 Back
BackPrinciples of Chemical Equilibrium: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Principles of Chemical Equilibrium
Introduction to Chemical Equilibrium
Chemical equilibrium occurs when the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products over time. This concept is fundamental in understanding reversible reactions and their behavior in closed systems.
Dynamic Equilibrium: Both forward and reverse reactions continue to occur, but there is no net change in concentrations.
Equilibrium Expression: The ratio of product and reactant concentrations, each raised to the power of their stoichiometric coefficients, defines the equilibrium constant.
Example: The decomposition and formation of molecules over time illustrate the approach to equilibrium.
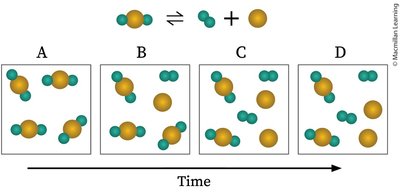
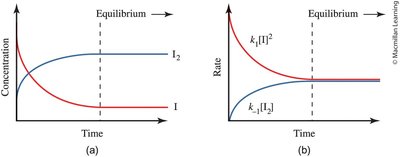
Concentration and Rate Changes During Equilibrium
As a reaction approaches equilibrium, the concentrations of reactants and products change until they stabilize. The rates of the forward and reverse reactions also adjust and become equal at equilibrium.
Concentration vs. Time: Reactant concentration decreases, product concentration increases, and both plateau at equilibrium.
Rate vs. Time: The rate of the forward reaction decreases, while the reverse reaction rate increases, until both are equal.
Equilibrium Constant Expressions
The equilibrium constant (K) quantifies the ratio of product to reactant concentrations at equilibrium. For a general reaction:
Expression: for A ⇌ B
Reverse Reaction:
Stoichiometric Variations: Changing the coefficients alters the equilibrium constant's form.
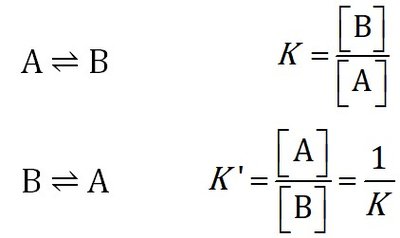
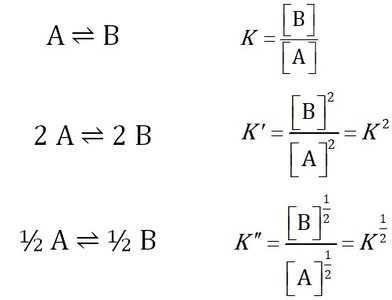
Combining Equilibrium Constants
When reactions are combined, their equilibrium constants multiply to give the overall equilibrium constant. This is useful for multi-step reactions.
Overall Equilibrium Constant:
Expression: for the combined reaction 2A ⇌ C

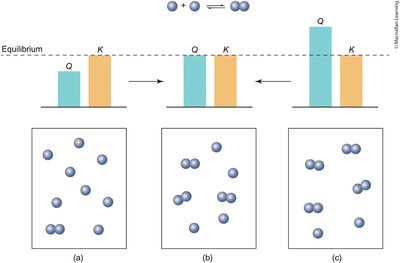
Reaction Quotient (Q) vs. Equilibrium Constant (K)
The reaction quotient (Q) is calculated using current concentrations, while K uses equilibrium concentrations. Comparing Q and K predicts the direction the reaction will shift to reach equilibrium.
If Q < K: Reaction shifts right (toward products).
If Q > K: Reaction shifts left (toward reactants).
If Q = K: System is at equilibrium.
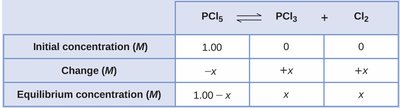
ICE Tables and Equilibrium Calculations
ICE tables (Initial, Change, Equilibrium) are used to organize concentration data and solve for equilibrium concentrations. This method is essential for quantitative equilibrium problems.
Setup: List initial concentrations, changes (using x), and equilibrium concentrations.
Example: For PCl5 ⇌ PCl3 + Cl2, the ICE table helps track changes and solve for x.
Solving Equilibrium Problems Using Quadratic Equations
Some equilibrium calculations require solving quadratic equations. The equilibrium constant expression is set up, and the quadratic formula is used to solve for unknown concentrations.
Equilibrium Expression:
Quadratic Setup:
Quadratic Formula:
Calculation: Substitute values and solve for x.
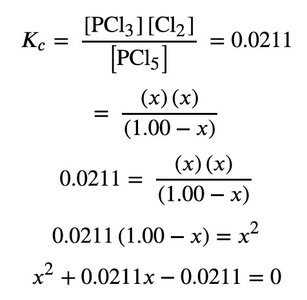
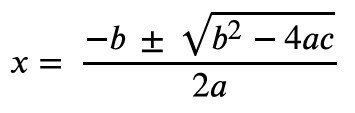
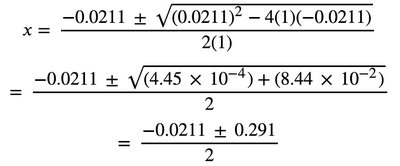
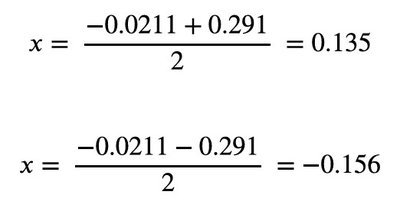
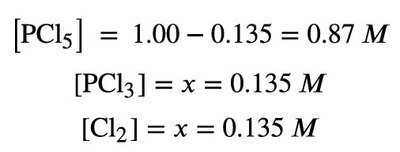
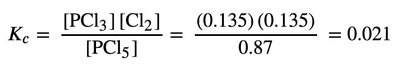
Equilibrium Calculations with Simplifying Assumptions
When the equilibrium constant is very small or large, simplifying assumptions can be made to avoid solving complex equations. This is checked by ensuring the change is less than 5% of the initial concentration.
Assumption: If x is small, (initial - x) ≈ initial.
Check: Validate the assumption by comparing x to the initial value.
Example: Formic acid ionization in water.

Le Châtelier's Principle: Effect of Volume and Pressure
Le Châtelier's Principle predicts how a system at equilibrium responds to changes in volume and pressure. The system shifts to counteract the change, favoring the side with fewer or more moles of gas as appropriate.
Decrease Volume: Increases pressure, shifts equilibrium to side with fewer moles of gas.
Increase Volume: Decreases pressure, shifts equilibrium to side with more moles of gas.
Table: Summarizes the effect of volume changes on equilibrium position.

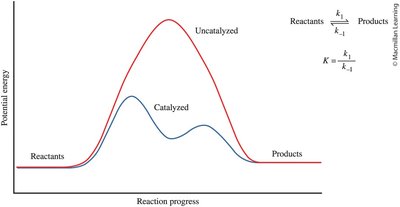
Effect of Catalysts on Equilibrium
Catalysts lower the activation energy for both forward and reverse reactions, allowing equilibrium to be reached faster. However, they do not change the equilibrium position or the equilibrium constant.
Potential Energy Diagram: Shows the effect of a catalyst on reaction progress.
Equilibrium Constant: Remains unchanged by the presence of a catalyst.
Worked Example: Volume Change and Equilibrium Shift
Applying Le Châtelier's Principle and ICE tables to a real equilibrium problem demonstrates how changes in volume affect equilibrium concentrations and the direction of the reaction.
Example: COCI2 decomposition in a sealed container.
Calculation: ICE table, equilibrium constant expression, and quadratic solution.
Result: Reaction shifts to the right, favoring products, as predicted by Le Châtelier's Principle.

