dating ancient objects, food preservation, and power generation.
Atomic Models and the Gold Foil Experiment
Development of Atomic Models
The structure of the atom was elucidated through a series of experiments, most notably the Gold Foil Experiment, which disproved the "plum pudding" model and led to the nuclear (planetary) model.
Plum Pudding Model: Proposed by J.J. Thomson, this model depicted the atom as a sphere of positive charge with embedded electrons.
Rutherford Model: Proposed by Ernest Rutherford, this model described the atom as mostly empty space with a dense, positively charged nucleus at the center and electrons orbiting around it.
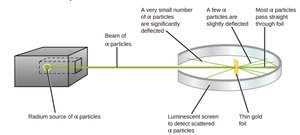


The Gold Foil Experiment (~1910)
The Gold Foil Experiment involved shooting alpha particles at a thin sheet of gold foil. Most particles passed through, but some were deflected at large angles, indicating a small, dense nucleus.
Alpha particles: Helium nuclei with a +2 charge and high mass.
Key Observations: Most alpha particles passed through, but a few were deflected or reflected, suggesting the presence of a dense nucleus.
Conclusion: Atoms are mostly empty space, with most of the mass concentrated in the nucleus.
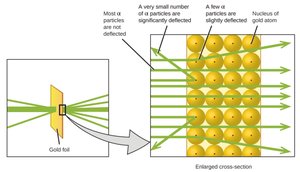
Structure of the Nucleus
Protons and Neutrons
The nucleus contains protons (positively charged) and neutrons (neutral). The number of protons determines the element, while the number of neutrons can vary, resulting in isotopes.
Proton: Charge = +1e, mass ≈ 1 amu.
Neutron: Charge = 0, mass ≈ 1 amu.
Isotopes: Atoms of the same element with different numbers of neutrons.
Radioactivity and Types of Decay
Overview of Radioactive Decay
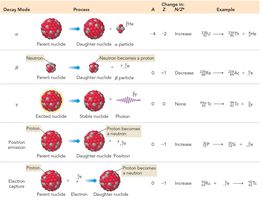
Unstable nuclei undergo radioactive decay to achieve stability. The main types of decay are alpha (𝛼), beta (β), positron emission, electron capture, and gamma (γ) emission.
Alpha Decay (𝛼): Emission of a helium nucleus (4He2+), decreases mass number by 4 and atomic number by 2.
Beta Decay (β): Emission of an electron (0-1e), increases atomic number by 1.
Positron Emission: Emission of a positron (0+1e), decreases atomic number by 1.
Electron Capture: A proton captures an electron and becomes a neutron, decreases atomic number by 1.
Gamma Emission (γ): Emission of high-energy photons, does not change mass or atomic number.
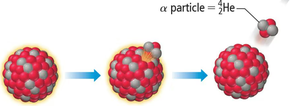
Alpha Decay
Alpha decay involves the emission of an alpha particle, resulting in a new element with a lower atomic and mass number.
General Equation:
Example:
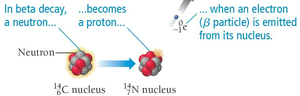
Beta Decay
Beta decay occurs when a neutron converts into a proton, emitting an electron (beta particle).
General Equation:
Example:
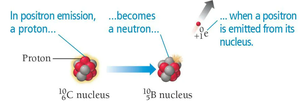
Positron Emission
Positron emission occurs when a proton converts into a neutron, emitting a positron.
General Equation:
Example:
Electron Capture
Electron capture involves a proton in the nucleus capturing an inner orbital electron and converting into a neutron.
General Equation:
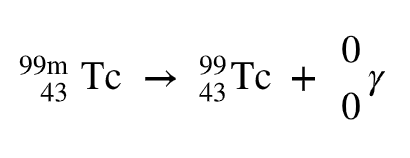
Gamma Ray Emission
Gamma emission involves the release of high-energy photons from an excited nucleus, with no change in atomic or mass number.
General Equation:
Summary Table: Nuclear Decay Modes
Decay Mode | Process | Change in A | Change in Z | Example |
|---|---|---|---|---|
Alpha (𝛼) | Emission of 4He nucleus | -4 | -2 | |
Beta (β) | Neutron to proton, electron emitted | 0 | +1 | |
Gamma (γ) | Excited to stable nucleus, photon emitted | 0 | 0 | |
Positron | Proton to neutron, positron emitted | 0 | -1 | |
Electron Capture | Proton + electron to neutron | 0 | -1 |
Applications of Radioactivity
Medical and Industrial Uses
Radioactivity has many practical applications, including medical diagnostics, cancer therapy, dating ancient objects, food preservation, and pest control.
Medical Imaging: Radioisotopes are used as tracers to diagnose organ function and detect cancer.
Food Preservation: Gamma rays are used to sterilize food and eliminate pathogens.
Dating: Radioactive isotopes such as carbon-14 and uranium are used to date archaeological and geological samples.
Pest Control: Radiation is used to sterilize insects, reducing pest populations.

Stability of Nuclei and the Island of Stability
Forces in the Nucleus
The stability of a nucleus depends on the balance between the repulsive electromagnetic force (between protons) and the attractive strong nuclear force (between nucleons).
Stable Nuclei: Have a specific ratio of neutrons to protons (N/Z ratio).
Island of Stability: Region where nuclei are stable; nuclei outside this region undergo decay to reach stability.
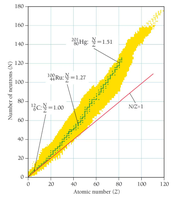
Decay Pathways
Above the island: Too many neutrons; undergo beta decay to convert neutrons to protons.
Below the island: Too many protons; undergo positron emission or electron capture to convert protons to neutrons.
Decay Series
Multiple Decays to Stability
Some heavy nuclei require a series of decays (alpha and beta) to reach a stable isotope. For example, uranium-238 decays through a series of steps to become lead-206. 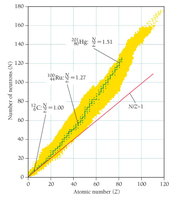
Detection and Measurement of Radioactivity
Detecting Radioactivity
Several instruments are used to detect and measure radioactivity:
MOSFET Dosimeter: Measures changes in voltage due to radiation-induced defects in a transistor.
Thermoluminescent Dosimeter: Measures light emitted from a material when heated after exposure to radiation.
Scintillation Counter: Detects light emitted by excited electrons returning to ground state after radiation exposure.
Geiger Counter: Measures conductivity changes in ionized argon gas caused by radiation.
Units of Radioactivity
Becquerel (Bq): 1 decay per second.
Curie (Ci): Bq.
Roentgen (R): Measures exposure to ionizing radiation in air.
Gray (Gy): Absorbed dose, 1 Gy = 1 J/kg tissue.
Rad: 100 rad = 1 Gy.
Rem: Dose equivalent, accounts for type of radiation and tissue sensitivity.
Sievert (Sv): 1 Sv = 100 rem.
Biological Effects of Radiation
Acute and Chronic Effects
Radiation can damage biological molecules by ionizing them, leading to cell death, cancer, or genetic defects.
Acute Damage: High doses kill cells, especially in the immune and digestive systems.
Cancer Risk: Repeated low doses can cause mutations leading to cancer.
Genetic Defects: Damage to reproductive cells can cause birth defects.
Half-Life and Radioactive Decay Calculations
Half-Life Concept
The half-life of a radioactive isotope is the time required for half of the sample to decay. It is used to calculate the remaining amount of a substance after a given time.
Formula:
Example: If you start with 16 g of (half-life 59.4 days), after 178.2 days, g remain.
Nuclear Fission and Fusion
Fission
Fission is the splitting of a heavy nucleus into two lighter nuclei, releasing a large amount of energy due to the conversion of mass into energy ().
Example:
Applications: Nuclear power plants, atomic bombs.
Fusion
Fusion is the joining of two light nuclei to form a heavier nucleus, also releasing energy. Fusion powers the sun and stars.
Example:
Applications: Potential for clean energy, but requires extremely high temperatures and pressures.
Summary
Nuclear chemistry explores the structure and transformations of atomic nuclei, the mechanisms of radioactive decay, and their profound applications in science and society. Understanding these principles is essential for fields ranging from medicine to energy production.
