 Back
BackPhysical Properties of Solutions – General Chemistry Chapter 13 Study Notes
Study Guide - Smart Notes

Physical Properties of Solutions
Introduction to Solutions
A solution is a homogeneous mixture composed of two or more substances. The component present in the largest amount is called the solvent, while the component present in the smaller amount is called the solute. Understanding the nature of solutions is fundamental in chemistry, as many chemical reactions occur in solution.
Solvent: Substance in greatest quantity in a solution.
Solute: Substance dissolved in the solvent.
Example: Dissolving sugar in water forms a sugar solution.
Types of Solutions: Saturated, Unsaturated, Supersaturated
The solubility of a solute is the maximum amount that can dissolve in a given quantity of solvent at a specific temperature. Solutions can be classified based on the amount of solute dissolved:
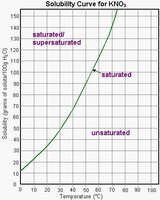
Saturated solution: Contains the maximum amount of solute that can dissolve at a given temperature.
Unsaturated solution: Contains less than the maximum amount of solute.
Supersaturated solution: Contains more than the maximum amount of solute; unstable and can precipitate excess solute.



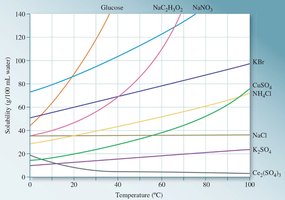
Solubility Curves and Temperature Effects
Solubility curves graphically represent how the solubility of a substance changes with temperature. For most solids, solubility increases with temperature, while for gases, solubility decreases as temperature rises.
Example: The solubility curve for potassium nitrate (KNO3) shows increased solubility with higher temperature.

Intermolecular Forces and Solubility
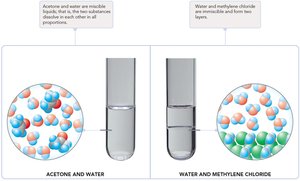
The ability of a solute to dissolve in a solvent depends on the similarity of their intermolecular forces (IMFs). The rule "like dissolves like" means that polar solutes dissolve in polar solvents, and nonpolar solutes dissolve in nonpolar solvents.
Miscible: Liquids that mix in all proportions (similar IMFs).
Immiscible: Liquids that do not mix (different IMFs).
Example: Oil (nonpolar) and water (polar) are immiscible.

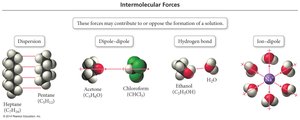
Factors Affecting Solubility: Pressure and Henry's Law
Pressure significantly affects the solubility of gases in liquids. According to Henry's Law, the solubility of a gas is directly proportional to the pressure of the gas above the solution.
Henry's Law equation:
Where: C = molar concentration (mol/L) P = pressure (atm) k = Henry's law constant (mol/(L·atm))
Concentration Units and Calculations
Concentration expresses the amount of solute in a given amount of solution or solvent. Several units are used in chemistry:
Unit | Definition | Units |
|---|---|---|
Molarity (M) | amount solute (mol) / volume solution (L) | mol/L |
Molality (m) | amount solute (mol) / mass solvent (kg) | mol/kg |
Mole fraction (χ) | amount solute (mol) / total amount (mol) | None |
Parts by mass | mass solute / mass solution × multiplication factor | ppm, ppb |
Percent by mass (%) | mass solute / mass solution × 100 | % |

Colligative Properties of Solutions
Colligative properties depend on the number of solute particles in solution, not their identity. The main colligative properties are:
Vapor pressure lowering (ΔP)
Boiling point elevation (ΔTb)
Freezing point depression (ΔTf)
Osmotic pressure (π)
Vapor Pressure Lowering and Raoult's Law
The addition of a nonvolatile solute to a solvent lowers the vapor pressure of the solution. This is described by Raoult's Law:
P = vapor pressure of solution
P∘ = vapor pressure of pure solvent
χ = mole fraction
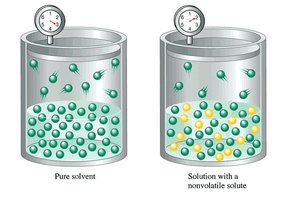
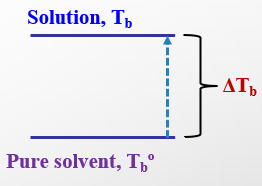
Boiling Point Elevation and Freezing Point Depression
Adding solute to a solvent increases the boiling point and decreases the freezing point. The magnitude of these changes is given by:
Freezing point depression:
Boiling point elevation:
m = molality
Kf = freezing point depression constant
Kb = boiling point elevation constant
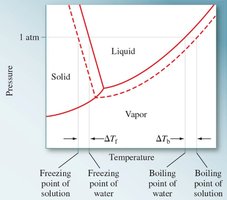
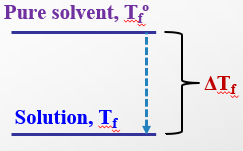
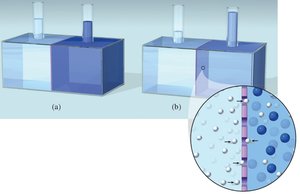
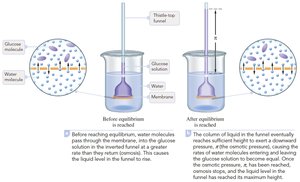
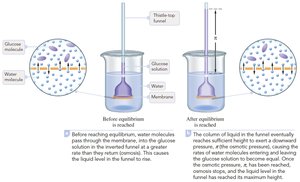
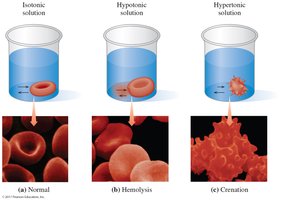
Osmosis and Osmotic Pressure
Osmosis is the movement of solvent molecules through a semipermeable membrane from a region of lower solute concentration to higher solute concentration. Osmotic pressure (π) is the pressure required to stop this flow.
Osmotic pressure equation:
M = molarity
R = gas constant (0.08206 L·atm/(mol·K))
T = temperature (K)
Electrolyte Solutions and the van't Hoff Factor
Electrolytes dissociate in water, producing more particles per formula unit. The van't Hoff factor (i) accounts for the number of particles produced and is incorporated into colligative property equations:
For ideal solutions, use the theoretical van't Hoff factor based on the number of ions produced per formula unit.
Practice Problems and Applications
To master these concepts, practice problems involving solution preparation, concentration calculations, and colligative property effects are essential. Refer to the recommended end-of-chapter problems for further study.
Additional info: These notes cover all major aspects of Chapter 13: Physical Properties of Solutions, including definitions, equations, and practical examples. Images were included only when directly relevant to the explanation and reinforce key concepts.
