 Back
BackQuantum Mechanics, Electron Configurations, and Periodic Trends: Study Notes
Study Guide - Smart Notes

Quantum Mechanical Model of the Atom
Introduction to Quantum Mechanics
The quantum mechanical model describes the behavior of electrons in atoms using mathematical functions and probability. Unlike the Bohr model, which treats electrons as particles in fixed orbits, the quantum model incorporates both wave and particle properties, as described by Schrödinger's equation.
Atomic Orbital: A region in space where there is a high probability of finding an electron.
Schrödinger Equation: The fundamental equation of quantum mechanics for electrons in atoms.
Key equation:
Where is the wave function, is the Hamiltonian operator, and is the energy.
Energy Levels and Electron Transitions
Energy Changes in the Hydrogen Atom
When an electron transitions between energy levels in a hydrogen atom, it absorbs or emits energy in the form of a photon. The energy change is given by:
where J, = energy levels.

Example: Calculate the energy of a photon emitted as an electron relaxes from to using the above formula.
Quantum Numbers and Atomic Orbitals
Quantum Numbers: The Electron's Address
Each electron in an atom is described by a unique set of four quantum numbers, which specify its energy, shape, orientation, and spin.
Quantum Number (QN) | What it Tells Us | Allowed Values |
|---|---|---|
Principal QN, | Energy level/shell and orbital size | 1, 2, 3, ... |
Angular Momentum QN, | Shape and subshell | 0 to |
Magnetic QN, | Specific orbital (orientation) | - to + |
Spin QN, | Direction of spin | +1/2 or -1/2 |
Example: For , possible values are 0 and 1. For , possible values are -1, 0, 1.
Shapes of Atomic Orbitals
The shape of an orbital is determined by the angular momentum quantum number ():
0 | 1 | 2 | 3 | |
|---|---|---|---|---|
Orbital Shape | s | p | d | f |
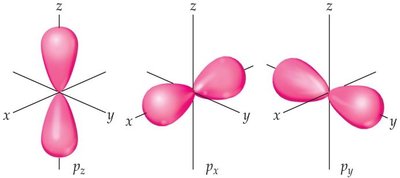
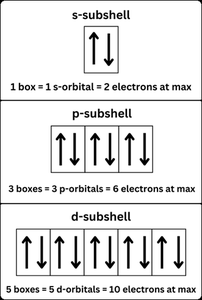
s orbitals: Spherical shape.
p orbitals: Dumbbell-shaped, oriented along x, y, or z axes.
d orbitals: Cloverleaf shapes, more complex orientation.

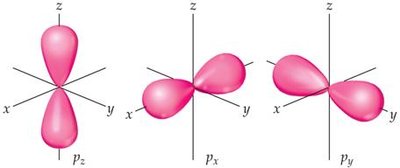
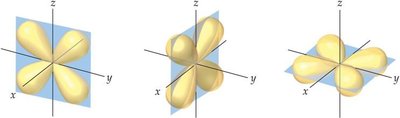
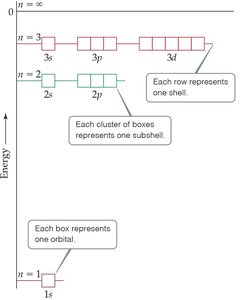
Subshells and Allowed Quantum Numbers
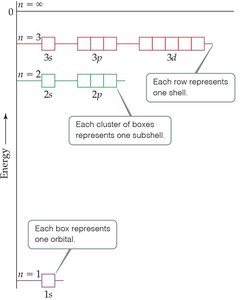
Each principal quantum number contains subshells, each defined by a value of .
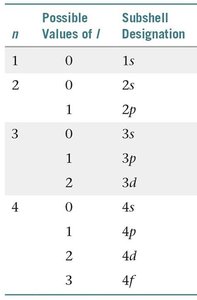
Example: For , possible subshells are 3s (), 3p (), and 3d ().
Electron Configuration and Orbital Diagrams
Filling Order and Principles
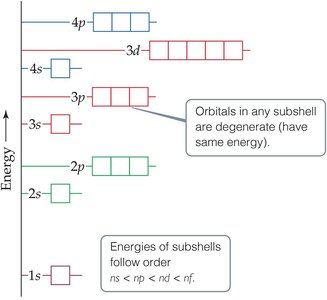
Electrons fill orbitals in a specific order, governed by three main principles:
Aufbau Principle: Electrons occupy the lowest energy orbitals first.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers.
Hund’s Rule: Electrons fill degenerate orbitals singly before pairing up (the "bus seat" rule).
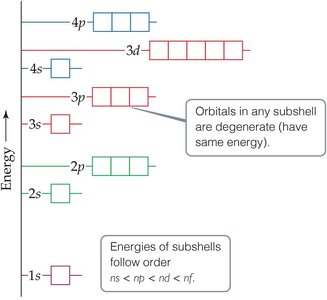
Orbital Energy Diagrams
Orbital diagrams visually represent the arrangement of electrons in an atom. Each box represents an orbital, and arrows represent electrons with their spins.
Electron Configuration Notation
Electron configuration uses numbers and letters to indicate the distribution of electrons among the atom's orbitals. For example, the configuration for oxygen (8 electrons) is:
Oxygen: 1s2 2s2 2p4
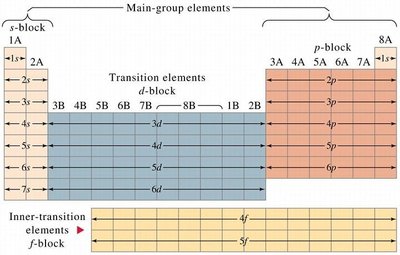
Periodic Table and Electron Configuration
The periodic table is structured according to electron configurations. The s, p, d, and f blocks correspond to the filling of respective subshells.

Example: Scandium (Sc): 1s2 2s2 2p6 3s2 3p6 4s2 3d1
Summary Table: Quantum Numbers and Subshells
n | Possible Values of l | Subshell Designation |
|---|---|---|
1 | 0 | 1s |
2 | 0, 1 | 2s, 2p |
3 | 0, 1, 2 | 3s, 3p, 3d |
4 | 0, 1, 2, 3 | 4s, 4p, 4d, 4f |
Key Learning Objectives
Identify and sketch the shapes of the s, p, and d orbitals.
Write a set of quantum numbers to represent a given electron.
Explain how periodic law is related to the arrangement of electrons.
Define and apply the Aufbau Principle, Pauli Exclusion Principle, and Hund’s Rule in determining electron configurations.
Write electron configurations and draw orbital diagrams for elements in the periodic table.
