 Back
BackPhase Equilibria, Vapor Pressure, and Chemical Equilibrium: Study Guide
Study Guide - Smart Notes

Topic 5A: Vapor Pressure
Origin and Characterization of Vapor Pressure
Vapor pressure is the pressure exerted by a vapor in equilibrium with its liquid (or solid) phase at a given temperature. It is a fundamental property that reflects the tendency of molecules to escape from the liquid phase into the gas phase.
Dynamic Equilibrium: In a closed system, the rate of evaporation equals the rate of condensation, resulting in a constant vapor pressure.
Boiling Point: The temperature at which the vapor pressure equals atmospheric pressure (1 atm).
Intermolecular Forces (IMF): Stronger IMFs result in lower vapor pressures and higher boiling points.
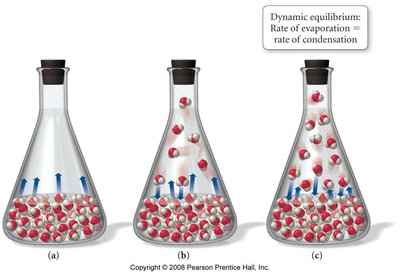
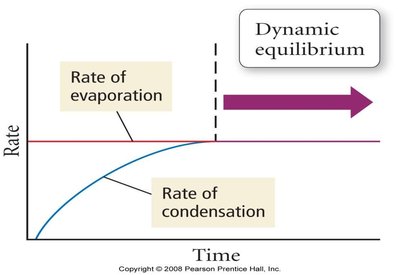
Variation of Vapor Pressure with Temperature
Vapor pressure increases with temperature as more molecules have sufficient kinetic energy to escape the liquid phase.
Boltzmann Distribution: At higher temperatures, a greater fraction of molecules have enough energy to overcome IMFs.
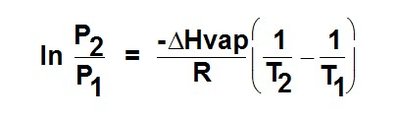
Clausius-Clapeyron Equation: Describes the relationship between vapor pressure and temperature:
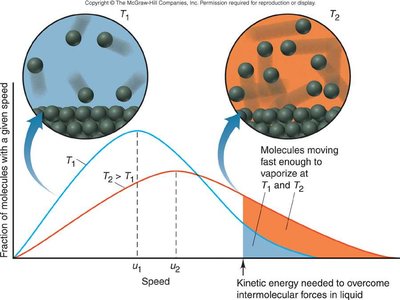
Ranking Boiling Points by IMF
Boiling points can be ranked based on the strength of intermolecular forces present in the molecules:
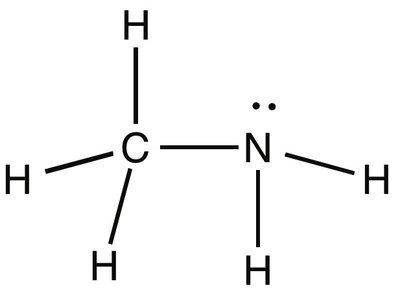
Hydrogen Bonding: Highest boiling point (e.g., CH3NH2)
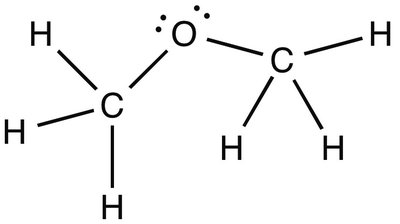
Dipole-Dipole: Intermediate boiling point (e.g., CH3OCH3)
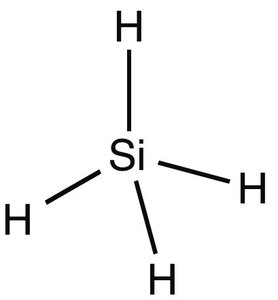
London Dispersion: Lowest boiling point (e.g., Ne, CH4, SiH4)

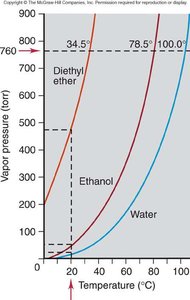
Clausius-Clapeyron Equation and Applications
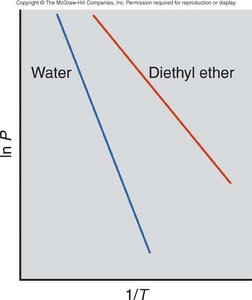
The Clausius-Clapeyron equation allows calculation of vapor pressure at different temperatures and comparison of enthalpy of vaporization () for different substances.
Example: Water has a higher and lower vapor pressure than diethyl ether at the same temperature.
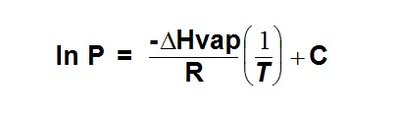
Topic 5B: Phase Equilibria in One Component Systems
Phase Diagrams and Key Points
Phase diagrams show the regions of stability for solid, liquid, and gas phases as a function of temperature and pressure.
Triple Point: The unique set of conditions where all three phases coexist.
Critical Point: The endpoint of the liquid-gas boundary; above this, the substance is a supercritical fluid.
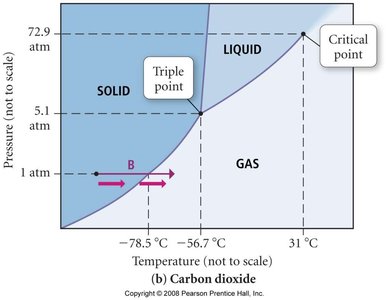
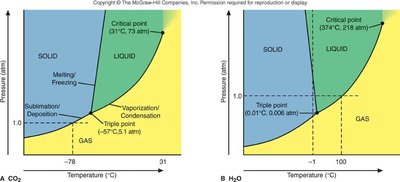
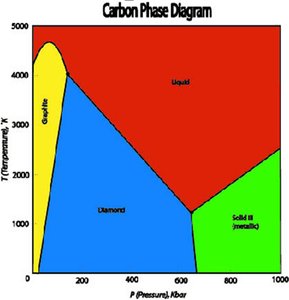
Phase Diagrams for Carbon
Carbon exhibits multiple solid phases, including graphite and diamond. Graphite is the most stable at standard conditions, while diamond requires high pressure to form.
Supercritical Fluids
Above the critical temperature and pressure, a substance forms a supercritical fluid, which has properties of both liquids and gases and can act as a solvent for various materials.

Topic 5C: Phase Equilibria in Two Component Systems
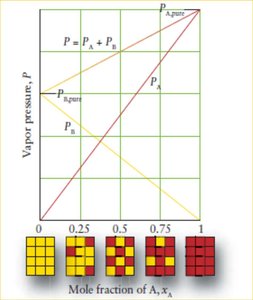
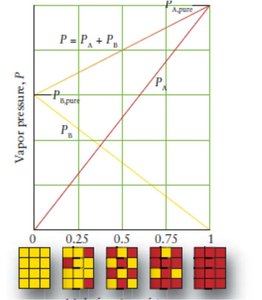
Raoult’s Law and Vapor Pressure of Mixtures
In mixtures, the vapor pressure of each component is proportional to its mole fraction. The total vapor pressure is the sum of the partial pressures.
Raoult’s Law: ,
Ideal Solutions: Obey Raoult’s Law exactly.
Non-Ideal Solutions: Show positive or negative deviations due to intermolecular interactions.

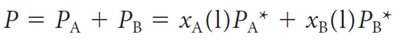
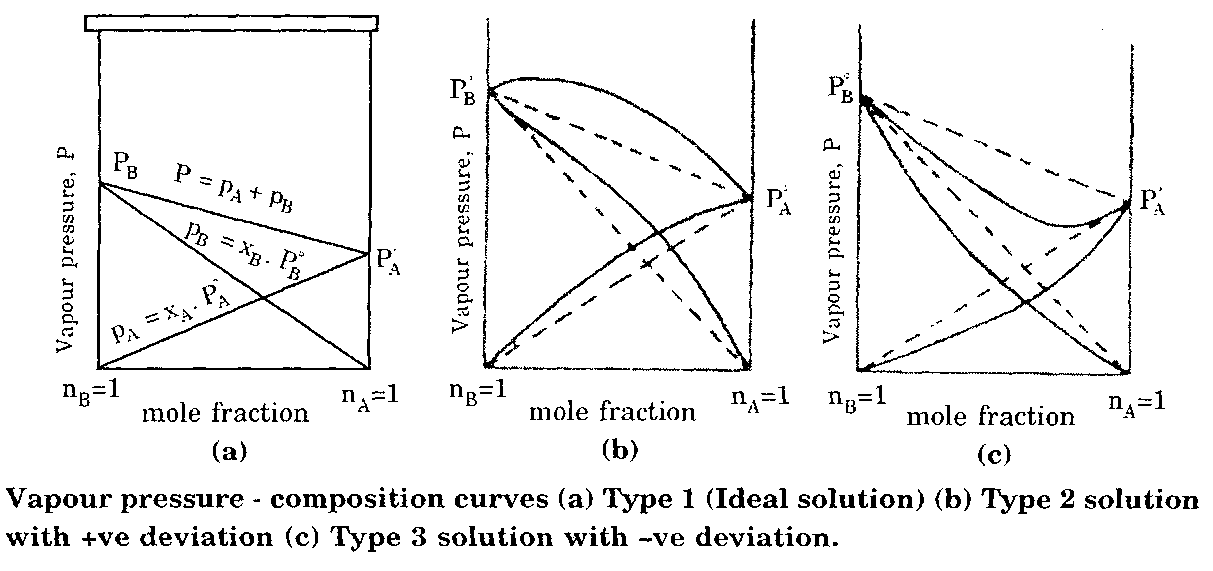
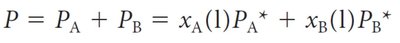
Example Calculation
To calculate the vapor pressure of a solution:
Find moles of solute and solvent.
Calculate mole fraction of solvent.
Apply Raoult’s Law:

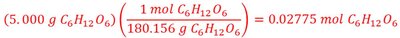

Topic 5D: Solubility
Solubility Principles
Solubility is determined by the compatibility of intermolecular forces between solute and solvent. "Like dissolves like" is a guiding principle.
Polar solutes: Dissolve in polar solvents (e.g., NaCl in water).
Nonpolar solutes: Dissolve in nonpolar solvents (e.g., vitamin A in fats).
Gas solubility: Proportional to partial pressure (Henry’s Law: ).
Topic 5E: Molality
Definition and Properties
Molality () is defined as moles of solute per kilogram of solvent. It is temperature independent, making it useful for colligative property calculations.
Formula:
Comparison: Molarity depends on volume, which changes with temperature; molality does not.
Topic 5F: Colligative Properties
Boiling Point Elevation and Freezing Point Depression
Colligative properties depend only on the number of solute particles, not their identity. Key properties include boiling point elevation, freezing point depression, and osmotic pressure.
Boiling Point Elevation:
Freezing Point Depression:
Van’t Hoff Factor (): Accounts for dissociation of electrolytes.
Osmotic Pressure
Osmosis is the movement of solvent through a semipermeable membrane. Osmotic pressure () is the pressure required to stop this flow.
Formula:
Topic 5G: Chemical Equilibrium
Dynamic Equilibrium and Equilibrium Constant
Chemical equilibrium occurs when the forward and reverse reaction rates are equal, and concentrations of reactants and products remain constant.
Equilibrium Constant (): Ratio of product to reactant concentrations at equilibrium, raised to their stoichiometric coefficients.
Law of Mass Action:
Topic 5H: Alternative Forms of the Equilibrium Constant
Conversion between and
For reactions involving gases, equilibrium constants can be expressed in terms of concentrations () or partial pressures ().
Relationship: , where is the change in moles of gas.
Topic 5I: Equilibrium Calculations
Using Equilibrium Tables
Equilibrium tables (ICE tables: Initial, Change, Equilibrium) are used to calculate concentrations at equilibrium given initial conditions and .
Quadratic Equations: Sometimes required for solving equilibrium concentrations.
Topic 5J: Response of Equilibria to Changes in Conditions (Le Châtelier’s Principle)
Le Châtelier’s Principle
If a system at equilibrium is disturbed by a change in concentration, pressure, or temperature, it will shift to partially offset the disturbance.
Concentration: Adding reactants shifts equilibrium toward products.
Pressure/Volume: Increasing pressure favors the side with fewer moles of gas.
Temperature: Increasing temperature favors endothermic direction.
Catalysts: Speed up attainment of equilibrium but do not affect position or .
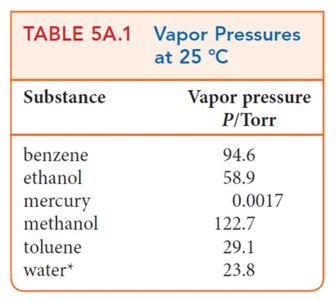
Example Table: Vapor Pressures at 25°C
Substance | Vapor Pressure (Torr) |
|---|---|
Benzene | 94.6 |
Ethanol | 58.9 |
Mercury | 0.0017 |
Methanol | 122.7 |
Toluene | 29.1 |
Water | 23.8 |
Example: Calculate the vapor pressure of water at 90°C for a solution prepared by dissolving 5.000 g of glucose in 100.0 g H2O. The vapor pressure of pure water at 90°C is 524 torr.
Find moles of solute and solvent.
Calculate mole fraction of solvent.
Apply Raoult’s Law:
Summary: This guide covers the essential concepts of phase equilibria, vapor pressure, solubility, colligative properties, and chemical equilibrium, providing definitions, equations, and examples for each topic.
