 Back
BackQuantum-Mechanical Model of the Atom: Structure, Properties, and Electron Behavior
Study Guide - Smart Notes

The Quantum-Mechanical Model of the Atom
Introduction to Quantum Mechanics
The quantum-mechanical model of the atom explains the unique and often counterintuitive behavior of electrons and other subatomic particles. Early twentieth-century scientists such as Einstein, Bohr, de Broglie, Planck, Heisenberg, Dirac, and Schrödinger contributed to the foundation of quantum theory, which describes matter at the atomic and subatomic level.
Subatomic particles: Electrons, protons, neutrons
Wave–matter duality: Electrons exhibit both particle-like and wave-like properties depending on experimental conditions.
Key concept: Direct observation of electrons changes their behavior due to their extremely small size.
Wave–Matter Duality
Electrons and other subatomic particles display duality, meaning they can behave as both particles and waves. This duality is central to quantum mechanics and is observed in phenomena such as interference and diffraction.
Particulate behavior: Mass, volume
Wave behavior: Energy, frequency, wavelength
The Nature of Light and Electromagnetic Radiation
Wave Nature of Light
Light is a form of electromagnetic radiation, consisting of oscillating electric and magnetic fields perpendicular to each other and to the direction of travel. All electromagnetic waves travel at the speed of light in a vacuum.
Electric field: Region where charged particles experience force
Magnetic field: Region where magnetized particles experience force
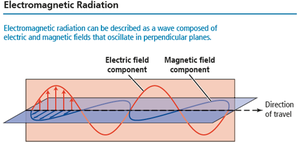
Characteristics of Energy Waves
Energy waves are described by their amplitude and wavelength:
Amplitude: Height of the wave; determines light intensity (brightness)
Wavelength (λ): Distance between consecutive crests or troughs; determines color
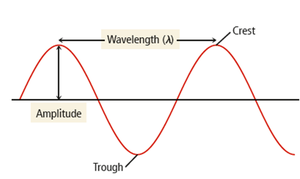
Wavelength and Amplitude
Wavelength and amplitude are independent properties. Wavelength determines the color of light, while amplitude determines its brightness.
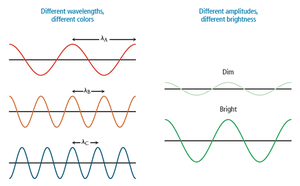
Color and the Visible Spectrum
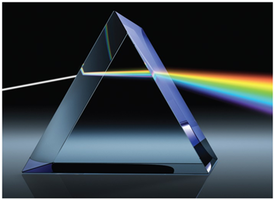
The color of light is determined by its wavelength or frequency. White light is a mixture of all visible wavelengths. When an object absorbs some wavelengths and reflects others, it appears colored.
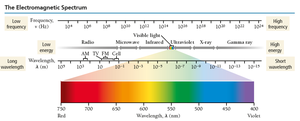
The Electromagnetic Spectrum
Visible light is only a small part of the electromagnetic spectrum, which includes radio waves, microwaves, infrared, ultraviolet, X-rays, and gamma rays. Shorter wavelengths correspond to higher energy and frequency.
Radio waves: Lowest energy
Gamma rays: Highest energy
Ionizing radiation: High-energy radiation (UV, X-ray, gamma) can damage biological molecules
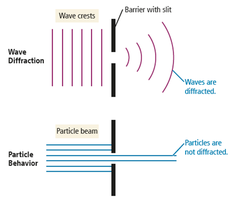
Wave Behavior: Interference and Diffraction
Interference
When waves interact, they can combine constructively (in phase, larger wave) or destructively (out of phase, cancel each other).
Diffraction
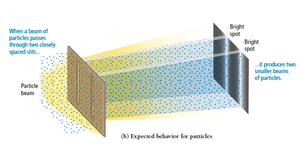
Waves bend around obstacles or openings comparable in size to their wavelength, producing diffraction patterns. Particles do not diffract.
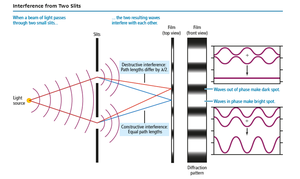
Two-Slit Interference
Passing light through two slits produces an interference pattern, a hallmark of wave behavior.
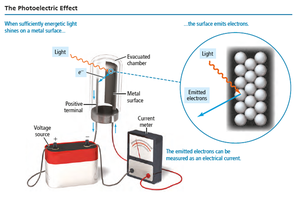
The Photoelectric Effect
Einstein's Discovery
When light shines on a metal surface, electrons are emitted (photoelectrons). This phenomenon is called the photoelectric effect.
Classical vs Quantum Explanation
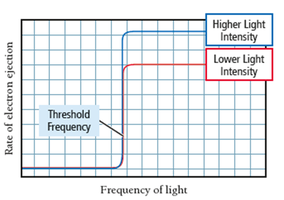
Classical theory: Predicted that increasing light intensity or decreasing wavelength would eject more electrons, with a lag time for dim light.
Quantum theory: Experimental results showed a minimum frequency (threshold frequency) was required, regardless of intensity. High-frequency light caused immediate electron emission.
Energy of a Photon
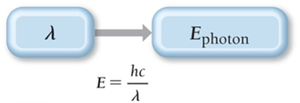
Einstein proposed that light energy is delivered in packets called photons. The energy of a photon is directly proportional to its frequency and inversely proportional to its wavelength:
Planck's constant (h): Fundamental constant
Speed of light (c): Constant value
Equation:
Atomic Spectra and the Bohr Model
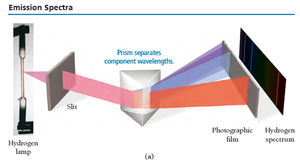
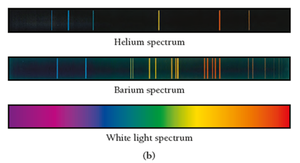
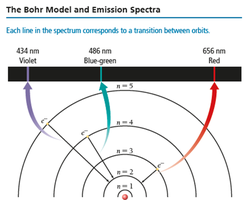
Emission Spectra
When atoms absorb energy, they emit light at specific wavelengths, producing a unique emission spectrum. Each element has its own line spectrum, which can be used for identification.

The Bohr Model
Bohr's model explains energy transitions in atoms. Electrons travel in fixed orbits (stationary states) at specific distances from the nucleus. Energy is quantized, and electrons emit photons when transitioning between orbits.
Wave Behavior of Electrons and de Broglie Relation
de Broglie Wavelength
de Broglie proposed that particles, including electrons, have wave-like properties. The wavelength of a particle is inversely proportional to its momentum:
Electron Diffraction
Electron beams produce interference patterns, confirming their wave nature.
Complementary Properties and Heisenberg's Uncertainty Principle
Wave-Particle Duality and Uncertainty
Electrons exhibit both particle (position) and wave (interference) properties, but these are complementary. The more precisely one property is known, the less precisely the other can be known.
Heisenberg's Uncertainty Principle
The product of uncertainties in position and velocity is inversely proportional to mass:
Quantum Numbers and Atomic Orbitals
Principal Quantum Number (n)
Defines the energy level and shell of an electron. Higher n values correspond to higher energy and larger orbitals.
Angular Momentum Quantum Number (l)
Defines the shape of the orbital. Values range from 0 to n-1:
l = 0: s orbital (spherical)
l = 1: p orbital (dumbbell)
l = 2: d orbital (complex)
l = 3: f orbital (more complex)
Magnetic Quantum Number (ml)
Specifies the orientation of the orbital in space. Values range from -l to +l.
Spin Quantum Number
Describes the intrinsic spin of the electron, which can be in one of two quantized states.
Atomic Orbitals and Subshells
Orbitals are regions of high probability for finding electrons. Each set of quantum numbers (n, l, ml) describes one orbital. Orbitals with the same n are in the same shell; those with the same n and l are in the same subshell.
Electron Transitions and Atomic Spectra
Electron Excitation and Relaxation
Electrons absorb energy to move to higher energy levels (excitation) and emit photons when returning to lower levels (relaxation). The energy of the emitted photon equals the difference between the two energy levels.
Energy Transitions in Hydrogen
The wavelength of light emitted or absorbed during electron transitions can be calculated using energy difference equations.
Radial Nodes and Orbital Shapes
Radial Nodes
Radial nodes are distances from the nucleus where the probability of finding an electron is zero. The number of radial nodes is given by:
Number of radial nodes = n - l - 1
Orbital Shapes
s orbitals: Spherical shape
p orbitals: Dumbbell shape
d orbitals: Four-lobed or toroidal shapes
f orbitals: Eight-lobed or toroidal shapes
Summary Table: Quantum Numbers and Orbitals
Quantum Number | Symbol | Meaning | Possible Values |
|---|---|---|---|
Principal | n | Energy level, shell | 1, 2, 3, ... |
Angular Momentum | l | Orbital shape | 0 to n-1 |
Magnetic | ml | Orbital orientation | -l to +l |
Spin | ms | Electron spin | +1/2 or -1/2 |
Additional info: All equations are provided in LaTeX format for clarity. Images included are directly relevant to the explanation of each paragraph and reinforce key concepts in quantum mechanics and atomic structure.
