 Back
BackPeriodic Properties of the Elements: Structure, Electron Configuration, and Trends
Study Guide - Smart Notes

Periodic Properties of the Elements
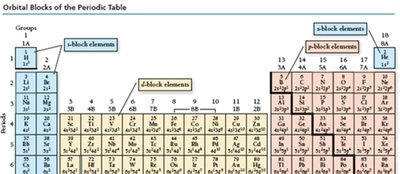
The Modern Periodic Table: Structure and Classification
The periodic table is a systematic arrangement of elements based on increasing atomic number. Its structure reveals recurring chemical properties and is foundational to understanding atomic behavior.
Periods: Horizontal rows in the table, indicating increasing principal quantum number (energy level).
Groups/Families: Vertical columns; elements in the same group share similar chemical properties due to similar valence electron configurations.
Classification: Elements are categorized as metals, nonmetals, and metalloids. The table is further divided into main-group elements (predictable properties) and transition/inner transition metals (less predictable properties).
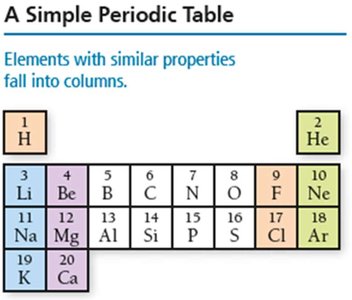
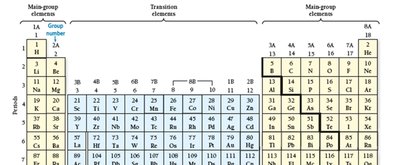
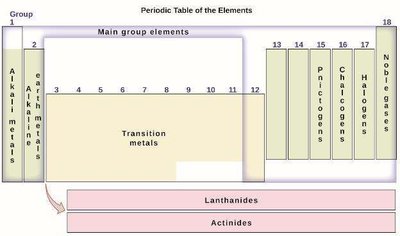
Example: Elements in Group 1 (alkali metals) are highly reactive and share similar properties, such as forming 1+ cations.
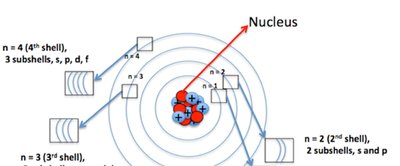
Electron Configuration and Quantum Theory
Quantum-mechanical theory describes how electrons occupy orbitals in atoms. The arrangement of electrons, or electron configuration, determines chemical properties and reactivity.
Orbitals: Regions in space where electrons are likely to be found (s, p, d, f types).
Aufbau Principle: Electrons fill orbitals from lowest to highest energy.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers; each orbital holds a maximum of two electrons with opposite spins.
Hund's Rule: Electrons occupy degenerate orbitals singly before pairing.
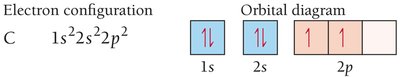
Example: The electron configuration for carbon is .
Quantum Numbers and Electron Spin
Each electron in an atom is described by four quantum numbers: principal (n), angular momentum (l), magnetic (ml), and spin (ms). The spin quantum number (ms) can be +½ or -½, representing spin up or spin down.
Spin: Fundamental property; electrons in the same orbital must have opposite spins.
Orbital Diagrams: Visual representations using boxes and arrows to show electron arrangement and spin.

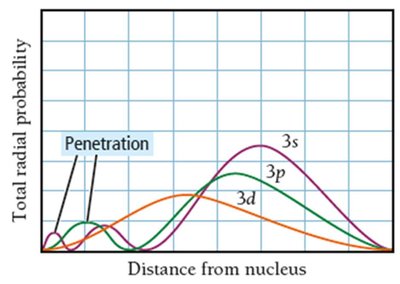
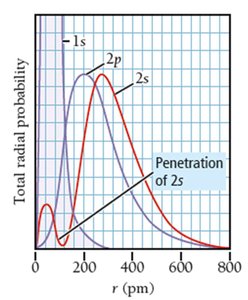
Sublevel Energy Splitting, Shielding, and Penetration
In multi-electron atoms, sublevels (s, p, d, f) within the same principal shell have different energies due to electron-electron repulsion, shielding, and penetration effects.
Shielding: Inner electrons block outer electrons from the full nuclear charge, reducing attraction.
Penetration: Some orbitals (e.g., s) allow electrons to get closer to the nucleus, increasing attraction and lowering energy.
Energy Order: For a given shell, .

Coulomb's Law and Effective Nuclear Charge
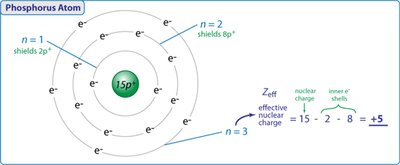
Coulomb's law quantifies the attractive and repulsive forces between charged particles. In atoms, the effective nuclear charge () is the net positive charge experienced by an electron, accounting for both attraction to the nucleus and repulsion by other electrons.
Coulomb's Law:
Effective Nuclear Charge: (Z = atomic number, S = shielding constant)
Writing Electron Configurations
Electron configurations can be written using the periodic table as a guide. Noble gas notation simplifies the representation by using the symbol of the preceding noble gas in brackets.
Steps:
Locate the element and determine its atomic number.
Fill orbitals in the order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, etc.
Use noble gas notation for inner electrons.
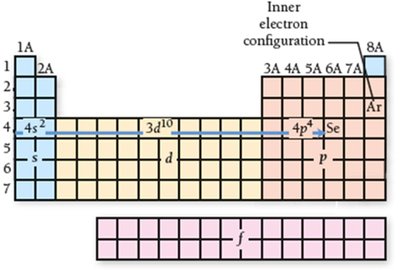
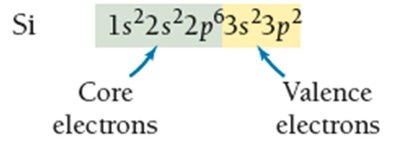
Valence and Core Electrons
Electrons in the highest principal energy level are called valence electrons and are responsible for chemical bonding. Electrons in lower energy levels are core electrons.
Valence electrons: Participate in bonding and determine reactivity.
Core electrons: Do not participate in bonding; shield valence electrons from the nucleus.
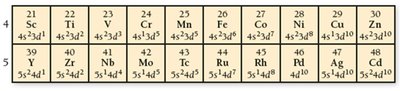
Transition Metals and Electron Configuration Anomalies
Transition and inner transition metals may have irregular electron configurations due to stability associated with half-filled or fully filled d sublevels.
Examples: Chromium (Cr): ; Copper (Cu):
Reason: Enhanced stability for half-filled or fully filled d orbitals.
Electron Configuration of Ions
When atoms form cations, electrons are removed first from the valence shell. For anions, electrons are added to the valence shell.
Cations: Remove electrons from the highest energy orbital (usually ns before (n-1)d).
Anions: Add electrons to the lowest available energy orbital.
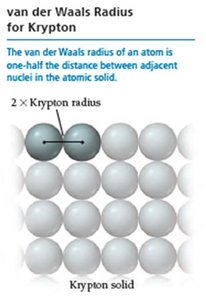
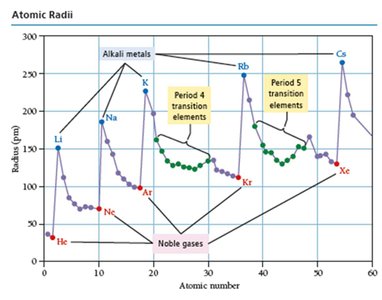
Periodic Trends: Atomic Radii
Atomic radius is a measure of the size of an atom. It varies systematically across the periodic table due to changes in effective nuclear charge and principal quantum number.
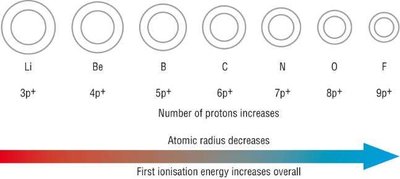
Across a period: Atomic radius decreases due to increasing .
Down a group: Atomic radius increases due to higher principal quantum number (n).
Types of radii: Van der Waals radius (nonbonding), covalent radius (bonding).
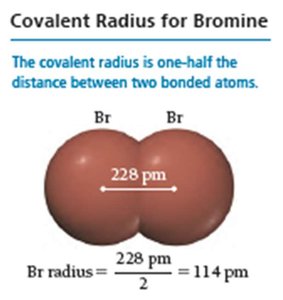

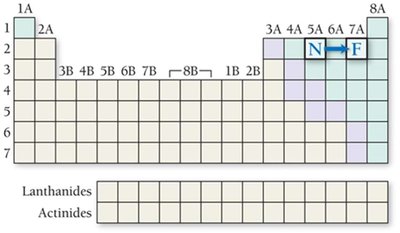
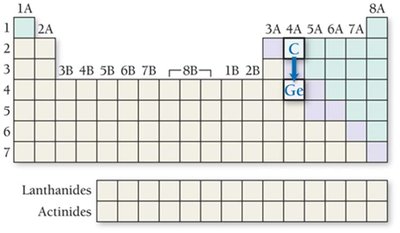
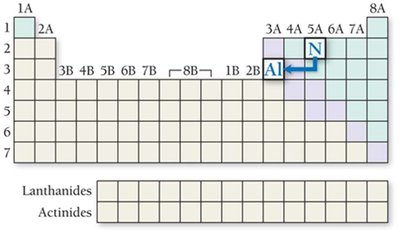
Radii of Ions: Cations and Anions
Ionic radii differ from atomic radii due to changes in electron number and effective nuclear charge.
Cations: Smaller than their parent atoms; loss of electrons increases .
Anions: Larger than their parent atoms; gain of electrons decreases .
Isoelectronic species: Ions with the same electron configuration; higher nuclear charge results in smaller radius.
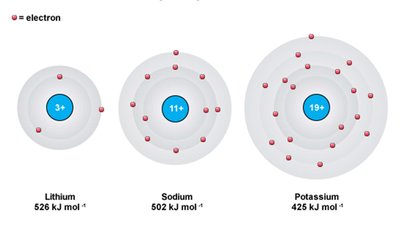
Periodic Trend: Ionization Energy
Ionization energy (IE) is the energy required to remove an electron from an atom in the gas phase. It reflects the strength of attraction between the nucleus and valence electrons.
First IE: Decreases down a group (electrons farther from nucleus), increases across a period (higher ).
Successive IE: Each additional electron removed requires more energy, with a large jump when core electrons are removed.
Exceptions: Sublevel structure and electron pairing can cause deviations from the general trend.
Periodic Trend: Electron Affinity
Electron affinity (EA) is the energy change when an atom gains an electron. It is typically exothermic for most elements, especially nonmetals.
Trend: EA becomes more negative (more exothermic) across a period; halogens have the highest EA.
Exceptions: Groups 2A and 8A have low or positive EA due to sublevel structure.
Periodic Trend: Metallic Character
Metallic character describes how closely an element's properties match those of metals. It is related to ease of electron loss and physical properties.
Trend: Metallic character decreases across a period and increases down a group.
Metals: Malleable, ductile, conduct electricity, form cations.
Nonmetals: Brittle, insulators, form anions.
Magnetic Properties: Paramagnetism and Diamagnetism
Magnetic properties of atoms and ions depend on electron configuration.
Paramagnetic: Atoms/ions with unpaired electrons; attracted to magnetic fields.
Diamagnetic: Atoms/ions with all electrons paired; slightly repelled by magnetic fields.
Summary Table: Periodic Properties and Trends
Property | Trend Across a Row | Trend Down a Column | Reason |
|---|---|---|---|
Atomic Radius | Decreases | Increases | Effective nuclear charge increases across; principal quantum number increases down |
Ionization Energy | Increases | Decreases | Electrons held more tightly across; farther from nucleus down |
Electron Affinity | Becomes more negative | No definite trend | Greater tendency to gain electrons across |
Metallic Character | Decreases | Increases | Ease of electron loss increases down |
Additional info: These notes expand on the original lecture slides and images by providing definitions, examples, and context for each concept, ensuring a comprehensive and self-contained study guide for general chemistry students.
