 Back
BackPeriodicity, Electronic Structure, and Ionic Bonding: Study Notes for General Chemistry
Study Guide - Smart Notes

Periodicity and the Electronic Structure of Atoms
Periodic Trends in Properties of Elements
The periodic table displays repeating patterns, known as periodicity, in the properties of elements. These patterns are explained by the arrangement of electrons in atoms, which is described by electron configuration.
Periodicity: The presence of repeating patterns in element properties.
Electron Configuration: The distribution of electrons in atomic orbitals determines element properties.
The Beginning of Quantum Mechanics
Quantum mechanics revolutionized our understanding of atomic structure by introducing probabilistic models for subatomic particles, replacing deterministic classical physics.
Classical Physics: Predicts future conditions from current conditions (e.g., Newton's Laws).
Quantum Mechanics: Describes subatomic phenomena probabilistically.
Wave Properties of Light
Light exhibits both wave and particle properties. Key wave characteristics include wavelength, amplitude, and frequency. The energy of a wave depends on its amplitude and frequency.
Wavelength (\lambda): Distance between crests.
Frequency (\nu): Number of crests passing a point per unit time.
Velocity:
Energy: Proportional to amplitude and frequency.
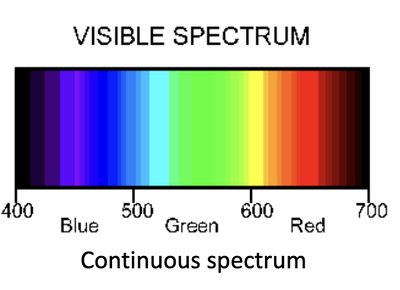
Electromagnetic Radiation and the Spectrum
Electromagnetic radiation consists of oscillating electric and magnetic fields, traveling through space. The visible spectrum is a small portion of the entire electromagnetic spectrum.
Speed of Light (c): m/s
Relationship:
Continuous Spectrum: White light contains all visible wavelengths.
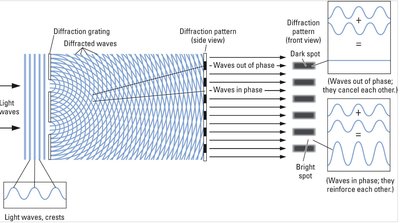
Properties of Waves: Diffraction and Interference
Light waves can diffract and interfere, producing patterns of bright and dark spots. Diffraction occurs when waves encounter obstacles or openings similar in size to their wavelength.
Diffraction: Bending of waves around obstacles.
Interference: Waves in phase reinforce (bright spots); out of phase cancel (dark spots).
The Photoelectric Effect and Quantum Theory
The photoelectric effect demonstrates the particle nature of light. Electrons are ejected from metal surfaces when exposed to light above a threshold frequency, regardless of intensity.
Threshold Frequency: Minimum frequency required to eject electrons.
Work Function (\phi): Minimum energy needed to remove an electron.
Energy of Photon:
Kinetic Energy:
Calculating Energy and Wavelength for Photoelectric Effect
To determine the maximum wavelength that causes the photoelectric effect, convert energy per mole to energy per photon, then use the photon energy to calculate wavelength.
Energy per photon:
Wavelength:

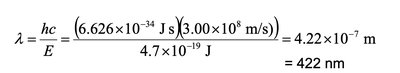
Wave-Particle Duality
Light exhibits both wave and particle properties, a concept known as wave-particle duality. This duality is also observed in electrons.
Wave Behavior: Diffraction and interference.
Particle Behavior: Photoelectric effect and emission spectra.
Bohr Model and Hydrogen Atom Spectra
The Bohr model explains the emission and absorption spectra of hydrogen by quantizing electron orbits. Energy transitions correspond to specific wavelengths.
Energy Transition:
Bohr Equation:
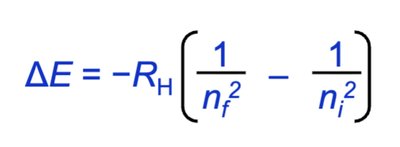
Quantum Mechanical Model and Quantum Numbers
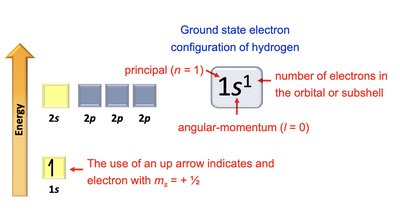
Quantum mechanics describes electrons as standing waves, characterized by quantum numbers. Each atomic orbital is defined by a set of quantum numbers.
Principal Quantum Number (n): Specifies size and energy level.
Angular Momentum Quantum Number (\ell): Specifies shape.
Magnetic Quantum Number (m_\ell): Specifies orientation.
Electron Spin Quantum Number (m_s): Specifies spin direction (+½ or -½).



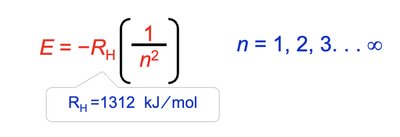
Energy Levels in Hydrogen Atom
For hydrogen, the energy of an orbital depends only on the principal quantum number (n). All subshells with the same n have equal energy.
Energy Equation: , where
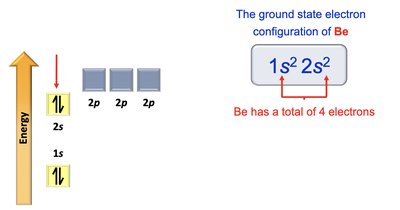
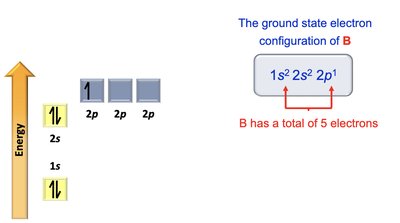
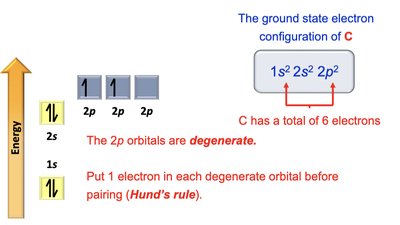
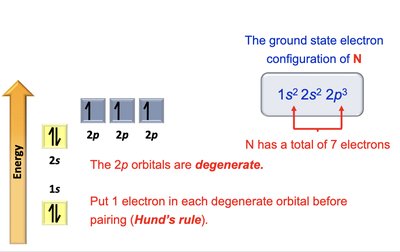
Electron Configurations and Orbital Diagrams
Electron configurations describe how electrons are distributed in atomic orbitals. The Aufbau principle, Pauli exclusion principle, and Hund's rule guide the filling of orbitals.
Aufbau Principle: Electrons fill lowest energy orbitals first.
Pauli Exclusion Principle: Maximum of two electrons per orbital, with opposite spins.
Hund's Rule: Electrons occupy degenerate orbitals singly before pairing.
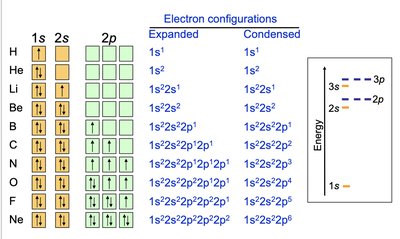
Examples of Electron Configurations
Ground state electron configurations for hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, and oxygen illustrate the application of quantum numbers and orbital filling rules.
Hydrogen: 1s1
Helium: 1s2
Lithium: 1s2 2s1
Beryllium: 1s2 2s2
Boron: 1s2 2s2 2p1
Carbon: 1s2 2s2 2p2
Nitrogen: 1s2 2s2 2p3
Oxygen: 1s2 2s2 2p4



Condensed Electron Configurations
Condensed electron configurations use noble gas notation to simplify the representation of electron arrangements.
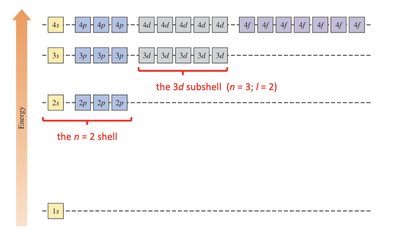
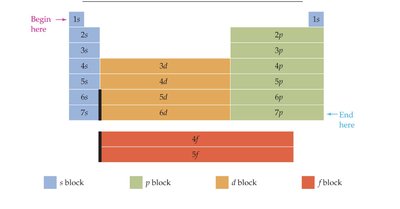
Orbital Filling Order and Periodic Table
The periodic table structure reflects the order of orbital filling, which follows the sequence: 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p, etc.
Valence Electrons and Noble Gas Notation
Valence electrons are those in the highest principal energy shell. Noble gas notation uses the electron configuration of the preceding noble gas as a core.
Valence Electrons: Determine chemical reactivity and periodic properties.
Noble Gas Notation: [He], [Ne], [Ar], [Kr], [Xe], [Rn]



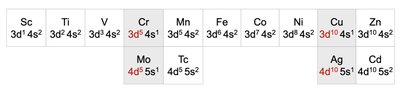
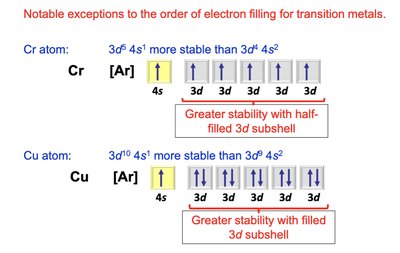
Transition Metals and Electron Configuration Anomalies
Transition metals often exhibit exceptions to the expected order of orbital filling, due to extra stability from half-filled or fully-filled d subshells.
Example: Chromium (Cr) and Copper (Cu) have anomalous configurations.

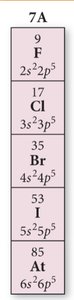

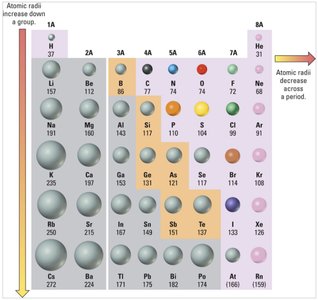
Periodic Trends: Atomic Radii
Atomic radius decreases across a period (left to right) and increases down a group (top to bottom). Cations are smaller than their neutral atoms, while anions are larger.
Trend: Decrease across period, increase down group.
Cations: Smaller due to loss of electrons.
Anions: Larger due to gain of electrons.
Ionic Compounds: Periodic Trends and Bonding Theory
Ionic Bonding and Classification of Elements
Ionic bonds form between metals and nonmetals, involving the transfer of electrons from the metal to the nonmetal. Elements are classified as main group metals, transition metals, metalloids, and nonmetals.
Electronic Configurations of Ions
Ion formation involves removing electrons for cations and adding electrons for anions, often resulting in a noble gas configuration.
Isoelectronic: Ions with the same number of electrons as a noble gas.
Transition Metal Cations: ns electrons are lost first.
Paramagnetism and Diamagnetism
Atoms with unpaired electrons are paramagnetic (attracted to magnetic fields), while those with all paired electrons are diamagnetic (repelled by magnetic fields).
Periodic Trends: Ionization Energy and Electron Affinity
Ionization energy is the energy required to remove an electron from a gas-phase atom. Electron affinity is the energy change when an electron is added to a gas-phase atom.
Ionization Energy: Increases across a period, decreases down a group.
Electron Affinity: Usually negative (favorable), increases across a period.
The Octet Rule and Stoichiometry of Ionic Compounds
Main group elements tend to gain or lose electrons to achieve eight valence electrons (octet rule), predicting ionic charges and chemical formulas.
Examples: NaCl, Na2S, CaCl2, Al2O3
Energy in Ionic Compound Formation and Crystal Lattice
Ionic compounds form stable crystal lattices, releasing large amounts of energy. The Born-Haber cycle is used to calculate lattice energies.
Lattice Energy (U): Energy required to break up an ionic solid into gaseous ions.
Coulomb's Law:
Trend: Larger ion size decreases lattice energy; higher ion charge increases lattice energy.
Covalent Bonding and Electron-Dot Structures
Lewis Bonding Theory
Lewis theory explains bonding by emphasizing valence electrons. Lewis dot structures model the arrangement of electrons in molecules.
Bond Types: Ionic (transfer), covalent (sharing), metallic (pooling).
Properties of Covalent Bonds
Covalent bonds involve shared electron pairs. Bond length and strength depend on the number of shared electrons and atomic size.
Bond Length: Shorter bonds are stronger; multiple bonds are shorter than single bonds.
Bond Energy: Energy required to break a bond; stronger bonds have higher bond energies.
Bond Polarity and Electronegativity
Bond polarity arises from unequal sharing of electrons. Electronegativity measures an atom's ability to attract shared electrons.
Electronegativity Trend: Increases across a period, decreases down a group.
Dipole Moment:
Electron Dot Structures and the Octet Rule
Electron dot structures show valence electrons as dots. The octet rule guides the formation of stable covalent bonds.
Single, Double, Triple Bonds: Atoms share one, two, or three pairs of electrons.
Formal Charge: Used to determine the most stable Lewis structure.
Table: Types of Chemical Bonds
Types of Atoms | Type of Bond | Characteristic |
|---|---|---|
Metal and nonmetal | Ionic | Electrons transferred |
Nonmetal and nonmetal | Covalent | Electrons shared |
Metal and metal | Metallic | Electrons pooled |
Table: Maximum Number of Electrons in Subshells
Subshell | Number of Orbitals | Maximum Electrons |
|---|---|---|
s | 1 | 2 |
p | 3 | 6 |
d | 5 | 10 |
f | 7 | 14 |
Table: Lattice Energy Trends for Metal Chlorides
Metal Ion | Lattice Energy (kJ/mol) |
|---|---|
Li+ | 834 |
Na+ | 788 |
K+ | 701 |
Cs+ | 657 |
Table: Effect of Ionic Charge on Lattice Energy
Anion | Na+ | Mg2+ | Al3+ |
|---|---|---|---|
OH- | 900 | 3006 | 5627 |
O2- | 2481 | 3791 | 15916 |
Table: Expanded and Condensed Electron Configurations
Element | Expanded | Condensed |
|---|---|---|
H | 1s1 | 1s1 |
He | 1s2 | 1s2 |
Li | 1s22s1 | 1s22s1 |
Be | 1s22s2 | 1s22s2 |
B | 1s22s22p1 | 1s22s22p1 |
C | 1s22s22p2 | 1s22s22p2 |
N | 1s22s22p3 | 1s22s22p3 |
O | 1s22s22p4 | 1s22s22p4 |
F | 1s22s22p5 | 1s22s22p5 |
Ne | 1s22s22p6 | 1s22s22p6 |
Example:
Bond Energy Calculation: For CH4 + 2 O2 → CO2 + 2 H2O, sum the energies of bonds broken and formed to estimate ΔE.
Additional info: Academic context was added to clarify quantum numbers, periodic trends, and the Born-Haber cycle for lattice energy calculation.
