 Back
BackPredicting Precipitation Using Ksp and Solution Mixing
Study Guide - Smart Notes

Q1. Does a precipitate form when 0.10 L of M Pb(NO) is added to 0.40 L of M Na$_2$SO?
Background
Topic: Solubility Product (Ksp) and Precipitation
This question tests your ability to predict whether a precipitate will form when two solutions are mixed, based on the concentrations of ions and the solubility product constant (Ksp) for the possible precipitate.
Key Terms and Formulas
Ksp (Solubility Product Constant): The equilibrium constant for the dissolution of a sparingly soluble salt.
Q (Reaction Quotient): Calculated using the concentrations of ions after mixing. Compare Q to Ksp to predict precipitation.
Key formula:
If , a precipitate forms. If , no precipitate forms.
Step-by-Step Guidance
Calculate the total volume after mixing: .
Find the moles of Pb and SO from each solution:
Moles Pb:
Moles SO:
Calculate the concentrations of Pb and SO after mixing, using the total volume:
Set up the expression for using the calculated concentrations:
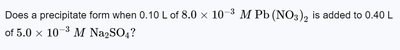
Try solving on your own before revealing the answer!
