 Back
BackQuantum-Mechanical Model of the Atom: Structure, Light, and Atomic Spectra
Study Guide - Smart Notes

The Quantum-Mechanical Model of the Atom
Introduction to Quantum Mechanics
The quantum-mechanical model explains the behavior of electrons in atoms, which is fundamentally different from the macroscopic world. Early 20th-century scientists such as Einstein, Bohr, de Broglie, Planck, Heisenberg, Dirac, and Schrödinger contributed to our understanding of subatomic particles—electrons, protons, and neutrons.
Wave–matter duality: Electrons exhibit both particle-like and wave-like properties depending on experimental conditions.
Subatomic behavior: Direct observation of electrons alters their behavior due to their small size.
Electron cloud: Electrons are best described as a cloud of probable positions rather than fixed orbits.
The quantum model forms the foundation of chemistry, explaining periodic trends, chemical bonding, atomic colors, and reactivity.
The Nature of Light
Electromagnetic Radiation and Wave Properties
Light is a form of electromagnetic radiation, consisting of oscillating electric and magnetic fields perpendicular to each other and to the direction of travel.
Electric field: Region where charged particles experience a force.
Magnetic field: Region where magnetized particles experience a force.
Speed of light (c): In a vacuum, all electromagnetic waves travel at m/s.
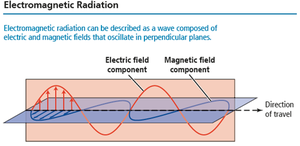
Wave Characteristics: Amplitude, Wavelength, and Frequency
Energy waves are described by amplitude, wavelength (), and frequency ().
Amplitude: Height of the wave; determines light intensity (brightness).
Wavelength (): Distance between consecutive crests or troughs; determines color.
Frequency (): Number of waves passing a point per second; measured in hertz (Hz).
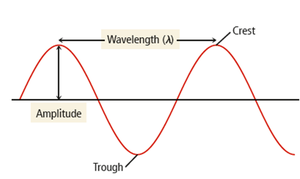
Wavelength and amplitude are independent properties. Wavelength determines color (intensive property), while amplitude determines brightness (extensive property).
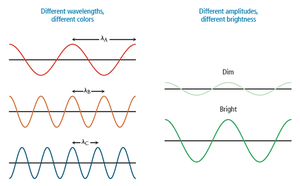
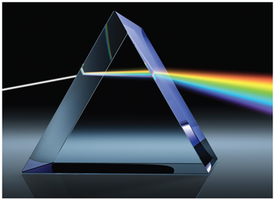
Color and the Visible Spectrum
The color of light is determined by its wavelength or frequency. White light is a mixture of all visible wavelengths (ROYGBIV). Objects appear colored due to selective absorption and reflection of certain wavelengths.
Relationship Between Wavelength and Frequency
Wavelength and frequency are inversely proportional for electromagnetic waves:
If wavelength decreases, frequency increases, and vice versa.
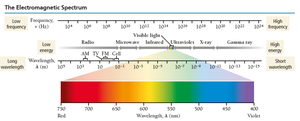
The Electromagnetic Spectrum
Visible light (400–700 nm) is a small part of the electromagnetic spectrum. Shorter wavelengths (higher frequency) have higher energy. Gamma rays have the highest energy; radio waves have the lowest.
Ionizing radiation: High-energy electromagnetic radiation (UV, X-ray, gamma) can damage biological molecules.
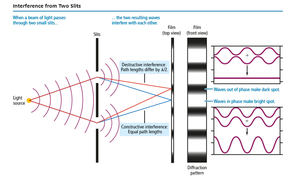
Wave Behavior: Interference and Diffraction
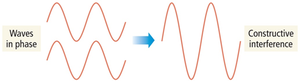
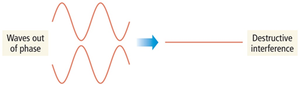
Interference
When waves interact, they can combine constructively (in phase) or destructively (out of phase).
Constructive interference: Waves add to form a larger wave.
Destructive interference: Waves cancel each other out.
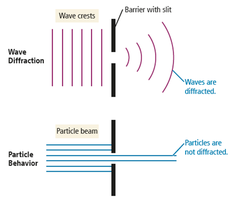
Diffraction
Waves bend around obstacles or openings similar in size to their wavelength, producing diffraction patterns. Particles do not diffract.
Two-Slit Interference
Passing light through two slits creates an interference pattern due to differences in path length, resulting in alternating bright and dark spots.
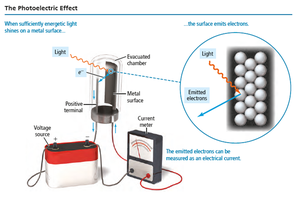
The Photoelectric Effect
Einstein’s Explanation
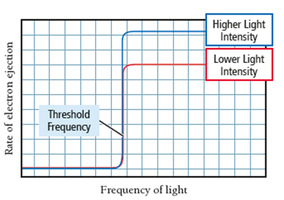
When light shines on a metal surface, electrons (photoelectrons) are emitted. Classical theory could not explain the observed threshold frequency and immediate electron emission with high-frequency light.
Quantum theory: Light energy is delivered in packets called quanta or photons.
Threshold frequency: Minimum frequency required for electron emission, regardless of intensity.
Photon energy:
Planck’s constant (h): J·s
Atomic Spectra and the Bohr Model
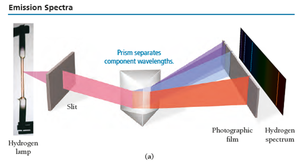
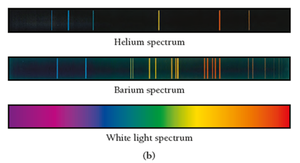
Emission Spectra
Atoms and molecules emit light when they absorb energy. The emitted light, passed through a prism, produces a unique pattern of wavelengths (line spectra) for each element.

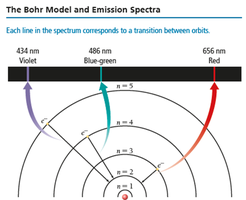
The Bohr Model of the Atom
Bohr proposed that electrons travel in quantized orbits (stationary states) around the nucleus. The energy of an electron is proportional to its distance from the nucleus. Electrons emit photons when transitioning between orbits, with the photon's energy equal to the difference between orbit energies.
Wave Behavior of Electrons: de Broglie and Electron Diffraction
de Broglie Relation
de Broglie proposed that particles, such as electrons, have wave-like properties. The wavelength of a particle is inversely proportional to its momentum:
For small particles like electrons, wave behavior is significant.
Electron Diffraction
Electron beams produce interference patterns, demonstrating their wave nature.
Heisenberg’s Uncertainty Principle
Wave-Particle Duality and Uncertainty
The position and velocity of an electron cannot both be known precisely at the same time. The more accurately one is known, the less accurately the other can be determined.
Uncertainty principle:
Indeterminacy: Electron paths are probabilistic, not deterministic.
Schrödinger’s Equation and Quantum Numbers
Wave Functions and Orbitals
Schrödinger’s equation calculates the probability of finding an electron with a specific energy at a specific location. Solutions yield wave functions (), which describe orbitals—regions of high probability for electron location.
Quantum Numbers
Quantum numbers describe the properties of atomic orbitals:
Principal quantum number (n): Energy level;
Angular momentum quantum number (l): Orbital shape; (s), $1 (d), $3$ (f)
Magnetic quantum number (): Orbital orientation; to
Spin quantum number (): Electron spin; or
Atomic Orbitals: Shapes and Probability Distributions
Probability and Radial Distribution Functions
The probability density function describes the likelihood of finding an electron at a specific point. The radial distribution function shows the total probability at a certain distance from the nucleus, with a maximum at the most probable radius.
Shapes of Atomic Orbitals
s-orbitals (l = 0): Spherical shape
p-orbitals (l = 1): Dumbbell shape, oriented along axes
d-orbitals (l = 2): Four-lobed or two-lobed with a ring
f-orbitals (l = 3): Eight-lobed or two-lobed with a ring
Electron Transitions and Atomic Spectra
Electron Excitation and Relaxation
Electrons transition between energy levels by absorbing or emitting photons. The energy of the photon equals the difference between the initial and final states. Each line in the emission spectrum corresponds to a specific transition.
Quantum Number | Symbol | Meaning | Possible Values |
|---|---|---|---|
Principal | n | Energy level, size | 1, 2, 3, ... |
Angular momentum | l | Shape | 0 to n-1 |
Magnetic | ml | Orientation | -l to +l |
Spin | ms | Spin direction | +1/2, -1/2 |
Key Equations:
Example: Calculate the energy of a photon with wavelength 500 nm:
J
Additional info: These notes cover the quantum-mechanical model of the atom, the nature of light, atomic spectra, and the fundamental quantum numbers, which are essential for understanding atomic structure and properties in general chemistry.
