 Back
BackChapter 7 Chemistry
Study Guide - Smart Notes

Periodic Properties of Elements
Effective Nuclear Charge (Zeff)
The effective nuclear charge is the net positive charge experienced by valence electrons in a many-electron atom. It is a crucial factor in determining atomic properties, as it reflects the balance between the attraction of the nucleus and the repulsion from other electrons.
Definition: The effective nuclear charge (Zeff) is the net positive charge "felt" by an electron in a multi-electron atom.
Formula: where is the atomic number (number of protons) and is the screening constant (approximate number of core electrons).
Screening Effect: Core electrons shield valence electrons from the full charge of the nucleus, reducing the effective nuclear charge.
Trends: Zeff increases from left to right across a period due to increasing nuclear charge with relatively constant shielding.
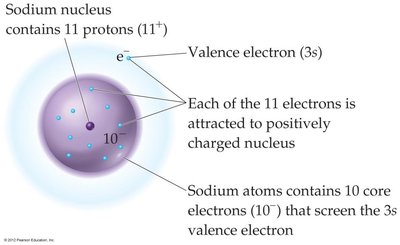
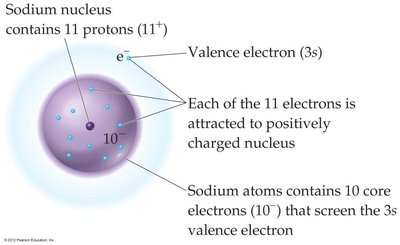
Example: For sodium (Na), , , so for the valence electron.
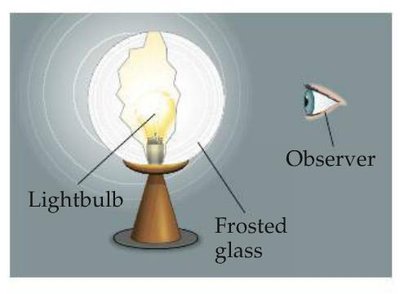
Analogy: The screening effect is like observing a lightbulb through frosted glass—the glass (core electrons) reduces the intensity of light (nuclear charge) reaching the observer (valence electron).
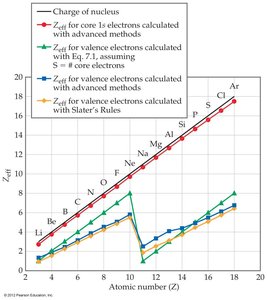
Summary of Trends: The effective nuclear charge increases steadily across a period, influencing many periodic properties.
Sizes of Atoms and Ions
Atomic Radii
The atomic radius is a measure of the size of an atom, typically defined as half the distance between the nuclei of two identical atoms bonded together. Atoms do not have sharply defined boundaries, so atomic radii are estimated from bonding distances.
Bond Length:
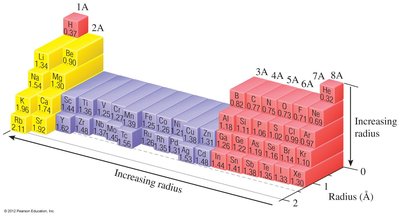
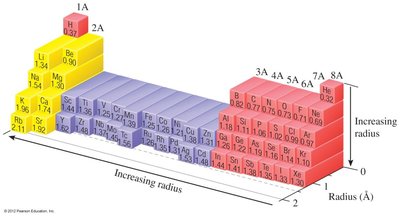
Trends:
Atomic radius decreases across a period (left to right) due to increasing Zeff.
Atomic radius increases down a group due to increasing principal quantum number (n).
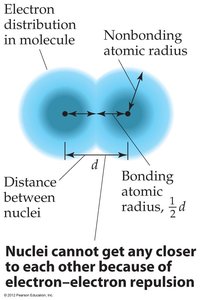
Example: The bond length of a C–S bond is .
Ionic Radii
The ionic radius is the radius of an ion in an ionic compound. It is determined from the distances between ions in a crystal lattice.
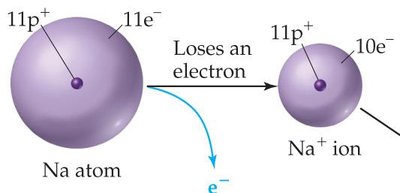
Cations (positive ions) are smaller than their parent atoms because they lose electrons, reducing electron-electron repulsion.
Anions (negative ions) are larger than their parent atoms due to increased electron-electron repulsion when electrons are added.
For ions with the same charge, ionic radius increases down a group as n increases.
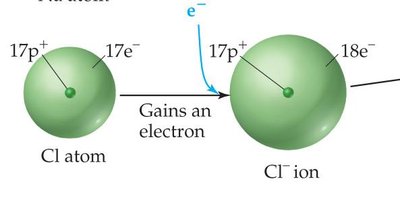
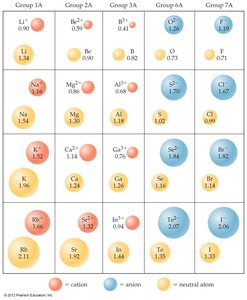
Isoelectronic Series: A group of ions with the same number of electrons. Within a series, ionic radius decreases as nuclear charge increases.
Ion | O2– | F– | Na+ | Mg2+ | Al3+ |
|---|---|---|---|---|---|
Atomic Number | 8 | 9 | 11 | 12 | 13 |
Electrons | 10 | 10 | 10 | 10 | 10 |
Ionic Radius (Å) | 1.26 | 1.19 | 1.16 | 0.86 | 0.68 |
Trend: As nuclear charge increases in an isoelectronic series, ionic radius decreases.
Ionization Energy
Definition and Trends
Ionization energy (IE) is the minimum energy required to remove an electron from a gaseous atom or ion. Successive ionization energies (I1, I2, I3, ...) refer to the removal of each subsequent electron.
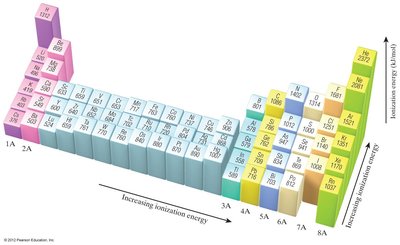
General Trend: Ionization energy increases across a period (left to right) and decreases down a group.
Each successive ionization energy is higher than the previous one: .
There is a large jump in ionization energy when removing a core electron.
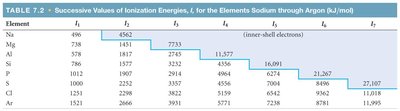
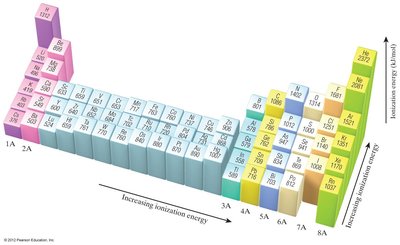
Example: The first ionization energy of sodium (Na) is 496 kJ/mol, but the second is much higher (4562 kJ/mol) because it involves removing a core electron.
Electron Affinity
Definition and Trends
Electron affinity (EA) is the energy change when an electron is added to a gaseous atom. A more negative value indicates a greater tendency to gain an electron.
General Trend: Electron affinity becomes more negative across a period (left to right) and less negative down a group.
Group 17 elements (halogens) have the most negative electron affinities, while Group 18 (noble gases) have positive or zero values due to their stable configurations.
Group 1 elements have more negative electron affinities than Group 2 elements.
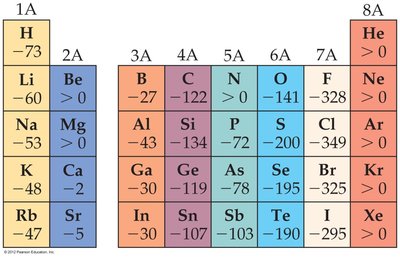
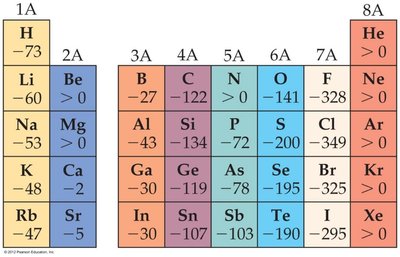
Example:
Comparison: Electron affinity is the opposite of ionization energy. Ionization energy measures the ease of losing an electron, while electron affinity measures the ease of gaining one.
Summary Table: Periodic Trends
Property | Across a Period (→) | Down a Group (↓) |
|---|---|---|
Effective Nuclear Charge (Zeff) | Increases | Slightly increases |
Atomic Radius | Decreases | Increases |
Ionic Radius | Decreases for isoelectronic ions | Increases |
Ionization Energy | Increases | Decreases |
Electron Affinity | Becomes more negative | Becomes less negative |
Additional info: These periodic trends are foundational for understanding chemical reactivity, bonding, and the structure of the periodic table. Mastery of these concepts is essential for predicting and rationalizing the behavior of elements in chemical reactions.
