Back
Backchem exam 4 (ch 17)-Organic Chemistry: Alkanes, Alkenes, Alkynes, and Functional Groups
Study Guide - Smart Notes

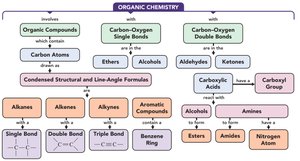
Organic Chemistry
Introduction to Organic Chemistry
Organic chemistry is the branch of chemistry that studies compounds containing carbon. Carbon atoms can bond together in various ways, forming a vast array of molecular structures. Organic compounds always contain carbon and hydrogen, and may also include other nonmetals such as oxygen, sulfur, nitrogen, phosphorus, or halogens.
Key Point: Organic compounds are found in everyday products like gasoline, medicines, plastics, and food.
Key Point: Organic compounds typically have low melting and boiling points, are not soluble in water, and are less dense than water.
Key Point: Many organic compounds burn vigorously in air, unlike most inorganic compounds.
Example: Vegetable oil (organic) floats on water and does not dissolve, while sodium chloride (inorganic) dissolves in water.


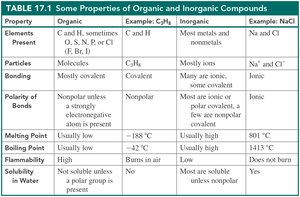
Properties of Organic and Inorganic Compounds
Organic and inorganic compounds differ in their physical and chemical properties. Organic compounds are mostly covalent, nonpolar, and have low melting and boiling points. Inorganic compounds are often ionic or polar covalent, and have high melting and boiling points.
Property | Organic | Inorganic |
|---|---|---|
Elements Present | C and H, sometimes O, S, N, P, or Cl | Most metals and nonmetals |
Bonding | Mostly covalent | Mostly ionic |
Polarity of Bonds | Nonpolar | Polar or ionic |
Melting Point | Usually low | Usually high |
Boiling Point | Usually low | Usually high |
Solubility in Water | Not soluble unless a polar group is present | Most are soluble unless nonpolar |
Flammability | Burns in air | Does not burn |
Alkanes
Structure and Bonding in Alkanes
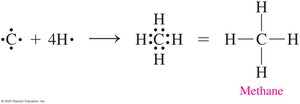
Alkanes are hydrocarbons containing only carbon and hydrogen atoms connected by single bonds. They are saturated hydrocarbons, meaning all carbon-carbon bonds are single bonds. The simplest alkane is methane, which has a tetrahedral structure.
Key Point: Hydrocarbons are organic compounds with only carbon and hydrogen.
Key Point: Methane (CH4) is the simplest alkane.
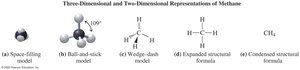
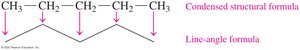
Key Point: Expanded structural formulas show all bonds; condensed formulas group atoms; line-angle formulas use zigzag lines for carbon skeletons.
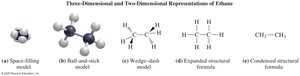
Naming Alkanes (IUPAC System)
Alkanes are named according to the International Union of Pure and Applied Chemistry (IUPAC) rules. The names reflect the number of carbon atoms in the main chain.
Key Point: The first four alkanes are methane, ethane, propane, and butane.
Key Point: Larger alkanes use prefixes: pent-, hex-, hept-, oct-, non-, dec-.
Number of Carbon Atoms | IUPAC Name | Molecular Formula | Condensed Structural Formula | Line-Angle Formula |
|---|---|---|---|---|
1 | Methane | CH4 | CH4 | |
2 | Ethane | C2H6 | CH3CH3 | |
3 | Propane | C3H8 | CH3CH2CH3 | |
4 | Butane | C4H10 | CH3CH2CH2CH3 | |
... | ... | ... | ... | ... |

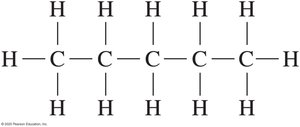
Drawing Structural Formulas for Alkanes
Alkanes can be represented in three ways: expanded, condensed, and line-angle formulas. For example, pentane (C5H12) can be drawn as follows:
Expanded formula: Shows all bonds between atoms.
Condensed formula: Groups hydrogen atoms with each carbon.
Line-angle formula: Uses zigzag lines; ends and corners represent carbon atoms.
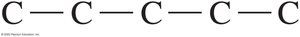
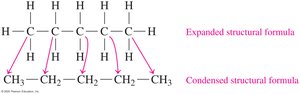
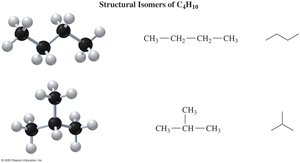
Isomers of Alkanes
Alkanes with four or more carbon atoms can have different arrangements, called structural isomers. Isomers have the same molecular formula but different bonding arrangements.
Key Point: Isomers differ in the connectivity of atoms, not just their spatial arrangement.
Example: C4H10 has two isomers: n-butane and isobutane.

Alkanes with Substituents
Alkanes can have branches or substituents, such as alkyl groups or halogens. Alkyl groups are named by replacing the -ane ending with -yl. Halogen substituents are named as fluoro, chloro, bromo, or iodo.
Key Point: Substituents are named and numbered according to their position on the main chain.
Key Point: The IUPAC name lists substituents in alphabetical order, using prefixes (di-, tri-, tetra-) for multiples.
Formula | Name |
|---|---|
CH3- | methyl |
CH3CH2- | ethyl |
CH3CH2CH2- | propyl |
F- | fluoro |
Cl- | chloro |
Br- | bromo |
I- | iodo |

Naming Alkanes with Substituents
To name an alkane with substituents:
Identify the longest carbon chain (main chain).
Number the chain from the end nearest a substituent.
List substituents in alphabetical order, indicating their position.
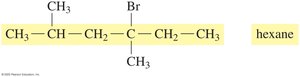
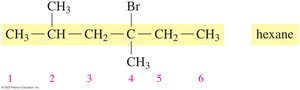
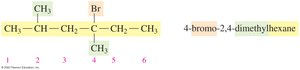
Drawing Structural Formulas from IUPAC Names
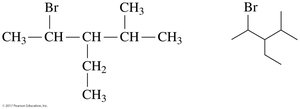
The IUPAC name provides information to draw condensed and line-angle formulas for alkanes with substituents. For example, 2,3-dimethylbutane:
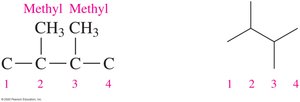
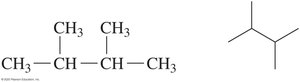
Uses and Reactions of Alkanes
Alkanes are used as fuels and solvents. The first four alkanes are gases at room temperature, while those with five to eight carbons are liquids. Alkanes undergo combustion reactions, producing energy.
Example: Propane combustion:
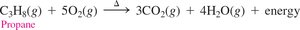

Alkenes, Alkynes, and Functional Groups
Functional Groups in Organic Chemistry
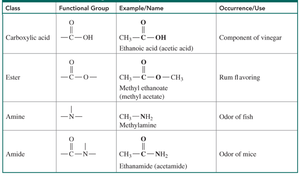
Functional groups are specific groups of atoms within molecules that determine the chemical properties of organic compounds. Compounds with the same functional group have similar properties and reactions.
Key Point: Functional groups allow classification and prediction of organic compound behavior.

Alkenes and Alkynes
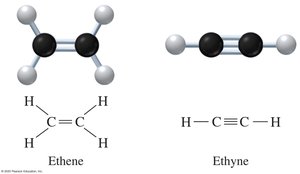
Alkenes and alkynes are unsaturated hydrocarbons. Alkenes contain at least one carbon-carbon double bond, while alkynes contain at least one carbon-carbon triple bond.
Key Point: Alkenes end with -ene; alkynes end with -yne.
Key Point: The main chain is numbered from the end nearest the double or triple bond.
Example: Ethene (C2H4), Ethyne (C2H2).
Alkane | Alkene | Alkyne |
|---|---|---|
CH3CH3 | H2C=CH2 | HC≡CH |
Propane | Propene | Propyne |

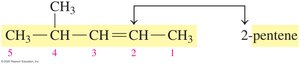
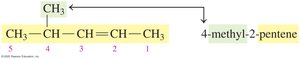
Naming Alkenes and Alkynes (IUPAC System)
The IUPAC rules for naming alkenes and alkynes are similar to those for alkanes, but the name reflects the presence and position of the double or triple bond.
Name the longest carbon chain containing the double or triple bond.
Number the chain from the end nearest the bond.
List substituents in alphabetical order, indicating their position.
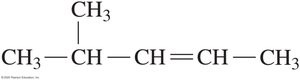

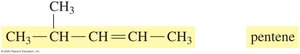
Classification by Functional Group
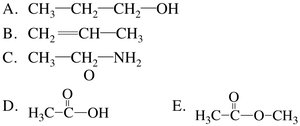
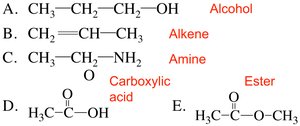
Organic compounds are classified by their functional groups, such as alcohols, alkenes, aldehydes, ketones, carboxylic acids, esters, amides, and amines.
Example: Alcohols contain an -OH group; alkenes contain a C=C bond; carboxylic acids contain a -COOH group.
Summary and Concept Map
Organic chemistry involves the study of compounds containing carbon atoms, classified by their functional groups. Alkanes, alkenes, and alkynes are hydrocarbons with different types of bonds. The IUPAC system is used to name these compounds, and their structural formulas can be drawn in various ways. Functional groups determine the properties and reactions of organic compounds.