 Back
BackPrinciples of Chemical Equilibrium: Structured Study Notes
Study Guide - Smart Notes

Principles of Chemical Equilibrium
The Nature of the Equilibrium State
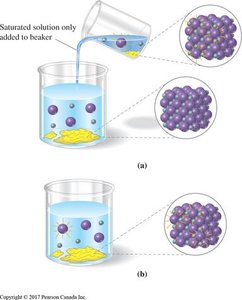
Chemical equilibrium occurs in a closed system at constant temperature when the rates of the forward and reverse reactions become equal, resulting in constant concentrations of reactants and products. The equilibrium state is dynamic, meaning both reactions continue to occur, but there is no net change in concentrations.
Reaction Quotient (Q): The ratio of product and reactant concentrations at any point in the reaction.
Equilibrium Constant (K): At equilibrium, Q becomes constant and is denoted as K, regardless of initial concentrations.
Dynamic Equilibrium: Forward and reverse reactions occur at equal rates.
Example: AgI(s) ↔ Ag+(aq) + I-(aq)
The Equilibrium Constant Expression
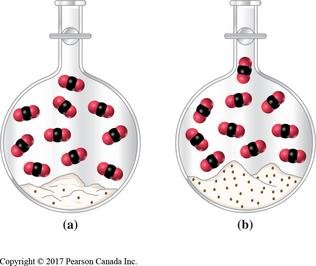
The equilibrium constant expression relates the concentrations (or partial pressures) of reactants and products at equilibrium for a given reaction.
General Form: For a reaction aA + bB ↔ cC + dD, the equilibrium constant is:
For Gases: Use partial pressures (KP).
For Solutions: Use concentrations (Kc).
Pure Solids and Liquids: Activities are set to 1 and are not included in the expression.
Activities of Substances
Activities are used in equilibrium expressions to account for non-ideal behavior. For most introductory purposes, activities are approximated as follows:
Substance | Activity | Comment |
|---|---|---|
Ideal gas, X(g) | PX is the partial pressure; P° = 1 bar | |
Pure solid or liquid | Activity is set to 1 | |
Solute in ideal aqueous solution | [X] is concentration; c° = 1 mol/L |

Relationships Involving Equilibrium Constants
Equilibrium constants change depending on how the chemical equation is manipulated:
Reversing the equation: Invert K ().
Multiplying coefficients: Raise K to the power of the factor.
Dividing coefficients: Take the root of K.
Combining reactions: Multiply the equilibrium constants.
Relationship Between KP and Kc
For reactions involving gases, KP and Kc are related by:
R: Gas constant
T: Temperature in Kelvin
Δngas: Change in moles of gas (products minus reactants)

The Magnitude of an Equilibrium Constant
The value of K indicates the extent of a reaction:
Large K (>1010): Reaction goes nearly to completion; products dominate.
Small K (<10-10): Reaction hardly occurs; reactants dominate.
Intermediate K: Significant amounts of both reactants and products are present.
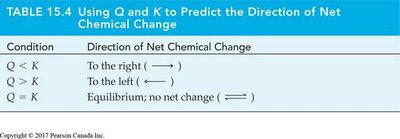
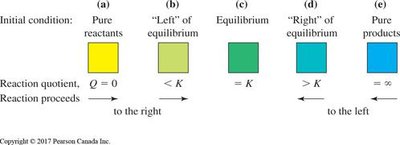
Predicting the Direction of Net Chemical Change
The reaction quotient Q is compared to K to predict the direction of change:
Condition | Direction of Net Chemical Change |
|---|---|
Q < K | To the right (forward reaction) |
Q > K | To the left (reverse reaction) |
Q = K | Equilibrium; no net change |
Altering Equilibrium Conditions: Le Châtelier’s Principle
Le Châtelier’s Principle states that if a system at equilibrium is disturbed by changing temperature, pressure, or concentration, the system will shift to counteract the disturbance and establish a new equilibrium.
Change in Concentration: Adding/removing reactants or products shifts equilibrium to consume or produce the changed species.
Change in Pressure/Volume: For gases, decreasing volume (increasing pressure) shifts equilibrium toward fewer moles of gas; increasing volume shifts toward more moles.
Change in Temperature: Raising temperature favors the endothermic direction; lowering temperature favors the exothermic direction.
Catalyst: Does not affect equilibrium position, only speeds up attainment of equilibrium.
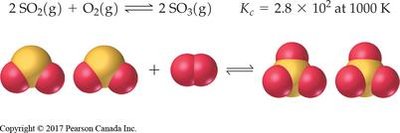
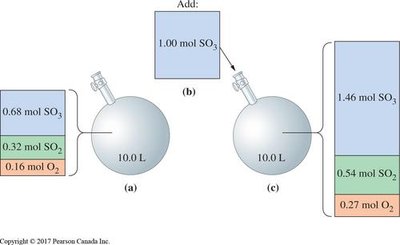
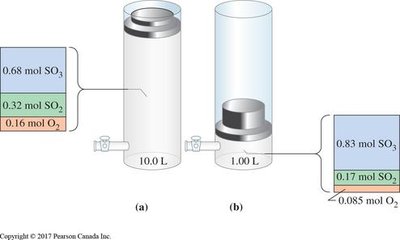

Worked Examples and Applications
Worked examples illustrate how to apply equilibrium concepts to calculate concentrations, partial pressures, and predict shifts in equilibrium.
Relating Equilibrium Concentrations: Use equilibrium constant expressions to solve for unknown concentrations.
Pure Solids and Liquids: Activities are set to 1; only gases and solutes are included in K expressions.
Interrelating KP and Kc: Use the relationship .
Predicting Direction: Compare Q and K to determine if the reaction will shift right or left.
Applying Le Châtelier’s Principle: Analyze effects of changes in concentration, volume, and temperature.





Summary Table: Using Q and K
Condition | Direction of Net Chemical Change |
|---|---|
Q < K | To the right (forward reaction) |
Q > K | To the left (reverse reaction) |
Q = K | Equilibrium; no net change |
Visual Representations
Graphs and diagrams help illustrate the approach to equilibrium, the effect of disturbances, and the molecular view of equilibrium.
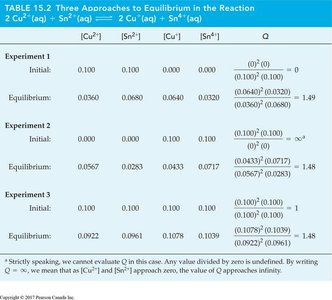
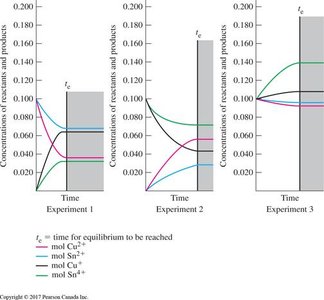

Additional info: These notes cover the core concepts of chemical equilibrium, including equilibrium constant expressions, the dynamic nature of equilibrium, Le Châtelier’s Principle, and the mathematical relationships between KP and Kc. Worked examples and tables provide practical applications and reinforce understanding for exam preparation.
