 Back
BackQuantum Chemistry Practice Guidance – CHEM 1042
Study Guide - Smart Notes

Q1. What is the frequency of electromagnetic radiation with a wavelength of 589 nm?
Background
Topic: Electromagnetic Radiation – Frequency and Wavelength Relationship
This question tests your understanding of how to relate the frequency and wavelength of light using the speed of light equation.
Key formula:
Where:
= speed of light ( m/s)
= wavelength (in meters)
= frequency (in Hz)
Step-by-Step Guidance
Convert the wavelength from nanometers to meters: .
Write the speed of light equation: .
Rearrange to solve for frequency: .
Plug in the values for and (in meters) to set up the calculation.
Try solving on your own before revealing the answer!
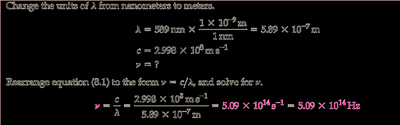
Final Answer: Hz
The frequency is calculated by dividing the speed of light by the wavelength in meters.
Q2. For radiation of wavelength 242.4 nm, what is the energy of (a) one photon, and (b) a mole of photons?
Background
Topic: Photon Energy – Planck’s Equation
This question tests your ability to use Planck’s equation to calculate the energy of a photon and then scale up to a mole of photons.
Key formula:
Where:
= energy (in Joules)
= Planck’s constant ( J·s)
= frequency (in Hz)
= speed of light ( m/s)
= wavelength (in meters)
Step-by-Step Guidance
Convert the wavelength from nanometers to meters: .
Calculate the frequency using .
Calculate the energy of one photon using .
To find the energy of a mole of photons, multiply the energy per photon by Avogadro’s number ( photons/mol).
Try solving on your own before revealing the answer!
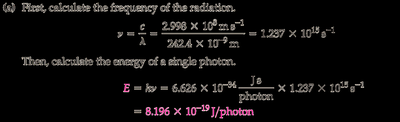
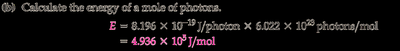
Final Answer: (a) J/photon, (b) J/mol
We first calculated the frequency, then used Planck’s equation for a single photon, and finally multiplied by Avogadro’s number for a mole.
Q3. Is there an energy level for the hydrogen atom having J?
Background
Topic: Quantization of Energy in Hydrogen Atom
This question tests your understanding of allowed energy levels in the hydrogen atom and the quantization condition.
Key formula:
Where:
= energy of the nth level
= Rydberg constant ( J)
= principal quantum number (must be integer)
Step-by-Step Guidance
Set J and J.
Rearrange the formula to solve for : .
Plug in the values and calculate .
Check if is an integer; only integer values correspond to allowed energy levels.
Try solving on your own before revealing the answer!

Final Answer: Not an allowed energy level
The calculated is not an integer, so this energy does not correspond to an allowed energy level for hydrogen.
Q4. Determine the wavelength of the line in the Balmer series of hydrogen corresponding to the transition from to .
Background
Topic: Hydrogen Atom Transitions – Balmer Series
This question tests your ability to calculate the energy and wavelength of a photon emitted during an electron transition in hydrogen.
Key formula:
Where:
= initial quantum number
= final quantum number
= Planck’s constant
= speed of light
Step-by-Step Guidance
Plug and into the energy difference formula.
Calculate for the transition.
Use to find the frequency.
Use to solve for the wavelength .
Try solving on your own before revealing the answer!
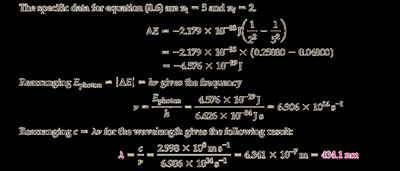
Final Answer: nm
The wavelength corresponds to the photon emitted during the to transition in the Balmer series.
Q5. Determine the kinetic energy of the electron ionised from a Li ion in its ground state, using a photon of frequency s.
Background
Topic: Ionization Energy and Conservation of Energy
This question tests your understanding of ionization of hydrogen-like ions and the calculation of kinetic energy after ionization.
Key formula:
Kinetic energy
Where:
= atomic number (for Li, )
= Rydberg constant
= principal quantum number (ground state )
= Planck’s constant
= photon frequency
Step-by-Step Guidance
Calculate the ground state energy for Li using with and .
Calculate the energy of the photon using .
Subtract the ionization energy from the photon energy to find the kinetic energy of the electron.
Try solving on your own before revealing the answer!
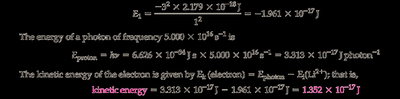
Final Answer: J
The kinetic energy is the difference between the photon energy and the ionization energy.
Q6. What is the wavelength associated with electrons traveling at one-tenth the speed of light?
Background
Topic: De Broglie Wavelength
This question tests your understanding of the wave-particle duality and how to calculate the wavelength of a moving particle.
Key formula:
Where:
= wavelength (in meters)
= Planck’s constant
= mass of electron ( kg)
= velocity ()
Step-by-Step Guidance
Calculate the velocity: m/s.
Plug the values for , , and into the de Broglie equation.
Set up the calculation for .
Try solving on your own before revealing the answer!

Final Answer: pm
The wavelength is calculated using the de Broglie relation for a fast-moving electron.
Q7. Calculating the uncertainty in the position of an electron
Background
Topic: Heisenberg Uncertainty Principle
This question tests your understanding of the uncertainty principle and how to relate uncertainties in position and momentum.
Key formula:
Where:
= uncertainty in position
= uncertainty in momentum ()
= Planck’s constant
= 3.14
Step-by-Step Guidance
Calculate the uncertainty in velocity: m/s.
Calculate the uncertainty in momentum: .
Use the uncertainty principle to solve for : .
Try solving on your own before revealing the answer!

Final Answer: About 10 atomic diameters
The uncertainty in position is significant due to the uncertainty in speed.
Q8. What is the energy difference between the ground state and the first excited state of an electron in a one-dimensional box 1.00 × 102 pm long? Hence, calculate the wavelength of the photon that could excite the electron from the ground state to the first excited state.
Background
Topic: Particle in a Box – Quantum Transitions
This question tests your understanding of quantum mechanics for a particle in a box and how to relate energy differences to photon wavelengths.
Key formula:
Where:
= quantum number ( for ground, for first excited)
= Planck’s constant
= mass of electron
= length of box (in meters)
= speed of light
Step-by-Step Guidance
Convert the box length from pm to meters: pm m.
Calculate and using for and .
Find .
Calculate the wavelength of the photon using .
Try solving on your own before revealing the answer!
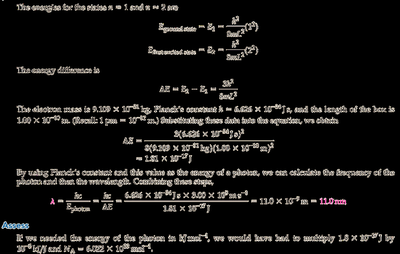
Final Answer: nm
The energy difference and photon wavelength are calculated using quantum mechanics for a particle in a box.
