 Back
BackPredicting Chemical Change: Energetic and Entropic Factors in Reaction Directionality
Study Guide - Smart Notes

Chemical Equilibrium and Reaction Directionality
Introduction to Chemical Change
Chemical reactions can be predicted and analyzed by considering their directionality and extent. The likelihood of a reaction proceeding to products or remaining as reactants is determined by thermodynamic and kinetic factors.
Thermodynamics governs the directionality and extent of reactions.
Kinetics determines the mechanism and rate of reactions.
Central Goal: Predicting Reaction Directionality
The main objective is to qualitatively reason whether a chemical reaction is product-favored or reactant-favored at equilibrium. This involves understanding the factors that influence equilibrium position.
Product-favored reactions yield more products at equilibrium.
Reactant-favored reactions retain more reactants at equilibrium.
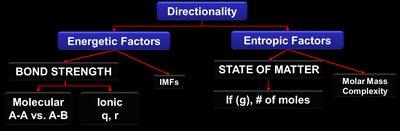
Factors Affecting Reaction Directionality
Energetic Factors
Energetic stability is determined by the potential energy of reactants and products. Lower potential energy corresponds to greater stability.
Bond Strength: Stronger bonds (shorter, more polar) result in lower potential energy and greater stability.
Intermolecular Forces (IMFs): Stronger IMFs (e.g., hydrogen bonding, dipole-dipole, dispersion) lower the potential energy of substances.
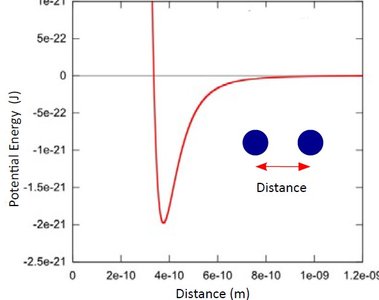
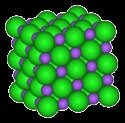
Ionic Compounds: Stability increases with higher ionic charge and smaller ionic radius, as described by Coulomb's Law:
Bond Energies and Reaction Energetics
The enthalpy change () of a reaction is calculated using bond energies:
Exothermic reactions () are energetically favored.
Endothermic reactions () are less favored.
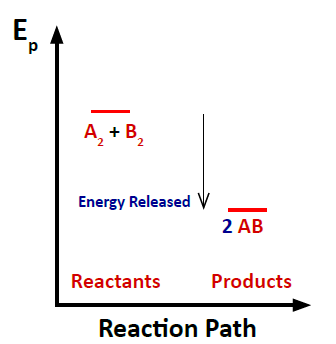
Example: Double Bonds
Double bonds consist of a sigma and a pi bond. The pi bond is weaker and more easily broken. 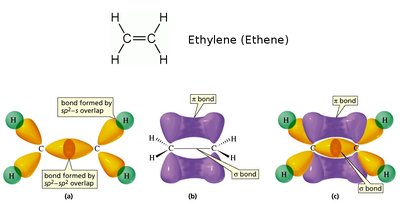
Entropic Factors
Entropic stability is determined by the number of configurations (microstates) available to the system.
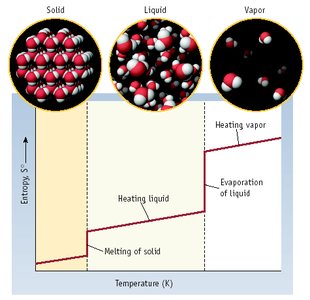
Entropy (S): A measure of disorder or the number of possible configurations.
State of Matter: Gases have higher entropy than liquids, which have higher entropy than solids.
Molar Mass and Complexity: Larger and more complex molecules have higher entropy.
Number of Particles: More moles of gas increase entropy.
Standard Molar Entropy
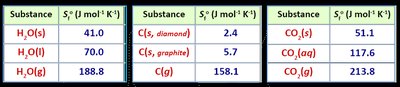
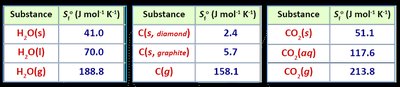
Standard molar entropy () is the entropy of one mole of a substance at 25°C and 1 atm.
Measured experimentally.
Increases with state changes and molecular complexity.
Comparing Energetic and Entropic Factors
Competition Between Enthalpy and Entropy
The net directionality of a reaction depends on the balance between enthalpy and entropy changes.
Lower enthalpy (more negative ) favors product formation.
Higher entropy (more positive ) favors product formation.
Sometimes, energetic and entropic factors compete, and the outcome depends on their relative magnitudes.
Summary Table: Factors Affecting Reaction Directionality
Factor | Effect on Stability | Example |
|---|---|---|
Bond Strength | Stronger bonds = lower Ep = more stable | H2O(l) vs O2(g) |
IMFs | Stronger IMFs = lower Ep = more stable | H2O(l) vs H2O(g) |
Ionic Charge/Size | Higher charge/smaller size = lower Ep = more stable | MgO vs Na2O |
State of Matter | Gas > Liquid > Solid (entropy) | CO2(g) vs CO2(s) |
Molar Mass/Complexity | Higher mass/complexity = higher S | Cl2(g) vs H2(g) |
Number of Particles | More moles of gas = higher S | 2 mol CO2(g) vs 1 mol CO2(g) |
Key Equations
Potential Energy (Coulomb's Law):
Entropy:
Enthalpy Change:
Conclusion
Predicting chemical change requires evaluating both energetic and entropic factors. Product-favored reactions typically release energy and increase entropy, but the balance between these factors determines the equilibrium position. Understanding these principles is essential for analyzing and predicting chemical reactions in general chemistry.
