 Back
BackQuantum-Mechanical Model of the Atom: Electromagnetic Radiation, Photons, and Bohr's Model
Study Guide - Smart Notes

Quantum-Mechanical Model of the Atom
Electromagnetic Radiation and Atomic Structure
The study of light and its properties is fundamental to understanding the electronic structure of atoms. Light interacts with matter, revealing information about atomic energy levels and electron arrangements. The colors emitted by heated elements are directly related to their electronic structure.

Overview of Key Topics
Electromagnetic Radiation
Quantized Energy and Photons
Bohr’s Model of the Hydrogen Atom
These topics are central to understanding how light reveals atomic structure and how quantum mechanics describes atomic behavior.

The Dual Nature of Light
Wave and Particle Properties
Light exhibits both wave-like and particle-like characteristics. This duality is essential for explaining phenomena such as interference, diffraction, and the photoelectric effect.
Wave Description: Light possesses wavelength, frequency, amplitude, and energy.
Electromagnetic Radiation: Light consists of fluctuating electric and magnetic fields perpendicular to the direction of wave motion.
Wave Properties: Light shows interference and diffraction, similar to other waves.
Particle Description: Light can also be described as a particle with mass and momentum.

Atomic Emission Spectra
Hydrogen Emission Spectrum
When atoms are excited, they emit light at specific wavelengths, producing an emission spectrum. The hydrogen atom's emission spectrum is a key example, showing discrete lines corresponding to electronic transitions.
Emission Spectra: Each element produces a unique set of spectral lines.
Goal: To explain the origin of these lines using quantum theory.

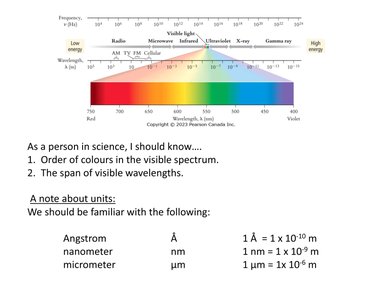
The Electromagnetic Spectrum
Visible Light and Units
The electromagnetic spectrum includes all types of electromagnetic radiation, from radio waves to gamma rays. Visible light is a small portion, with wavelengths from about 400 nm (violet) to 700 nm (red).
Order of Colors: Violet, Blue, Green, Yellow, Orange, Red
Units: Angstrom (Å), nanometer (nm), micrometer (μm)
Unit | Symbol | Conversion |
|---|---|---|
Angstrom | Å | 1 Å = 1 x 10-10 m |
Nanometer | nm | 1 nm = 1 x 10-9 m |
Micrometer | μm | 1 μm = 1 x 10-6 m |
Light as Electromagnetic Radiation
Relationship Between Wavelength and Frequency
All electromagnetic radiation travels at the speed of light (c) in a vacuum. The speed is related to wavelength (λ) and frequency (ν) by:
λ: Wavelength (meters)
ν: Frequency (s-1 or Hz)
c: Speed of light (3.00 x 108 m/s)
Frequency and wavelength are inversely related: as one increases, the other decreases.

Wave Properties: Interference and Diffraction
Examples of Wave Behavior
Light, as a wave, can exhibit interference and diffraction. These properties are observed in experiments such as ripple tanks and X-ray diffraction.
Interference: Constructive and destructive patterns from overlapping waves.
Diffraction: Bending and spreading of waves around obstacles.

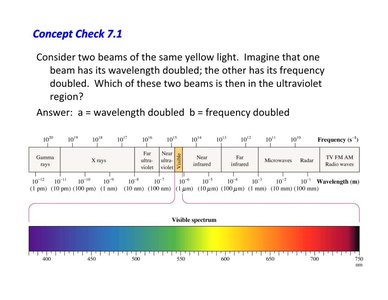
Concept Check: Wavelength and Frequency
Application to the Electromagnetic Spectrum
Doubling the wavelength or frequency of light shifts its position in the electromagnetic spectrum. Higher frequency corresponds to higher energy and shorter wavelength.
Light as a Particle: Quantized Energy and Photons
Planck's Quantum Theory
Max Planck proposed that electromagnetic radiation is quantized, coming in discrete units called quanta. The energy of a quantum is given by:
h: Planck's constant (6.63 x 10-34 J·s)
ν: Frequency of light
This explains why emission spectra have discrete lines rather than a continuum.

Einstein and the Photon Concept
Einstein extended Planck's ideas, suggesting that each quantum of light behaves as a particle (photon) with energy . Photons are packets of light energy.
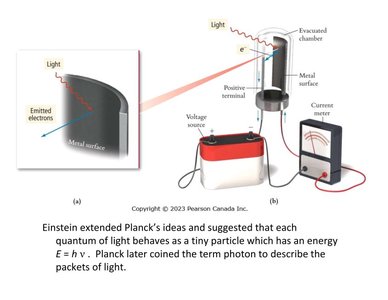
The Photoelectric Effect
Evidence for Particle Nature of Light
The photoelectric effect demonstrates that light can eject electrons from a metal surface, but only if the light has sufficient energy (frequency). This effect supports the particle theory of light.
Electrons are emitted only at certain frequencies.
Intensity affects the number of electrons, not their energy.

Mechanism of the Photoelectric Effect
Electrons are ejected from metal when struck by photons with energy greater than the binding energy (work function, φ).
If photon energy is less than φ, no electrons are ejected.
Excess energy becomes kinetic energy of the electron:
Number of electrons ejected depends on light intensity above the threshold.

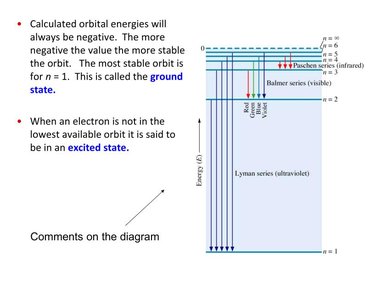
Bohr’s Model of the Hydrogen Atom
Explaining the Line Spectrum
Niels Bohr proposed that the line spectrum of hydrogen is due to electrons orbiting the nucleus in specific, quantized orbits. Each orbit corresponds to a specific energy level.

Electrons circle the nucleus only in allowable orbits with specific radii and energies.
An electron in an allowable orbit does not radiate energy.
Orbits are defined by the principal quantum number, n.
Values of n are integers (1, 2, 3, ...), with n = 1 closest to the nucleus.

Electron Transitions and Emission Lines
Electrons can "jump" between energy levels by absorbing or emitting photons. Moving to a higher orbit requires absorption of energy; moving to a lower orbit releases energy as light.

Calculated orbital energies are negative; the most stable orbit is n = 1 (ground state).
Electrons not in the lowest orbit are in an excited state.

Concept Check: Photoelectric Effect and Light Energy
Application to Electron Ejection
When a metal is struck by light of different frequencies, the kinetic energy of ejected electrons depends on the energy of the photons. Ultraviolet light, with higher energy, ejects electrons with greater kinetic energy than yellow light.
Additional info: These notes cover the foundational concepts of Chapter 7: Quantum-Mechanical Model of the Atom, including electromagnetic radiation, quantized energy, photons, and Bohr's model. The explanations are expanded for clarity and completeness, suitable for exam preparation in general chemistry.
