 Back
BackQuantum Mechanics, Electron Configurations, and Periodic Trends: Study Guide
Study Guide - Smart Notes

Quantum Mechanical Model of the Atom
Introduction to Quantum Mechanics
The quantum mechanical model describes the behavior of electrons in atoms using mathematical functions and principles. Unlike classical models, it accounts for the probabilistic nature of electron locations and energies.
Quantum mechanics: The branch of physics that explains the behavior of matter and energy at very small scales, including electron configurations and energy levels.
Atomic orbital: A region in space where there is a high probability of finding an electron.
Orbitals are described by mathematical functions, shapes, and quantum numbers.
Quantum Numbers and Their Significance
Types of Quantum Numbers
Quantum numbers are used to describe the properties and locations of electrons within atoms. Each electron in an atom is uniquely identified by a set of four quantum numbers.
Principal Quantum Number (n): Indicates the energy level or shell and orbital size. Values: 1, 2, 3, ...
Angular Momentum Quantum Number (l): Defines the shape and subshell. Values: 0 to n-1.
Magnetic Quantum Number (ml): Specifies the orientation of the orbital. Values: -l to +l.
Spin Quantum Number (ms): Indicates the direction of electron spin. Values: +1/2 or -1/2.
Quantum numbers can be likened to an "address" for electrons, specifying their location and properties within the atom.
Shapes of Atomic Orbitals
s, p, and d Orbitals
Atomic orbitals have distinct shapes, which influence the arrangement of electrons and chemical bonding.
s-orbital: Spherical shape, found in all energy levels.
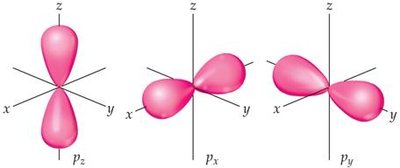
p-orbital: Dumbbell-shaped, oriented along x, y, and z axes.
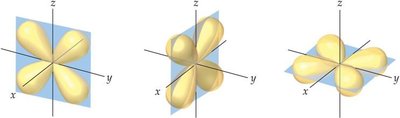
d-orbital: Cloverleaf shapes, more complex, found in higher energy levels.
Example: The 1s orbital is spherical, while the 2p orbitals are oriented along three axes.

Electron Configuration and Orbital Diagrams
Writing Electron Configurations
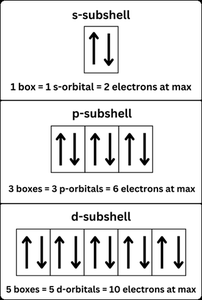
Electron configuration describes the distribution of electrons in an atom's orbitals, following specific rules to determine how electrons fill available energy levels.
Aufbau Principle: Electrons fill the lowest energy orbitals first.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of quantum numbers.
Hund's Rule: Electrons occupy orbitals singly before pairing up.
Example: Oxygen (8 electrons): 1s2 2s2 2p4
Periodic Table and Orbital Blocks
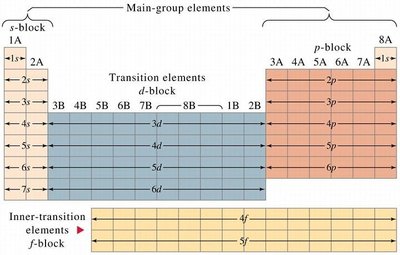
The periodic table is divided into blocks based on the type of orbitals being filled: s-block, p-block, d-block, and f-block. This arrangement helps predict electron configurations and periodic trends.
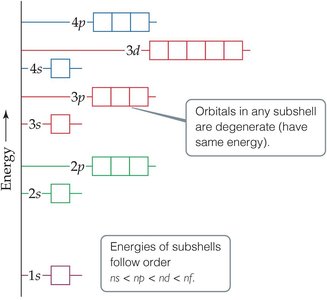
Energy Ordering of Orbitals
Orbitals fill in a specific order based on their energy, which can be visualized in an energy diagram.
Order:
Ground State and Excited State
Definitions and Differences
The ground state is the lowest energy configuration of an atom, where all electrons occupy the lowest available orbitals. An excited state occurs when one or more electrons absorb energy and move to higher energy levels, resulting in a less stable configuration.
Ground state: All electrons are in their lowest energy orbitals according to the Aufbau principle.
Excited state: Electrons have absorbed energy and moved to higher orbitals.
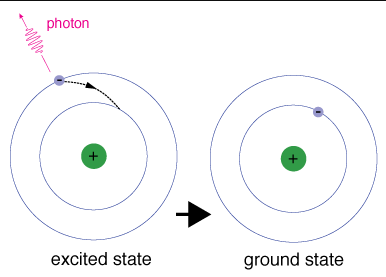
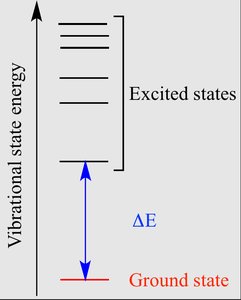
Importance of Ground State
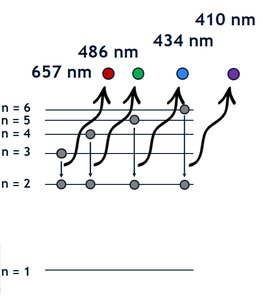
The concept of ground state is fundamental in spectroscopy and chemical reactivity. When an atom transitions from an excited state back to the ground state, it releases energy in the form of light or heat. This process is used to identify elements by their emission spectra.
Atoms tend to seek stability by achieving their lowest energy configuration.
Emission lines are unique to each element and can be used for identification.
Example: Sodium's spectrum is dominated by two yellow lines at 589.0 nm and 589.6 nm.
Key Terms and Concepts
Definitions
Excited state: An electron absorbs energy and moves to a higher energy level.
Electron configuration: The arrangement of electrons in an atom's orbitals.
Quantum mechanics: The study of matter and energy at atomic and subatomic scales.

Summary Table: Ground State Properties
The following table summarizes key properties and implications of the ground state in atoms:
Property | Explanation |
|---|---|
Lowest energy configuration | Electrons fill lowest energy orbitals first (Aufbau principle) |
Unique for each element | Determined by atomic number and electron distribution |
Energy release | Transition from excited to ground state emits energy (light/heat) |
Spectroscopy | Characteristic wavelengths emitted during transitions |
Chemical properties | Stability and reactivity explained by ground state configuration |

Practice and Learning Objectives
Identify and sketch the shapes of s, p, and d orbitals.
Write a set of quantum numbers to represent a given electron.
Explain how periodic law is related to arrangement of electrons.
Define and apply the Aufbau Principle, Pauli Exclusion Principle, and Hund’s Rule in determining electron configurations.
Write electron configurations and draw orbital diagrams for elements in the periodic table.
