 Back
BackQuantum-Mechanical Model of the Atom: Structure, Properties, and Electron Behavior
Study Guide - Smart Notes

The Quantum-Mechanical Model of the Atom
Introduction to Quantum Mechanics
The quantum-mechanical model of the atom revolutionized our understanding of matter at the subatomic level. Early twentieth-century scientists such as Albert Einstein, Niels Bohr, Louis de Broglie, Max Planck, Werner Heisenberg, P. A. M. Dirac, and Erwin Schrödinger contributed foundational work that explains the strange behavior of electrons, protons, and neutrons. - Quantum mechanics investigates the behavior of absolutely small particles, which differ fundamentally from macroscopic objects. - Wave–matter duality: Subatomic particles, like electrons, exhibit both particle-like and wave-like properties depending on experimental conditions. - Electrons are so small that direct observation alters their behavior, making their study reliant on indirect methods.
Key Features of the Quantum Mechanical Model
The quantum mechanical model explains how electrons exist and behave in atoms, emphasizing their wave nature over their particulate characteristics. - Electron cloud: Electrons are best described as a cloud of probable positions rather than as particles orbiting the nucleus. - This model forms the foundation of chemistry, explaining periodic trends, chemical bonding, atomic colors, and sizes. - It predicts why elements behave as metals or nonmetals, their reactivity, and periodic patterns.
The Nature of Light and Electromagnetic Radiation
Wave Nature of Light
Light is a form of electromagnetic radiation, composed of oscillating electric and magnetic fields perpendicular to each other. - Electric field: Region where charged particles experience force. - Magnetic field: Region where magnetized particles experience force. - All electromagnetic waves travel at the speed of light in a vacuum. 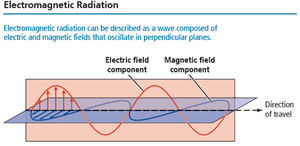
Characteristics of Energy Waves
- Amplitude: Height of the wave, measuring light intensity. - Wavelength (\lambda): Distance between consecutive crests or troughs, determining color. 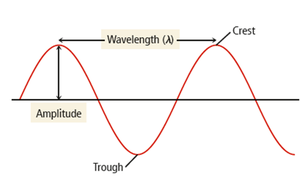 - Frequency (\nu): Number of waves passing a point per unit time, measured in hertz (Hz). - Total energy (E): Proportional to amplitude and frequency.
- Frequency (\nu): Number of waves passing a point per unit time, measured in hertz (Hz). - Total energy (E): Proportional to amplitude and frequency.
Relationship Between Wavelength and Frequency
Wavelength and frequency are inversely proportional for waves traveling at the same speed. The speed of light (c) is constant, so:
Color and Light
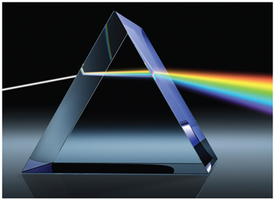
The color of light is determined by its wavelength or frequency. White light is a mixture of all visible wavelengths. When objects absorb some wavelengths and reflect others, the reflected colors are observed. 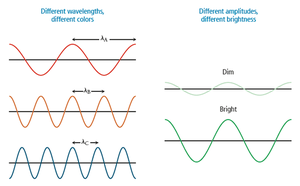
The Electromagnetic Spectrum
Visible light (400–700 nm) is only a small part of the electromagnetic spectrum. - Shorter wavelength (higher frequency) light has higher energy. - Radio waves have the lowest energy; gamma rays have the highest. - High-energy radiation (UV, X-ray, gamma) can damage biological molecules (ionizing radiation). 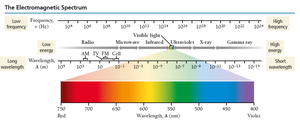
Wave Behavior: Interference and Diffraction
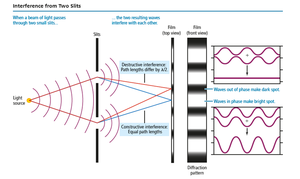
Interference
Waves interact through interference: - Constructive interference: Waves in phase add to make a larger wave. - Destructive interference: Waves out of phase cancel each other.
Diffraction
When waves encounter an obstacle or opening similar in size to their wavelength, they bend around it (diffraction). Particles do not diffract. Diffraction through two slits produces an interference pattern characteristic of waves. 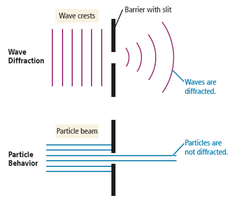
The Photoelectric Effect
Einstein's Observations
When light shines on a metal surface, electrons (photoelectrons) are emitted. This is the photoelectric effect. 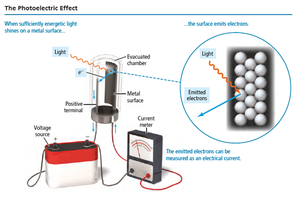
Classic vs Quantum Theory
- Classic theory: Increasing light intensity or decreasing wavelength should eject more electrons, with a lag time for dim light. - Quantum theory: A minimum frequency (threshold frequency) is required for electron emission, regardless of intensity. High-frequency light causes immediate emission. 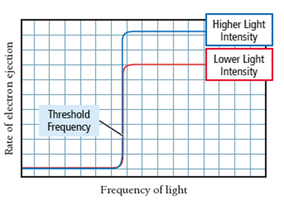
Photon Energy
Einstein proposed that light energy is delivered in packets called photons. The energy of a photon is: where is Planck's constant and is the speed of light. 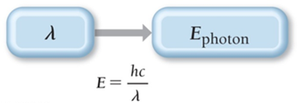
Atomic Spectra and the Bohr Model
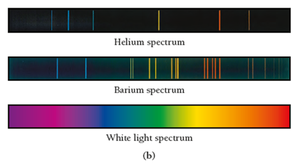
Emission Spectra
When atoms absorb energy, they emit light at specific wavelengths, producing a unique emission spectrum (line spectra). Each element has its own spectrum. 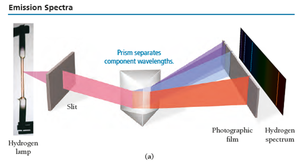

The Bohr Model
Bohr's model explains energy transitions in atoms. - Electrons travel in fixed orbits (stationary states) at set distances from the nucleus. - Energy is quantized; electrons emit photons when transitioning between orbits. - The energy of the photon equals the difference between orbit energies. 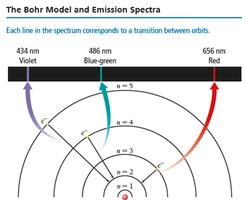
Wave Behavior of Electrons
De Broglie Relation
De Broglie proposed that particles have wave-like character. The wavelength of a particle is inversely proportional to its momentum: where is mass and is velocity. 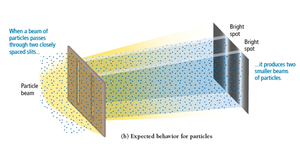
Complementary Properties and Uncertainty
Wave-Particle Duality
Electrons exhibit both particle (position) and wave (interference) properties, but both cannot be observed simultaneously.
Heisenberg's Uncertainty Principle
The product of uncertainties in position and velocity is inversely proportional to mass: This means that knowing one property precisely makes the other less certain.
Quantum Numbers and Atomic Orbitals
Quantum Numbers
The size, shape, and orientation of atomic orbitals are described by four quantum numbers:
Quantum Number | Symbol | Description |
|---|---|---|
Principal | n | Energy level, size of orbital |
Angular Momentum | l | Shape of orbital (s, p, d, f) |
Magnetic | ml | Orientation in space |
Spin | ms | Electron spin direction |
- Principal quantum number (n): Determines energy and size; larger n means higher energy and larger orbital. - Angular momentum quantum number (l): Determines shape; s (l=0), p (l=1), d (l=2), f (l=3). - Magnetic quantum number (ml): Specifies orientation; values range from -l to +l. - Spin quantum number (ms): Specifies electron spin; values are +1/2 or -1/2.
Energy Levels and Sublevels
- The number of subshells in a shell = n. - The number of orbitals in a subshell = 2l + 1.
Electron Transitions and Atomic Spectra
Electron Excitation and Relaxation
Electrons transition between energy levels by absorbing or emitting photons. The energy of the photon equals the difference between the initial and final states.
Probability Functions and Orbital Shapes
Probability Density and Radial Distribution
- Probability density: Likelihood of finding an electron at a specific point. - Radial distribution function: Total probability of finding an electron within a spherical shell at distance r from the nucleus.
Atomic Orbital Shapes
- s-orbitals (l=0): Spherical, lowest energy in each principal level. - p-orbitals (l=1): Dumbbell-shaped, three orientations per principal level above n=1.
- d-orbitals (l=2): Four-lobed or toroidal shapes, five orientations per principal level above n=2. - f-orbitals (l=3): Eight-lobed or toroidal shapes, seven orientations per principal level above n=3.
Orbital Phase
Orbitals are derived from wave functions, which can have positive or negative values (phases). Nodes are points where the wave function equals zero.
Summary Table: Quantum Numbers and Orbitals
n | l | ml | Orbital Name | Number of Orbitals |
|---|---|---|---|---|
1 | 0 | 0 | 1s | 1 |
2 | 0,1 | 0; -1,0,1 | 2s, 2p | 1 (2s), 3 (2p) |
3 | 0,1,2 | 0; -1,0,1; -2,-1,0,1,2 | 3s, 3p, 3d | 1 (3s), 3 (3p), 5 (3d) |
4 | 0,1,2,3 | 0; -1,0,1; -2,-1,0,1,2; -3,-2,-1,0,1,2,3 | 4s, 4p, 4d, 4f | 1 (4s), 3 (4p), 5 (4d), 7 (4f) |
Conclusion
The quantum-mechanical model provides a comprehensive framework for understanding atomic structure, electron behavior, and the properties of elements. It explains phenomena such as atomic spectra, chemical bonding, and the periodic table, forming the foundation for modern chemistry.
