 Back
BackPredicting Precipitation Using Ksp and Solution Mixing
Study Guide - Smart Notes

Q1. Does a precipitate form when 0.10 L of M Pb(NO) is added to 0.40 L of M Na$_2$SO?
Background
Topic: Solubility Product (Ksp) and Precipitation
This question tests your ability to predict whether a precipitate will form when two solutions are mixed, based on the concentrations of ions and the solubility product constant (Ksp) for the possible precipitate.
Key Terms and Formulas
Ksp: The solubility product constant, which represents the maximum product of ion concentrations that can exist in solution without forming a precipitate.
Q: The reaction quotient, calculated from the actual concentrations of ions after mixing. If , a precipitate forms.
Key formula:
Step-by-Step Guidance
Calculate the total volume after mixing: .
Find the new concentration of Pb after mixing, using dilution:
Find the new concentration of SO after mixing, using dilution:
Calculate using the formula with the concentrations you just found.
Try solving on your own before revealing the answer!
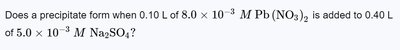
Final Answer: A precipitate will form if for PbSO.
After calculating the concentrations and , compare $Q$ to the known for PbSO (typically ). If $Q$ exceeds this value, a precipitate forms.
This approach uses dilution and the solubility product to predict precipitation.
