 Back
BackOrganic Chemistry: Isomerism, Functional Groups, and Chirality
Study Guide - Smart Notes

Organic Chemistry: Isomerism, Functional Groups, and Chirality
Isomerism in Organic Chemistry
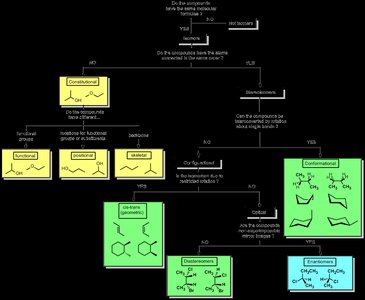
Isomers are compounds that have the same molecular formula but different structural arrangements or spatial orientations. Understanding isomerism is fundamental in organic chemistry, as it affects the physical and chemical properties of molecules.
Constitutional (Structural) Isomers: Molecules with the same molecular formula but different connectivity of atoms. Examples include functional isomers (different functional groups), positional isomers (same functional group, different position), and skeletal isomers (different carbon skeletons).
Conformational Isomers: Molecules that differ by rotation around single bonds. These are not true isomers but different spatial arrangements of the same molecule. Examples include staggered and eclipsed forms of ethane.
Stereoisomers: Molecules with the same connectivity but different spatial arrangement. Includes diastereomers (not mirror images) and enantiomers (non-superimposable mirror images).
Example: Butane and isobutane are constitutional isomers; cis- and trans-2-butene are stereoisomers.
Functional Groups in Organic Molecules
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. Identifying functional groups is essential for predicting reactivity and properties.
Alkane: Saturated hydrocarbons with only single bonds.
Alkene: Hydrocarbons containing at least one carbon-carbon double bond.
Alkyne: Hydrocarbons containing at least one carbon-carbon triple bond.
Aromatic Ring: Cyclic, planar ring with delocalized π electrons (e.g., benzene).
Alcohol: Contains a hydroxyl (-OH) group.
Ether: Contains an oxygen atom connected to two alkyl or aryl groups (R-O-R').
Aldehyde: Contains a carbonyl group (C=O) bonded to at least one hydrogen.
Ketone: Contains a carbonyl group (C=O) bonded to two carbon atoms.
Carboxylic Acid: Contains a carboxyl group (-COOH).
Ester: Contains a carbonyl group adjacent to an ether linkage (R-COO-R').
Amide: Contains a carbonyl group bonded to a nitrogen atom.
Amine: Contains a nitrogen atom bonded to carbon and/or hydrogen atoms.
Haloalkane: Contains a halogen atom (F, Cl, Br, I) bonded to an alkane.
Thiol: Contains a sulfhydryl (-SH) group.
Phenol: Contains a hydroxyl group attached to an aromatic ring.
Example: Ethanol (CH3CH2OH) is an alcohol; acetone (CH3COCH3) is a ketone.
Chirality and Chiral Centers
Chirality is a property of a molecule that is not superimposable on its mirror image. Chiral molecules have at least one chiral center, typically a carbon atom bonded to four different groups.
Chiral Center: A carbon atom with four unique substituents.
Enantiomers: Pair of chiral molecules that are mirror images but not superimposable.
Diastereomers: Stereoisomers that are not mirror images.
Identifying Chiral Centers: Mark the chiral carbon with a star (*) and circle the four unique groups.
Example: 2-butanol has a chiral center at the second carbon.
Hydrogen Counting in Molecular Structures
Organic molecules often omit hydrogen atoms in skeletal structures. It is important to identify and count all 'invisible' hydrogens to determine the molecular formula.
Each carbon in a skeletal structure is assumed to have enough hydrogens to complete four bonds.
Count all explicit and implicit hydrogens for accurate molecular formula.
Example: Cyclohexane (C6H12) has 12 hydrogens, though only the ring is drawn.
Summary Table: Types of Isomers
Type | Description | Example |
|---|---|---|
Constitutional Isomers | Same formula, different connectivity | Butane vs. Isobutane |
Conformational Isomers | Same formula, same connectivity, different spatial arrangement due to rotation | Staggered vs. Eclipsed ethane |
Stereoisomers | Same formula, same connectivity, different spatial arrangement | Cis- vs. Trans-2-butene |
Enantiomers | Non-superimposable mirror images | (R)- and (S)-2-butanol |
Diastereomers | Not mirror images, differ at some but not all chiral centers | Threo- vs. erythro-2,3-butanediol |
Key Equations and Notation
Molecular Formula: for alkanes
Chiral Center Notation: Marked with a star (*)
Isomerism:
Additional info: Academic context was added to clarify definitions, examples, and classification of isomers and functional groups, as well as the process for identifying chiral centers and counting hydrogens in skeletal structures.
