 Back
BackPhysical Properties of Solutions: Structure, Formation, and Concentration
Study Guide - Smart Notes

Physical Properties of Solutions
Definition and Components of Solutions
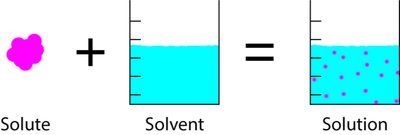
A solution is a homogeneous mixture composed of two or more substances. The substance present in the smaller amount is called the solute, while the substance present in the larger amount is called the solvent. When water is the solvent, the solution is referred to as an aqueous solution.
Solute: The component dissolved in the solvent.
Solvent: The medium in which the solute is dissolved.
Solution: The resulting mixture after the solute is dissolved in the solvent.
Solubility: The maximum amount of solute that can be dissolved in a given amount of solvent at equilibrium.
Example: Properties of Glucose
Glucose is a common solute in aqueous solutions. Its physical and thermochemical properties are important for understanding its behavior in solution.
Property | Value |
|---|---|
Molecular formula | C6H12O6 |
Molar mass | 180.16 g mol-1 |
Appearance | White powder |
Density | 1.54 g/cm3 |
Melting point | α-D-glucose: 146 °C, β-D-glucose: 150 °C |
Solubility in water | 909 g/L (25 °C) |
Specific heat capacity | 218.6 J K-1 mol-1 |
Std molar entropy | 209.2 J K-1 mol-1 |
Std enthalpy of formation | -1271 kJ/mol |
Std enthalpy of combustion | -2805 kJ/mol |

Role of Entropy in Solution Formation
Mixing and Entropy
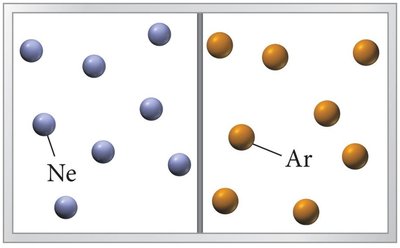
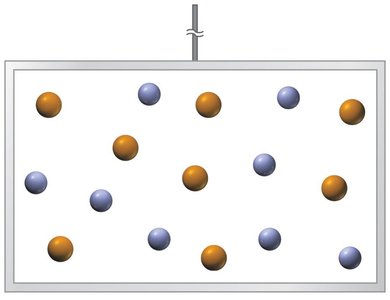
Entropy is a statistical phenomenon that describes the spreading out of energy in a system. In mixtures of non-interacting particles, such as ideal gases, mixing occurs spontaneously due to the increase in entropy.
Entropy: A measure of disorder or randomness in a system.
Mixing increases entropy, favoring solution formation.
Intermolecular Forces and Solution Formation
Types of Intermolecular Forces
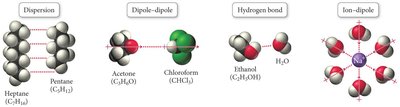
The formation of a solution depends on the relative strength of intermolecular forces between solute and solvent particles. The "Like Dissolves Like" rule states that substances with similar intermolecular forces are likely to be soluble in each other.
Dispersion forces: Present in non-polar molecules (e.g., heptane and pentane).
Dipole-dipole forces: Present in polar molecules (e.g., acetone and chloroform).
Hydrogen bonding: Strong interaction in molecules like ethanol and water.
Ion-dipole forces: Occur between ionic compounds and polar solvents (e.g., NaCl in water).
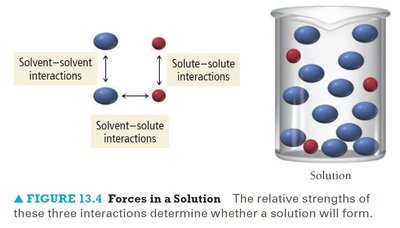
Forces in a Solution
Three types of interactions are important in solution formation: solvent-solvent, solute-solute, and solvent-solute interactions. The relative strengths of these interactions determine whether a solution will form.
Solvent-solvent interactions: Forces between solvent molecules.
Solute-solute interactions: Forces between solute molecules.
Solvent-solute interactions: Forces between solvent and solute molecules.
Relative Interactions and Solution Formation
The likelihood of solution formation can be summarized in the following table:
Solvent-solute interactions | Solvent-solvent and solute-solute interactions | Outcome |
|---|---|---|
> | Solution generally forms | |
= | Solution generally forms | |
< | Solution may or may not form, depending on disparity |
Solubility and Structure
The solubility of a substance is influenced by its molecular structure. Hydrophilic molecules (e.g., vitamin C) dissolve well in water, while hydrophobic molecules (e.g., vitamin A) dissolve well in nonpolar solvents.
Hydrophilic: Water-loving, polar, dissolves in water.
Hydrophobic: Water-fearing, nonpolar, dissolves in nonpolar solvents.
Molecular View of Dissolution
Steps in Solution Formation
The process of dissolution involves three steps, each associated with an enthalpy change:
Step 1: Separation of solvent molecules () - endothermic.
Step 2: Separation of solute molecules () - endothermic.
Step 3: Mixing of solute and solvent () - exothermic.
Total enthalpy change:
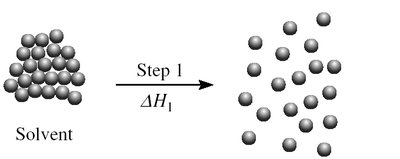
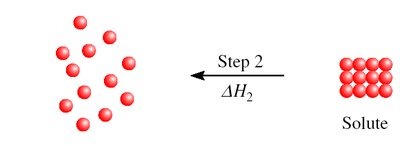
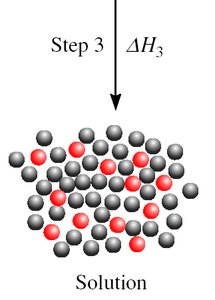
Enthalpy of Dissolution
Enthalpy Changes in Solution Formation
The enthalpy change when one mole of a substance is dissolved in excess solvent is called the enthalpy of solution (). For ionic compounds, the enthalpy of hydration () is always large and exothermic.
Endothermic if
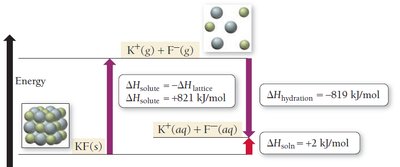
Solution Equilibrium
Saturated, Unsaturated, and Supersaturated Solutions
When dissolution and re-crystallization rates are balanced, a dynamic equilibrium is achieved, and the solution is saturated. An unsaturated solution contains less solute than the solvent can dissolve, while a supersaturated solution contains more solute than is present in a saturated solution.
Saturated solution: Maximum solute dissolved at equilibrium.
Unsaturated solution: Less solute than capacity.
Supersaturated solution: More solute than saturation, unstable.



Supersaturated Solutions and Nucleation
Supersaturated solutions are typically prepared by dissolving more solute at higher temperatures and then cooling. The solid remains in solution due to an energy barrier called the nucleation energy barrier. Adding a seed crystal removes this barrier and allows the solid to precipitate.
Nucleation: Process of forming solid from solution.
Supersaturated solutions are unstable and can rapidly crystallize.

Factors Affecting Solubility
Temperature and Pressure
The solubility of most solid substances in liquid solvents increases with temperature, while the solubility of gases in liquids generally decreases with temperature. Pressure strongly affects the solubility of gases in liquids, as described by Henry's law.
Henry's law:
= equilibrium concentration of gas (mol L-1)
= Henry's constant (mol L-1 atm-1)
= partial pressure of gas (atm)


Concentration Units
Molarity
Molarity (M) is the most common unit of concentration, defined as moles of solute per liter of solution.
1.0 M = 1.0 mol/L
Low concentration: dilute; high concentration: concentrated
Molality
Molality (m) is defined as moles of solute per kilogram of solvent. It is useful when temperature changes, as it is independent of volume.
Example: Dissolving 19.8 g NaCl in 245 g water yields m
Conversion Between Molality and Molarity
Conversion requires the density of the solution. For example, a 1.16 M solution of sucrose with density 1.18 g/mL has a molality of 1.48 m.
Calculate moles of solute and mass of solvent per liter.
Mole Fraction
Mole fraction (X) is a dimensionless measure of concentration, defined as the ratio of moles of a component to total moles in solution.
Ranges from 0 to 1
Weight Fraction
Weight fraction (w) is commonly used in analytical chemistry, defined as the mass of a component divided by the total mass of the solution.
Expressed as % or ppm
w = 1% means 0.01 g solute in 1 g solution
w = 1 ppm means g solute in 1 g solution
Summary
Understanding the physical properties of solutions, including their formation, equilibrium, and concentration units, is essential in general chemistry. The interplay of intermolecular forces, entropy, and enthalpy governs whether solutions form and how solutes behave in solvents. Temperature and pressure are key factors affecting solubility, especially for gases. Accurate measurement of concentration is fundamental for laboratory and analytical work.
