 Back
BackL12: Phase Changes and Thermodynamics of Phase Transitions
Study Guide - Smart Notes

Phase Changes and Intermolecular Forces
Overview of Phase Changes
Phase changes are physical transformations between the solid, liquid, and gas states of matter. These transitions are driven by the addition or removal of energy, which affects the kinetic energy of particles and the strength of intermolecular forces (IMFs) holding them together.
Melting (Fusion): Solid to liquid, requires energy input.
Freezing: Liquid to solid, releases energy.
Vaporization (Boiling/Evaporation): Liquid to gas, requires energy input.
Condensation: Gas to liquid, releases energy.
Sublimation: Solid to gas, requires energy input.
Deposition: Gas to solid, releases energy.
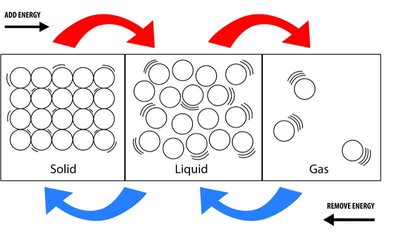
Properties of Phases
The three main phases of matter differ in compressibility, density, particle motion, and the strength of intermolecular forces:
Phase | Compressibility | Density | Particle Motion | IMF Strength |
|---|---|---|---|---|
Solid | Not compressible | High | Fixed positions | Strongest |
Liquid | Not compressible | High | Relative motion | Intermediate |
Gas | Compressible | Low | Free motion | Weakest |
Intermolecular Forces and Phase Behavior
Types of Intermolecular Forces (IMFs)
IMFs are the forces that hold molecules together and determine the physical properties of substances. The main types are:
Dispersion (London) Forces: Present in all molecules; strength increases with molecular size and shape.
Dipole-Dipole Forces: Occur in polar molecules; strength increases with molecular polarity.
Hydrogen Bonding: Strongest IMF, occurs when H is bonded to N, O, or F.
Order of strength: Hydrogen Bonding > Dipole-Dipole > Dispersion
Effect of IMFs on Melting and Boiling Points
Stronger IMFs result in higher melting and boiling points because more energy is required to overcome these forces.
Melting Point (Tmp): Temperature where solid becomes liquid.
Boiling Point (Tbp): Temperature where liquid becomes gas.
Sublimation Point: Temperature where solid becomes gas directly.
Vapor Pressure and Boiling
Vapor Pressure
Vapor pressure (Pvap) is the pressure exerted by a vapor in equilibrium with its liquid at a given temperature. It increases with temperature as more molecules have enough kinetic energy to escape the liquid phase.
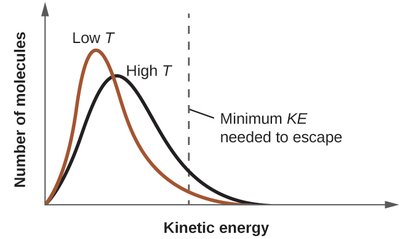
Vapor Pressure Trends
At higher temperatures, vapor pressure increases.
At a given temperature, substances with stronger IMFs have lower vapor pressures.
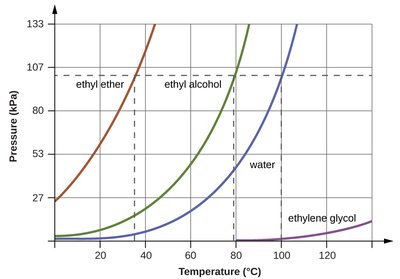
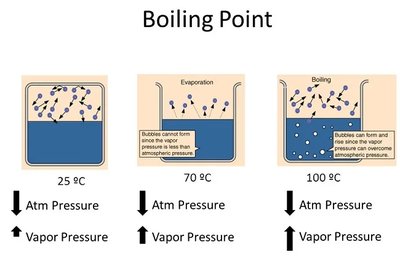
Boiling Point and Vapor Pressure
Boiling occurs when the vapor pressure equals the external (atmospheric) pressure. The normal boiling point is defined as the temperature at which vapor pressure is 1 atm.
Clausius-Clapeyron Equation
Quantitative Relationship
The Clausius-Clapeyron equation relates vapor pressure and temperature for a substance:
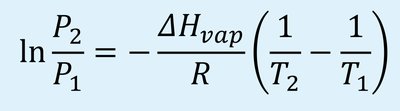
P1, P2: Vapor pressures at temperatures T1 and T2 (in Kelvin).
ΔHvap: Enthalpy of vaporization (J/mol).
R: Gas constant (8.3145 J/mol·K).
This equation allows calculation of vapor pressure at a new temperature if ΔHvap and a reference point are known.
Thermodynamics of Phase Changes
Enthalpy Changes
ΔHfus (Fusion): Energy required to melt 1 mole of solid to liquid (endothermic, positive value).
ΔHvap (Vaporization): Energy required to vaporize 1 mole of liquid to gas (endothermic, positive value).
ΔHsub (Sublimation): Energy required to convert 1 mole of solid to gas (endothermic, positive value).
Condensation, Freezing, Deposition: Opposite processes, same magnitude but negative sign (exothermic).
Calculating Heat for Phase Changes
The heat (q) required for a phase change is:
n: Number of moles of substance.
ΔHphase change: Enthalpy change for the specific transition.
Heating Curves
Heating curves show temperature changes as heat is added to a substance. There are plateaus at phase change temperatures where energy goes into changing phase, not raising temperature.
During a phase change, temperature remains constant until the transition is complete.
Between phase changes, temperature increases according to (where c is specific heat).
Summary Table: Enthalpy Changes for Phase Transitions
Process | Direction | ΔH sign | Energy Flow |
|---|---|---|---|
Melting (Fusion) | Solid → Liquid | Positive | Absorbed |
Freezing | Liquid → Solid | Negative | Released |
Vaporization | Liquid → Gas | Positive | Absorbed |
Condensation | Gas → Liquid | Negative | Released |
Sublimation | Solid → Gas | Positive | Absorbed |
Deposition | Gas → Solid | Negative | Released |
