 Back
BackPeriodicity and the Electronic Structure of Atoms: Periodic Trends and Quantum Mechanics
Study Guide - Smart Notes

Periodicity and the Electronic Structure of Atoms
Introduction to Periodicity
Periodicity refers to the presence of repeating patterns in the properties of elements as you move across the periodic table. Understanding periodicity is fundamental to predicting and explaining the behavior of elements based on their atomic structure.
Periodic Table Arrangement: The periodic table is organized so that elements with similar properties appear in the same columns (groups).
Goal: To explain elemental properties based on their electron configurations.
Example: Atomic radius trends across periods and groups.
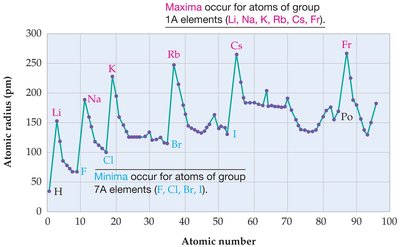
Atomic Radius and Periodic Trends
The atomic radius is a key property that exhibits periodic behavior. It is defined as the distance from the nucleus to the outermost electron shell. Atomic radius varies predictably across the periodic table:
Maxima: Occur for atoms of group 1A elements (Li, Na, K, Rb, Cs, Fr).
Minima: Occur for atoms of group 7A elements (F, Cl, Br, I).
Trend: Atomic radius generally decreases across a period (left to right) and increases down a group (top to bottom).
Reason: This trend is explained by changes in electron configuration and effective nuclear charge.
Example: The graph above shows atomic radius as a function of atomic number, with clear periodic maxima and minima corresponding to group 1A and 7A elements, respectively.
The Beginnings of Quantum Mechanics
Quantum mechanics revolutionized our understanding of atomic structure. Prior to the twentieth century, physical phenomena were believed to be deterministic, meaning future conditions could be predicted from present conditions (e.g., Newton's Laws of Motion). However, studies of subatomic particles revealed that their behavior is fundamentally probabilistic.
Key Physicists: Albert Einstein, Niels Bohr, Louis de Broglie, Max Planck, Werner Heisenberg, P. A. M. Dirac, and Erwin Schrödinger.
Quantum Mechanics: Provides a probabilistic model for electron behavior in atoms.
Impact: Quantum mechanics forms the foundation of modern chemistry.
Quantum-Mechanical Model of the Atom
The quantum-mechanical model explains how electrons exist and behave in atoms. This model is essential for understanding and predicting the properties of elements, which are directly related to electron behavior.
Explains:
Why some elements are metals and others are nonmetals
Why some elements gain one electron when forming an anion, while others gain two
Why some elements are highly reactive and others are inert
Why properties of elements exhibit periodic patterns
Foundation: Understanding the behavior of light and its interaction with matter was crucial to the development of quantum mechanics.
Key Terms and Concepts
Atomic Radius: The distance from the nucleus to the outermost electron shell.
Electron Configuration: The arrangement of electrons in an atom's orbitals.
Effective Nuclear Charge (Zeff): The net positive charge experienced by valence electrons.
Quantum Mechanics: The branch of physics that describes the behavior of subatomic particles using probabilistic models.
Relevant Equations
Effective Nuclear Charge: Where Z is the atomic number and S is the shielding constant.
Atomic Radius Trend: Atomic radius decreases across a period due to increasing Zeff, and increases down a group due to additional electron shells.
