 Back
BackQuantum Mechanics and Electron Configurations: The Nature of Light and Atomic Structure
Study Guide - Smart Notes

Quantum Mechanics and Electron Configurations
Introduction to Atomic Structure and Electromagnetic Radiation
Understanding atomic structure requires a deep knowledge of electromagnetic radiation and how electrons are arranged in atoms. This chapter explores the dual nature of light, the quantization of energy, and how these principles explain the properties and behaviors of elements.
The Nature of Electromagnetic Radiation
What is Electromagnetic Radiation?
Electromagnetic radiation is a form of energy that travels through space as oscillating electric and magnetic fields. It includes a wide range of phenomena, from radio waves to gamma rays.
Examples: Radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, gamma rays.
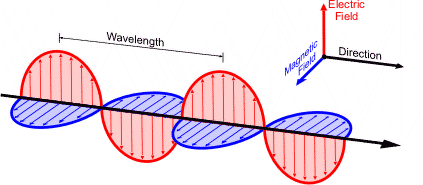
Wave Properties of Light
Light exhibits wave-like behavior, characterized by several key properties:
Wavelength (λ): The distance between two consecutive peaks of a wave, measured in meters (m).
Frequency (ν): The number of wave cycles that pass a given point per second, measured in hertz (Hz).
Amplitude: The height of the wave, related to the intensity of the radiation.
Speed of Light (c): All electromagnetic waves travel at the speed of light in a vacuum, m/s.
The relationship between wavelength, frequency, and speed of light is given by:
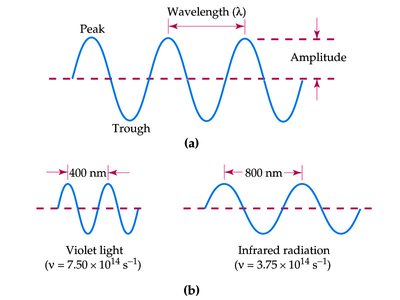
The Electromagnetic Spectrum
The electromagnetic spectrum encompasses all types of electromagnetic radiation, arranged by wavelength and frequency. Visible light is only a small portion of the spectrum.
Shorter wavelength = higher frequency = higher energy
Longer wavelength = lower frequency = lower energy
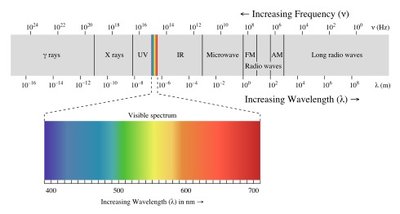
Comparing Wave Properties
Different types of electromagnetic radiation can be compared by their intensity, frequency, and wavelength. For example, violet light has a shorter wavelength and higher frequency than infrared radiation.
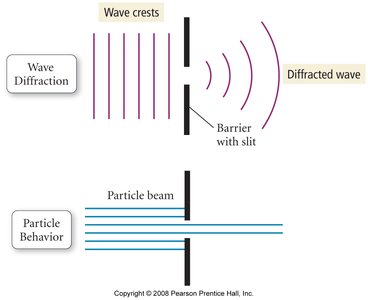
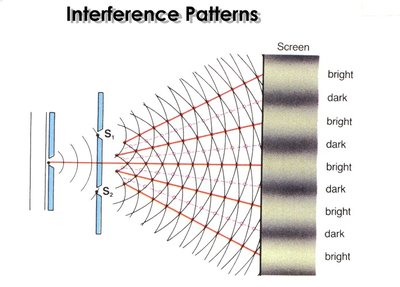
Wave Phenomena: Diffraction and Interference
Light as a wave can exhibit phenomena such as diffraction and interference:
Diffraction: The bending of waves around obstacles or through slits.
Interference: The combination of two or more waves resulting in constructive (bright) or destructive (dark) patterns.
Particle Nature of Light: The Photoelectric Effect
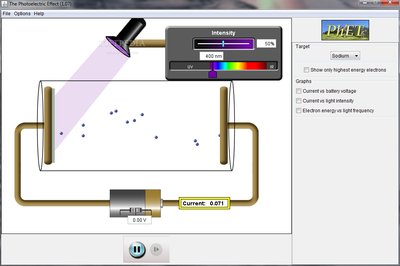
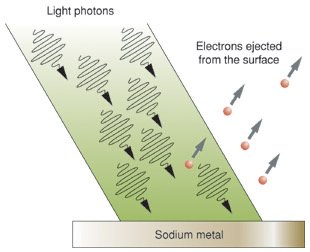
Evidence for Light as a Particle
Some phenomena, such as the photoelectric effect, cannot be explained by the wave model alone. When light of sufficient frequency shines on a metal surface, electrons are ejected. This effect demonstrates that light also behaves as a stream of particles called photons.
Threshold frequency: Electrons are only ejected if the light's frequency is above a certain value, regardless of intensity.
Photon energy: where is Planck's constant ( J·s).
Wave-Particle Duality
Light exhibits both wave-like and particle-like properties, a concept known as wave-particle duality. This duality is fundamental to quantum mechanics and is especially important at atomic and subatomic scales.
Energy of a photon:
Relationship to wavelength:
Atomic Spectra and the Bohr Model
Atomic Emission and Absorption Spectra
Atoms emit or absorb light at specific wavelengths, producing line spectra unique to each element. This is evidence for quantized energy levels in atoms.
Emission spectrum: Produced when electrons fall from higher to lower energy levels, emitting photons.
Absorption spectrum: Produced when electrons absorb photons and move to higher energy levels.

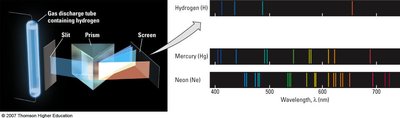
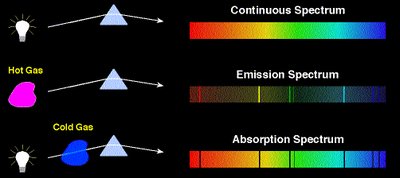
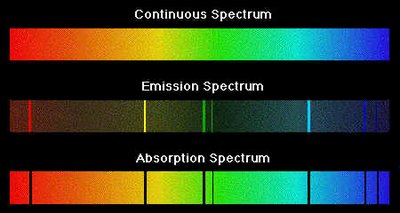
The Bohr Model of the Atom
The Bohr model proposed that electrons move in fixed orbits around the nucleus, each with a specific energy. Transitions between these orbits explain the observed spectral lines.
Energy quantization: Electrons can only occupy certain energy levels.
Photon emission/absorption: Occurs when electrons move between energy levels, with energy difference .
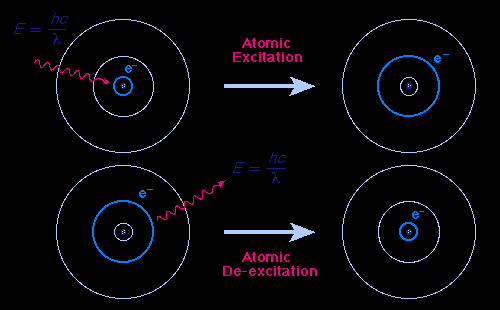
Quantum Mechanics and Atomic Orbitals
Wave Nature of Matter
Louis de Broglie proposed that all matter has wave properties, with wavelength given by:
Electrons: Their wavelengths are comparable to atomic dimensions, affecting their behavior in atoms.
Heisenberg Uncertainty Principle
The Heisenberg Uncertainty Principle states that it is impossible to simultaneously know both the exact position and momentum of an electron. This principle limits our ability to describe electrons as particles in fixed orbits.
Schrödinger Equation and Atomic Orbitals
Quantum mechanics describes electrons as wavefunctions (ψ), which give the probability of finding an electron in a particular region of space, called an atomic orbital.
Quantum numbers: Each orbital is defined by a set of quantum numbers derived from the Schrödinger equation.
Electron Configurations and the Periodic Table
Core and Valence Electrons
Electrons in an atom are arranged in shells and subshells. Core electrons are those in filled inner shells, while valence electrons are in the outermost shell and determine chemical reactivity.
Electron configuration: The distribution of electrons among orbitals, e.g., N: 1s2 2s2 2p3
Noble gas configuration: Uses the symbol of the previous noble gas to simplify notation, e.g., [He] 2s2 2p3
The Periodic Table and Periodic Trends
The modern periodic table is organized by increasing atomic number and reflects repeating patterns in electron configurations and properties.
Periodic Trends: Atomic Radius and Ionization Energy
Atomic Radius
The atomic radius is half the distance between the nuclei of two bonded atoms. It varies across the periodic table due to changes in nuclear charge and electron shielding.
Down a group: Atomic radius increases due to additional electron shells.
Across a period: Atomic radius decreases due to increasing effective nuclear charge.


Coulomb's Law and Effective Nuclear Charge
Coulomb's Law describes the force between charged particles:
Effective nuclear charge (Zeff): The net positive charge experienced by valence electrons, calculated as (where Z is the number of protons and S is the number of core electrons).
Ionic Radius
Ions are formed when atoms gain or lose electrons. Cations (positive ions) are smaller than their parent atoms, while anions (negative ions) are larger.
Ionization Energy
Ionization energy is the energy required to remove an electron from a gaseous atom. It is influenced by atomic radius and effective nuclear charge.
Down a group: Ionization energy decreases.
Across a period: Ionization energy increases.
Summary Table: Periodic Trends
Trend | Down a Group | Across a Period |
|---|---|---|
Atomic Radius | Increases | Decreases |
Ionization Energy | Decreases | Increases |
Effective Nuclear Charge | ~Constant | Increases |
Conclusion
The quantum mechanical model of the atom, incorporating the wave-particle duality of light and matter, explains the structure of atoms and the periodic trends observed in the elements. Understanding these principles is essential for further study in chemistry and the behavior of matter at the atomic level.
