 Back
BackProperties of Water: Structure, Behavior, and Chemical Significance
Study Guide - Smart Notes

Water: Structure and Polarity
Molecular Structure of Water
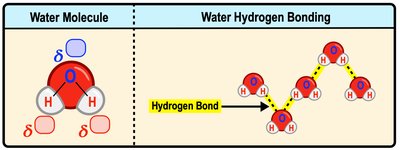
Water is a small, polar molecule composed of two hydrogen atoms and one oxygen atom (H2O). The oxygen atom is more electronegative, creating a partial negative charge, while the hydrogen atoms carry partial positive charges. This polarity allows water molecules to form hydrogen bonds with each other.
Polarity: Water's uneven charge distribution results in a dipole moment.
Hydrogen Bonding: Intermolecular attractions between the hydrogen of one water molecule and the oxygen of another.
Covalent Bonds: The bonds within a single water molecule are covalent.
Example: Water molecules bind to each other through hydrogen bonds, not covalent or ionic bonds.
Emergent Properties of Water
Overview of Emergent Properties
Water's hydrogen bonding gives rise to several emergent properties essential for life:
Adhesion: Water molecules stick to other polar surfaces.
Cohesion: Water molecules stick to each other.
Moderation of Temperature: Water resists temperature changes due to high specific heat.
Lower Density of Ice: Ice is less dense than liquid water, allowing it to float.
Universal Solvent: Water dissolves many substances, facilitating chemical reactions.

Properties of Water: Cohesion, Adhesion, and Surface Tension
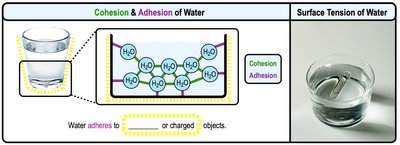
Cohesion and Adhesion
Cohesion refers to water molecules sticking to each other, while adhesion is their ability to stick to other substances. These properties are due to hydrogen bonding.
Surface Tension: The difficulty in breaking the surface of water, resulting from cohesive forces.
Biological Importance: Surface tension allows insects to walk on water and supports capillary action in plants.
Density of Water: Liquid vs. Solid
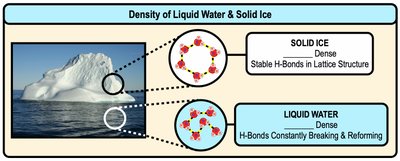
Density Differences and Biological Significance
Liquid water molecules are closely packed and constantly form and break hydrogen bonds. In solid ice, molecules are arranged in a stable lattice, making ice less dense than liquid water.
Ice Floats: Lower density of ice allows it to float, insulating aquatic life below.
Hydrogen Bonding: Stable hydrogen bonds in ice create a lattice structure.
Thermal Properties of Water
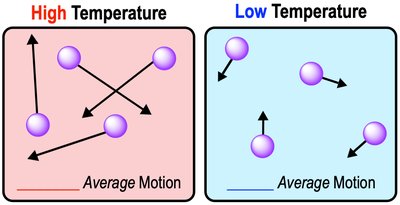
Kinetic Energy and Temperature
Kinetic energy is the energy of motion. Temperature measures the average kinetic energy of molecules in a substance. Water's high specific heat allows it to resist temperature changes, which is critical for maintaining stable environments.
Specific Heat: The amount of heat required to raise 1 gram of water by 1°C.
Heat Capacity: Water's high heat capacity stabilizes temperatures in organisms and environments.

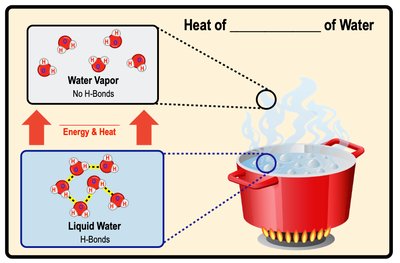
Heat of Vaporization
Evaporation is the phase transition from liquid to gas. Water has a high heat of vaporization due to strong hydrogen bonds, requiring significant energy to convert liquid water to vapor.
Heat of Vaporization: Amount of heat needed to convert 1 gram of liquid water to vapor.
Biological Importance: Evaporation cools surfaces, such as sweating in humans.
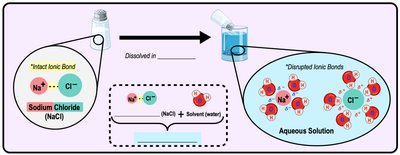
Water as the Universal Solvent
Dissolving Substances
Water is called the "universal solvent" because it dissolves many substances. The solvent is the substance doing the dissolving (usually water), while the solute is the substance being dissolved. Water forms a hydration shell around solute molecules, separating ions and facilitating reactions.
Aqueous Solution: A solution where water is the solvent.
Hydration Shell: Water molecules surround and stabilize ions or polar molecules.
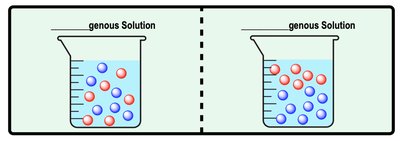
Homogenous vs. Heterogenous Solutions
Homogenous solutions are uniformly mixed, while heterogenous solutions are unevenly distributed.
Homogenous: All parts are equally distributed.
Heterogenous: Parts are unequally distributed.
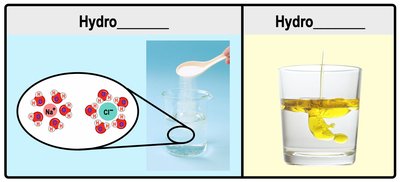
Hydrophilic vs. Hydrophobic Substances
Hydrophilic substances dissolve in water due to their attraction to it (water "loving"), typically polar or ionic molecules. Hydrophobic substances do not dissolve in water (water "fearing"), usually non-polar molecules like fats and oils.
Hydrophilic: Polar and ionic molecules (e.g., salts, ions).
Hydrophobic: Non-polar molecules (e.g., oils, waxes).
Acids, Bases, and pH
Acids and Bases
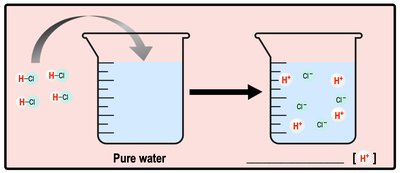
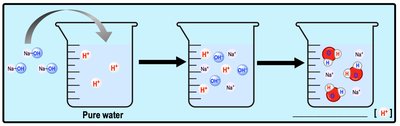
Acids increase the concentration of H+ ions in solution, while bases decrease it (often by increasing OH- ions). These changes affect biological processes and chemical reactions.
Acid: Donates H+ ions to solution.
Base: Accepts H+ ions or increases OH- concentration.
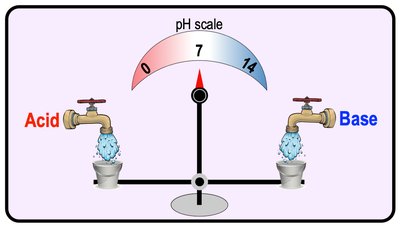
pH Scale
The pH scale measures the concentration of H+ ions in solution, ranging from 0 (acidic) to 14 (basic), with 7 being neutral. Biological systems are sensitive to pH changes.
Neutral Solution: [H+] = [OH-]
Acidic Solution: [H+] > [OH-]
Basic Solution: [H+] < [OH-]
Buffers and Biological Importance
Buffer Systems
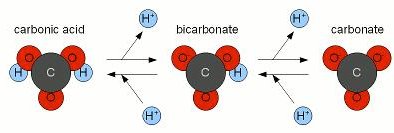
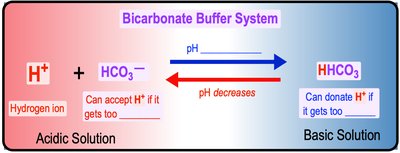
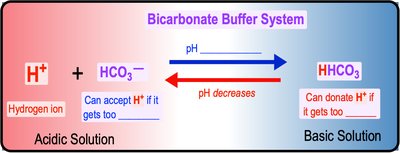
Buffers are substances that resist changes in pH when acids or bases are added. They maintain homeostasis by either donating or accepting H+ ions as needed. The bicarbonate buffer system is a key example in blood.
Buffer Action: Donates H+ when depleted, accepts H+ when in excess.
Bicarbonate Buffer System: Maintains blood pH by shifting between carbonic acid, bicarbonate, and carbonate forms.
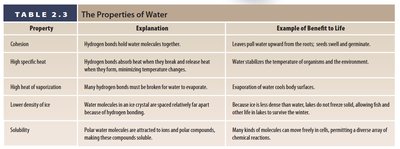
Summary Table: Properties of Water
Comparison of Water Properties
Property | Explanation | Example of Benefit to Life |
|---|---|---|
Cohesion | Hydrogen bonds hold water molecules together. | Leaves pull water upward from the roots; seeds swell and germinate. |
High specific heat | Hydrogen bonds absorb heat when they break and release heat when they form, minimizing temperature changes. | Water stabilizes the temperature of organisms and the environment. |
High heat of vaporization | Many hydrogen bonds must be broken for water to evaporate. | Evaporation of water cools body surfaces. |
Lower density of ice | Water molecules in ice are in a crystal lattice spaced relatively far apart because of hydrogen bonding. | Because ice is less dense than water, lakes do not freeze solid, allowing fish and other life to survive in winter. |
Solubility | Polar water molecules are attracted to ions and polar compounds, making these compounds soluble. | Many kinds of molecules can move freely in cells, permitting a diverse array of chemical reactions. |

