 Back
BackPeriodic Trends and Chemical Bonding: Study Notes for General Chemistry
Study Guide - Smart Notes

Periodic Trends and Bonding
Introduction
This section explores the periodic trends in ionisation energy, electron affinity, atomic and ionic radii, and electronegativity, as well as the fundamental types of chemical bonding. Understanding these trends and bonding types is essential for predicting chemical reactivity and the properties of elements and compounds.
Mathematical and Conceptual Foundations
Basic Calculations in Chemistry
Mole Calculations: The mass of a sample can be calculated using the number of moles and the molecular weight:
Density and Volume: The volume of a substance can be found using its mass and density:
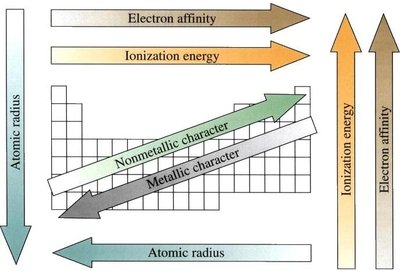
Periodic Trends
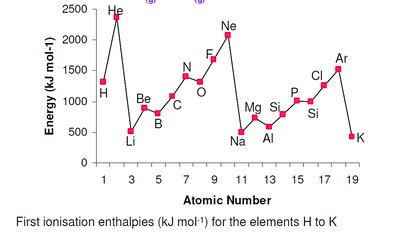
Ionisation Energy
Ionisation energy is the energy required to remove an electron from a gaseous atom or ion. It is a key indicator of an element's reactivity and its tendency to form cations.
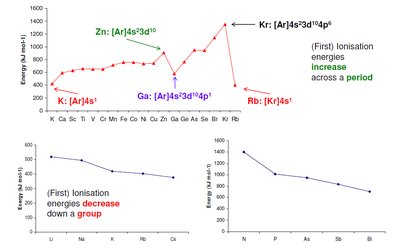
Trends:
Increases across a period (left to right) due to increasing nuclear charge and decreasing atomic radius.
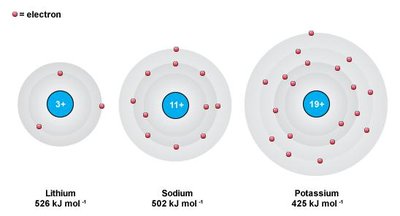
Decreases down a group due to increased atomic radius and electron shielding.
Anomalies: Occur when removing an electron results in or destroys a stable (full or half-full) subshell configuration.
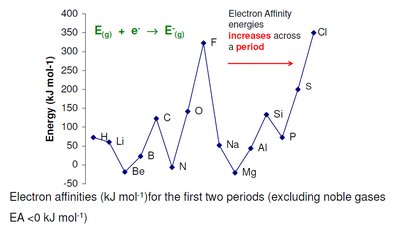
Electron Affinity
Electron affinity is the energy change when an electron is added to a neutral atom in the gas phase. A positive value (by the convention used here) indicates energy is released and the process is favorable.
Trends:
Generally increases (becomes more positive) across a period due to increasing nuclear charge.
Decreases down a group, except for the first member.
Noble gases have negative (unfavorable) electron affinities.
Anomalies: Occur when adding an electron results in or destroys a stable configuration.
Atomic and Ionic Radii
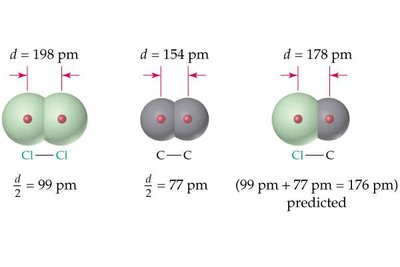
The atomic radius is defined as half the distance between the nuclei of two identical atoms bonded together. The ionic radius refers to the size of an ion in a crystal lattice.
Trends:
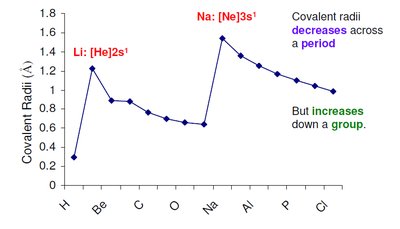
Atomic radius increases down a group (more electron shells).
Atomic radius decreases across a period (increased nuclear charge pulls electrons closer).
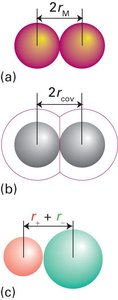
Types of Radii:
Metallic radius: Half the distance between nuclei in a metallic lattice.
Covalent radius: Half the distance between nuclei in a covalent bond.
Ionic radius: Distance between nuclei of neighboring cations and anions.
Electronegativity
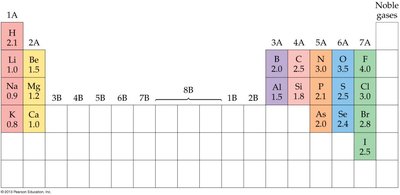
Electronegativity is a measure of an atom’s ability to attract electrons in a chemical bond. It is a dimensionless quantity and varies across the periodic table.
Trends:
Increases across a period (except noble gases).
Decreases down a group.
Applications: Used to predict bond polarity and the type of bonding between atoms.
Chemical Bonding
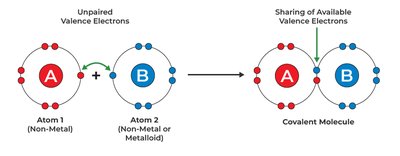
Types of Chemical Bonds
Ionic Bonds: Formed by the transfer of electrons from a metal to a non-metal, resulting in oppositely charged ions held together by electrostatic attraction.
Covalent Bonds: Formed by the sharing of electrons between non-metals.
Polar Covalent Bonds: Electrons are shared unequally due to differences in electronegativity.
Property | Covalent Bonds | Ionic Bonds |
|---|---|---|
State at Room Temp | Liquid or gaseous | Solid |
Polarity | Low | High |
Shape | Definite | No definite shape |
Melting/Boiling Point | Low | High |
Examples | CH4, HCl | NaCl, H2SO4 |
Occurs Between | Two non-metals | Metal and non-metal |
Electron Configurations and the Octet Rule
Atoms tend to gain, lose, or share electrons to achieve a stable configuration with eight valence electrons (the octet rule). Hydrogen and helium are exceptions, requiring only two electrons for stability.
Lewis Dot Structures: Visual representations of valence electrons as dots around the element symbol.
Bonding Capacity: The number of bonds an atom can form is related to the number of unpaired valence electrons.
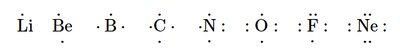
Ionic Bonding
Ionic bonding involves the complete transfer of electrons from a metal to a non-metal, resulting in the formation of cations and anions. The resulting electrostatic attraction forms an ionic compound.
Example: Formation of NaCl from Na and Cl atoms.
Predicting Formulas: Use the octet rule and ion charges to determine the empirical formula of ionic compounds.
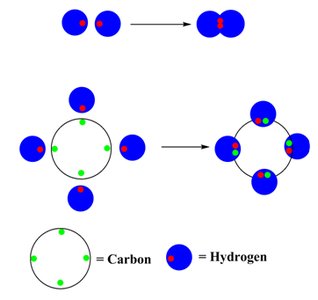
Covalent Bonding
Covalent bonding occurs when two atoms share pairs of electrons to achieve a full valence shell. The shared electrons count toward the octet for both atoms involved.
Single, Double, and Triple Bonds:
Single bond: One shared pair (e.g., H2)
Double bond: Two shared pairs (e.g., O2)
Triple bond: Three shared pairs (e.g., N2)
Bonding vs Non-Bonding Electrons:
Bonding electrons are shared between atoms.
Non-bonding (lone pair) electrons are not involved in bonding.
Summary Table: Periodic Trends
Trend | Across a Period | Down a Group |
|---|---|---|
Ionisation Energy | Increases | Decreases |
Electron Affinity | Increases | Decreases |
Atomic Radius | Decreases | Increases |
Electronegativity | Increases | Decreases |
Key Takeaways
Periodic trends in ionisation energy, electron affinity, atomic radius, and electronegativity are fundamental to understanding chemical reactivity and bonding.
Ionic bonding involves electron transfer; covalent bonding involves electron sharing.
The octet rule helps predict the stability and formulas of compounds.
Lewis dot structures are useful for visualizing valence electrons and predicting bonding patterns.
