 Back
BackPeriodicity and the Electronic Structure of Atoms: Quantum Theory, Light, and Atomic Spectra
Study Guide - Smart Notes

Periodicity and the Electronic Structure of Atoms
Quantum Mechanics and Electron Behavior
Quantum mechanics is the fundamental theory that explains the behavior of electrons in atoms. The quantum-mechanical model allows chemists to predict and understand atomic properties, including why elements exhibit periodic patterns, why some are metals or nonmetals, and why their reactivity varies.
Quantum mechanics forms the foundation of modern chemistry.
Electron behavior determines atomic properties and periodicity.
Understanding light was crucial to developing quantum theory.
Wave Properties of Light
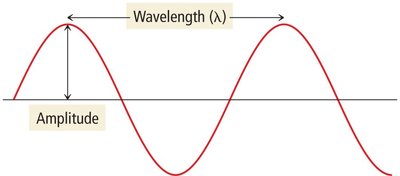
Light is an electromagnetic wave characterized by its wavelength, amplitude, and frequency. These properties determine the color and brightness of light.
Wavelength (\lambda): The distance between two consecutive crests of a wave. Measured in meters (m), centimeters (cm), or nanometers (nm).
Amplitude: The height of the wave from the center line to the crest, determining the brightness of light.
Frequency (\nu): The number of wave crests passing a point per unit time, measured in hertz (Hz), where 1 Hz = 1 s−1.
Velocity: The speed of the wave, given by , where is the speed of light ( m/s).
Continuous Spectrum of Light
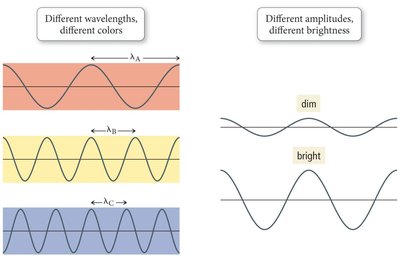
When white light is separated into its component wavelengths, a continuous spectrum is produced, displaying all visible colors. The color of light is determined by its wavelength or frequency, and its brightness by amplitude.
White light contains all visible wavelengths (colors).
Objects appear colored based on which wavelengths they reflect or absorb.
All colors travel at the same speed in a vacuum.

Electromagnetic Radiation and the Electromagnetic Spectrum
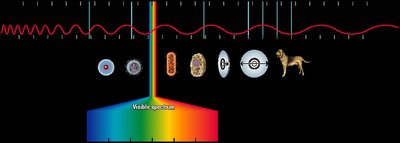
Electromagnetic radiation consists of oscillating electric and magnetic fields that propagate through space. The electromagnetic spectrum includes a wide range of wavelengths, from gamma rays to radio waves, but visible light is only a small portion.
Electromagnetic spectrum: Includes gamma rays, X-rays, ultraviolet (UV), visible, infrared (IR), microwave, and radio waves.
Energy increases from radio waves (low frequency, long wavelength) to gamma rays (high frequency, short wavelength).
Visible light ranges from about 400 nm (violet) to 700 nm (red).


Applications of Electromagnetic Radiation
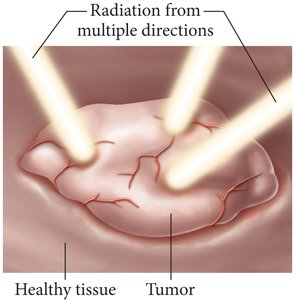
High-energy electromagnetic radiation is used in medical applications, such as radiation therapy for cancer treatment. Targeting tumors with radiation minimizes exposure to healthy tissue.
Radiation therapy uses focused beams to destroy cancer cells.
Different types of electromagnetic radiation have different biological effects.
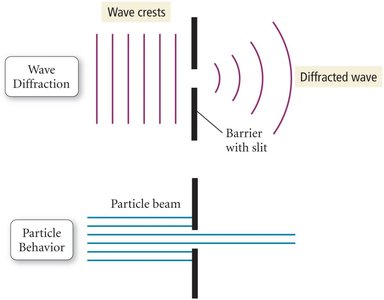
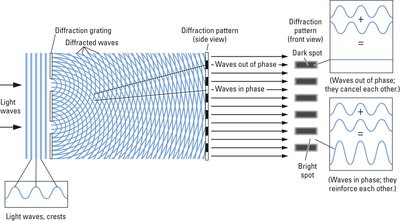
Wave Properties: Refraction and Diffraction
Light exhibits wave-like behaviors such as refraction and diffraction. Refraction occurs when light passes through a prism, separating into its component colors. Diffraction occurs when waves bend around obstacles or pass through slits.
Refraction: Bending of light as it passes through a medium, separating into colors.
Diffraction: Bending of waves around obstacles or through slits, producing interference patterns.
Particles do not diffract; only waves do.

The Photoelectric Effect
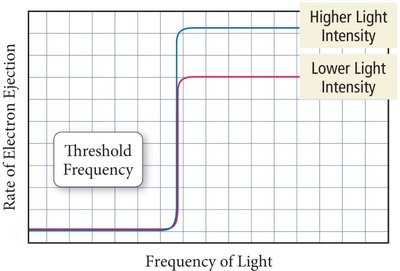
The photoelectric effect is the emission of electrons from a metal surface when exposed to light. Classical wave theory could not explain certain observations, such as the need for a minimum frequency (threshold frequency) for electron emission.
Electrons are ejected only if the light's frequency exceeds a threshold value.
Intensity affects the number of electrons ejected, but not their energy.
The energy required to eject an electron is called the work function (\phi).
Excess energy becomes the kinetic energy of the ejected electron.
Key equation:
Planck's Quantum Theory and Einstein's Photon Model
Planck proposed that energy is quantized and emitted in discrete packets called quanta. Einstein extended this idea, suggesting that light consists of particles called photons, each with energy proportional to its frequency.
Energy of a photon:
Planck's constant: J·s
Light exhibits both wave and particle properties (wave-particle duality).
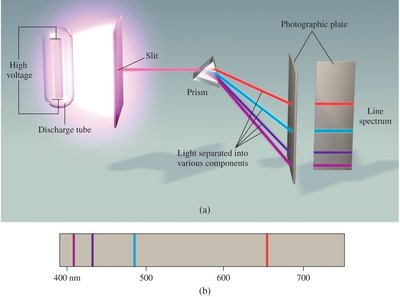
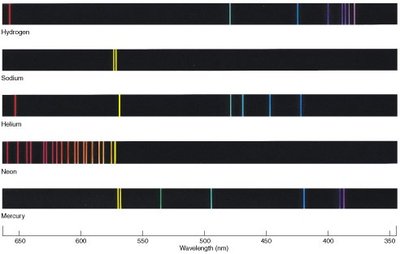
Continuous and Line Spectra
Heated solids emit continuous spectra, while excited atomic gases emit line spectra at specific wavelengths. Each element has a unique emission spectrum, which is used for identification.
Continuous spectrum: All wavelengths present, as in white light or heated solids.
Line spectrum: Only specific wavelengths emitted, characteristic of each element.
Visible spectrum: 400–700 nm.

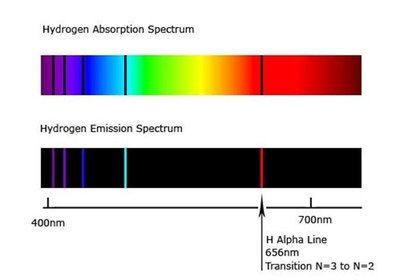
Emission vs. Absorption Spectra
Emission spectra show the wavelengths emitted by excited atoms, while absorption spectra show the wavelengths absorbed. These spectra are fundamental to understanding atomic structure and transitions.
Emission: Light emitted at specific wavelengths.
Absorption: Light absorbed at specific wavelengths, leaving dark lines in the spectrum.
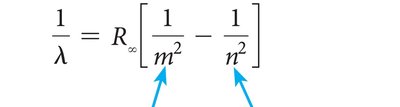
Rydberg's Spectrum Analysis and Equation
Johannes Rydberg analyzed the hydrogen spectrum and found it could be described by an equation involving the inverse squares of integers. This equation laid the groundwork for the Bohr model of the atom.
Rydberg equation:
is the Rydberg constant ( m−1).
and are integers ().
Additional info: The Bohr model, which will be discussed in subsequent lectures, uses quantized energy levels to explain the observed line spectra of hydrogen and other elements.
