 Back
BackOxidation–Reduction (Redox) Reactions: Classification, Oxidation States, and Balancing
Study Guide - Smart Notes

Oxidation–Reduction Reactions (Redox Reactions)
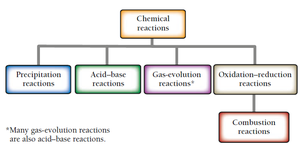
Classification of Chemical Reactions
Chemical reactions can be classified into several types based on the nature of the reactants and products. Understanding these categories helps in predicting reaction outcomes and identifying redox processes.
Precipitation reactions: Formation of a solid (precipitate) from two aqueous solutions.
Acid–base reactions: Formation of water upon mixing an acid and a base.
Gas-evolution reactions: Formation of a gas from two aqueous solutions. Many gas-evolution reactions are also acid–base reactions.
Oxidation–reduction (redox) reactions: Transfer of electrons between substances.
Combustion reactions: Reaction with oxygen, emitting heat and forming oxygen-containing compounds and often water.
Types of Chemical Reactions
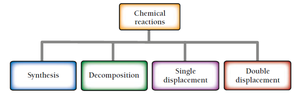
Chemical reactions can also be categorized by their structural changes:
Synthesis: Two or more substances combine to form one product.
Decomposition: One substance breaks down into two or more products.
Single displacement: One element replaces another in a compound.
Double displacement: Exchange of ions between two compounds.
Redox Reactions: Fundamentals
Definition and Examples
Oxidation–reduction (redox) reactions involve the transfer of electrons from one substance to another. These reactions are central to many chemical processes, including combustion, corrosion, and biological energy production.
Oxidation: Loss of electrons by a substance.
Reduction: Gain of electrons by a substance.
Oxidation and reduction always occur together; if one substance loses electrons, another must gain them.
Examples of redox reactions:
Combustion of hydrogen:
Rusting of iron:
Combustion of methane:

Identifying Redox Reactions
To determine whether a reaction is a redox reaction, look for electron transfer, reaction with elemental oxygen, or a metal reacting with a nonmetal.
Redox reactions involve changes in oxidation states.
In reactions between metals and nonmetals, the metal is oxidized and the nonmetal is reduced.
Oxidation States (Oxidation Numbers)
Rules for Assigning Oxidation States
Oxidation states are assigned to atoms in compounds to track electron transfer. The following rules are used:
Rule | Example |
|---|---|
1. The oxidation state of an atom in a free element is 0. | Cu, Cl2 (0 ox state) |
2. The oxidation state of a monatomic ion equals its charge. | Ca2+ (+2 ox state), Cl- (-1 ox state) |
3. The sum of oxidation states in a neutral molecule is 0; in an ion, it equals the ion's charge. | H2O: 2(+1) + 1(-2) = 0; NO3-: x + 3(-2) = -1 |
4. Group 1 metals always have +1; Group 2 metals always have +2. | NaCl: Na +1, Ca +2 |
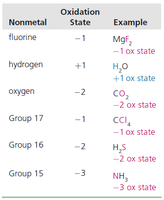
5. Assign nonmetals oxidation states by priority: F (-1), H (+1), O (-2), Group 17 (-1), Group 16 (-2), Group 15 (-3). | See table below |

Nonmetal | Oxidation State | Example |
|---|---|---|
Fluorine | -1 | MgF2 |
Hydrogen | +1 | H2O |
Oxygen | -2 | CO2 |
Group 17 | -1 | CCl4 |
Group 16 | -2 | H2S |
Group 15 | -3 | NH3 |
Sample Problems: Assigning Oxidation States
Assigning oxidation states helps identify which elements are oxidized or reduced in a reaction.
Zn: Oxidation state = 0 (elemental form).
Cu2+: Oxidation state = +2 (monatomic ion).
CaCl2: Ca = +2, Cl = -1 (sum = 0).
CF4: C = +4, F = -1 (sum = 0).
NO3-: N = +5, O = -2 (sum = -1).
SO3: S = +6, O = -2 (sum = 0).


Identifying Oxidation and Reduction in Reactions
Using Oxidation States to Track Electron Transfer
Changes in oxidation states indicate which elements are oxidized (increase in oxidation state) and which are reduced (decrease in oxidation state).
Oxidation: Increase in oxidation state (loss of electrons).
Reduction: Decrease in oxidation state (gain of electrons).
Example: C: 0 → +4 (oxidized), S: 0 → -2 (reduced)
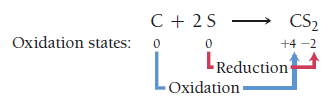
Sample Problem: Assigning Oxidation States and Identifying Changes
Assign oxidation states to each atom and identify which is oxidized and which is reduced.
Sn: 0 → +4 (oxidized)
N: +5 → +4 (reduced)

Balancing Redox Equations
The Half-Reaction Method
Redox reactions are often balanced using the half-reaction method, which separates the overall reaction into oxidation and reduction half-reactions. Each half-reaction is balanced for mass and charge, then combined to yield the balanced equation.
Assign oxidation states to all atoms.
Separate the reaction into two half-reactions.
Balance each half-reaction for mass (elements other than H and O).
Balance O by adding H2O; balance H by adding H+.
Balance charge by adding electrons.
Combine the half-reactions, ensuring electrons are canceled.
Verify the reaction is balanced for both mass and charge.

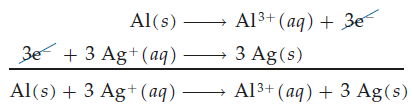

Summary Table: Oxidation States and Redox Identification
Compound | Oxidation State Assignment |
|---|---|
Zn | 0 |
Cu2+ | +2 |
CaCl2 | Ca: +2, Cl: -1 |
CF4 | C: +4, F: -1 |
NO3- | N: +5, O: -2 |
SO3 | S: +6, O: -2 |

Practice: Identifying Redox Reactions
Sample Reactions
Determine which reactions are redox and assign oxidation states:
(a) (Redox: Li oxidized, Cl reduced)
(b) (Redox: Al oxidized, Sn reduced)
(c) (Not redox: double displacement)
(d) (Redox: C oxidized, O reduced)
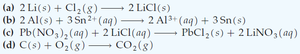
Conclusion
Redox reactions are fundamental to chemistry, involving electron transfer and changes in oxidation states. Mastery of assigning oxidation states, identifying redox processes, and balancing redox equations is essential for understanding chemical reactivity and predicting reaction outcomes.
