 Back
BackOrganic Chemistry Functional Groups, Nomenclature, and Chirality Practice
Study Guide - Smart Notes

Q1. Hydrocarbons containing only single bonds between the carbon atoms are called alkanes________.
Background
Topic: Hydrocarbon Classification
This question tests your understanding of the different types of hydrocarbons based on the types of bonds between carbon atoms.
Key Terms:
Alkanes: Hydrocarbons with only single bonds between carbon atoms.
Alkenes: Hydrocarbons with at least one double bond between carbon atoms.
Alkynes: Hydrocarbons with at least one triple bond between carbon atoms.
Aromatics: Hydrocarbons with conjugated ring systems (e.g., benzene).
Step-by-Step Guidance
Recall the definitions of alkanes, alkenes, alkynes, and aromatics.
Identify which class contains only single bonds between carbons.
Eliminate options that contain double or triple bonds, or ring structures.
Try solving on your own before revealing the answer!
Q2. The name of the following compound is carboclyic acid________.
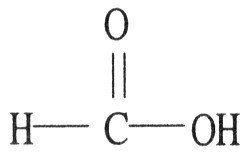
Background
Topic: Organic Nomenclature (IUPAC)
This question tests your ability to name an alkene with multiple double bonds using IUPAC rules.
Key Terms and Concepts:
Triene: Indicates three double bonds in the molecule.
Octa-: Indicates an eight-carbon chain.
Number the chain to give the double bonds the lowest possible numbers.
Step-by-Step Guidance
Identify the longest continuous carbon chain (should be 8 carbons for "octa").
Locate the positions of the double bonds and assign the lowest possible numbers to them.
Combine the positions and the suffix "triene" to form the name.
Try solving on your own before revealing the answer!
Q3. Which structure below represents a ketone?
Background
Topic: Functional Groups
This question tests your ability to recognize the ketone functional group among several structures.
Key Terms:
Ketone: A compound with a carbonyl group (C=O) bonded to two carbon atoms.
Aldehyde: A compound with a carbonyl group (C=O) bonded to at least one hydrogen atom.
Carboxylic Acid: A compound with a carbonyl group bonded to an -OH group.
Step-by-Step Guidance
Review the structures provided and identify the functional group in each.
Look for a carbonyl group (C=O) with two carbon atoms attached to it (not hydrogen or OH).
Eliminate structures that are aldehydes, carboxylic acids, or amines.
Try solving on your own before revealing the answer!
Q4. All of the following are achiral except ________, which is a chiral molecule.
Background
Topic: Chirality in Organic Molecules
This question tests your ability to identify chiral centers in organic molecules.
Key Terms:
Chiral molecule: A molecule that is not superimposable on its mirror image (has a stereocenter).
Achiral molecule: A molecule that is superimposable on its mirror image (no stereocenter).
Stereocenter: A carbon atom bonded to four different groups.
Step-by-Step Guidance
Examine each structure and identify any carbon atom bonded to four different groups.
Recall that a molecule with a stereocenter is chiral.
Eliminate molecules that are symmetrical or have no stereocenter.
Try solving on your own before revealing the answer!
Q5. Hydrocarbons containing carbon-carbon triple bonds are called ________.
Background
Topic: Hydrocarbon Classification
This question tests your knowledge of the types of hydrocarbons based on the presence of triple bonds.
Key Terms:
Alkynes: Hydrocarbons with at least one carbon-carbon triple bond.
Alkenes: Hydrocarbons with at least one carbon-carbon double bond.
Alkanes: Hydrocarbons with only single bonds.
Step-by-Step Guidance
Recall the definitions of alkynes, alkenes, and alkanes.
Identify which class contains a triple bond.
Eliminate options that do not fit the definition.
Try solving on your own before revealing the answer!
Q6. The addition of HBr to 2-butene produces ________.
Background
Topic: Electrophilic Addition to Alkenes
This question tests your understanding of Markovnikov's rule and the products of addition reactions to alkenes.
Key Terms and Concepts:
Markovnikov's Rule: In the addition of HX to an alkene, the hydrogen attaches to the carbon with more hydrogens, and the halide attaches to the carbon with fewer hydrogens.
2-butene: A four-carbon alkene with the double bond between carbons 2 and 3.
Step-by-Step Guidance
Draw the structure of 2-butene and identify the double bond.
Apply Markovnikov's rule to determine where H and Br will add.
Write the structure of the product and match it to the options given.
Try solving on your own before revealing the answer!
Q7. Name the following compounds:
Background
Topic: Organic Nomenclature (IUPAC)
This question tests your ability to name organic compounds based on their structure.
Key Terms and Concepts:
IUPAC Naming: Systematic method for naming organic compounds based on the longest carbon chain, functional groups, and substituents.
Step-by-Step Guidance
Identify the longest continuous carbon chain in each structure.
Number the chain to give the substituents the lowest possible numbers.
Name and locate each substituent (e.g., methyl, ethyl, etc.).
Combine the names and numbers according to IUPAC rules.
Try solving on your own before revealing the answer!
Q8. Draw the structures of the following compounds:
Meta-dichlorobenzene
2-ethyl-4-methylnonane
6-ethyl-3-octene
Background
Topic: Drawing Organic Structures
This question tests your ability to interpret IUPAC names and draw the corresponding structures.
Key Terms and Concepts:
Meta-: Substituents on a benzene ring separated by one carbon (positions 1,3).
Numbering: Assign numbers to the main chain to locate substituents and double bonds.
Step-by-Step Guidance
For each compound, start by drawing the parent structure (benzene ring, nonane, octene).
Number the carbons according to IUPAC rules.
Add the substituents (e.g., Cl, ethyl, methyl) at the correct positions.
For alkenes, indicate the position of the double bond.
Try drawing the structures on your own before checking the answer!
