 Back
BackL17: Reaction Rates, Rate Laws, and Colligative Properties: Study Notes
Study Guide - Smart Notes

Colligative Properties of Solutions
Raoult’s Law and Deviations
Raoult’s Law describes the vapor pressure of an ideal solution as a linear combination of the vapor pressures of the pure components, weighted by their mole fractions. Deviations from Raoult’s Law occur when solute-solvent interactions differ from those between pure components.
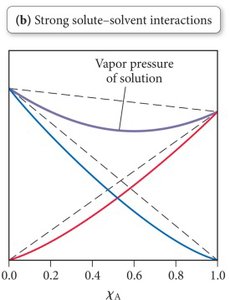
Strong solute-solvent interactions (A-B forces stronger than A-A or B-B): Lower vapor pressure than predicted by Raoult’s Law, especially near equal mole fractions. The solution resists vapor formation due to strong intermolecular forces.
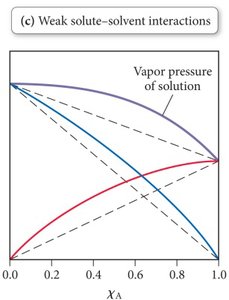
Weak solute-solvent interactions (A-B forces weaker than A-A or B-B): Higher vapor pressure than predicted, as weak interactions allow more molecules to escape into the vapor phase.
Intermolecular forces (IMFs) are always attractive and influence these deviations.
Example: A mixture of water and ethanol shows strong hydrogen bonding, resulting in a lower vapor pressure than predicted for an ideal solution.
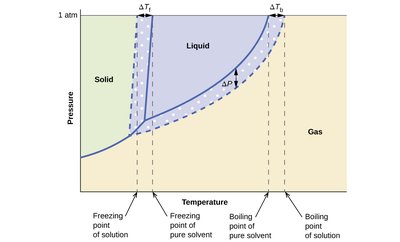
Freezing Point Depression and Boiling Point Elevation
Adding a solute to a solvent lowers the freezing point and raises the boiling point. These effects are called colligative properties and depend on the number of solute particles, not their identity.
Freezing point depression:
Boiling point elevation:
m is the molality of the solution, and are the freezing and boiling point constants for the solvent.
Example: Calculate the boiling point elevation for a solution of 15.6 g ethanol in 625 g water:
Moles ethanol:
Molality:
Boiling point elevation:
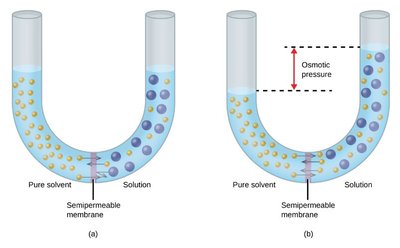
Osmosis and the van't Hoff Factor
Osmosis is the movement of solvent through a semipermeable membrane from a region of lower solute concentration to higher concentration. The osmotic pressure () is given by:
i is the van't Hoff factor (number of particles per formula unit), M is molarity, R is the gas constant, T is temperature in Kelvin.
Example: For a 0.085 M solution of Ca(ClO3)2 at 298 K, (1 Ca2+ + 2 ClO3-):
Chemical Kinetics: Reaction Rates and Rate Laws
Reaction Rate and Relative Rate
The reaction rate is the change in concentration of a reactant or product per unit time. For a general reaction :
Rate =
Negative for reactants (consumed), positive for products (formed).
Example: For :
Rate =
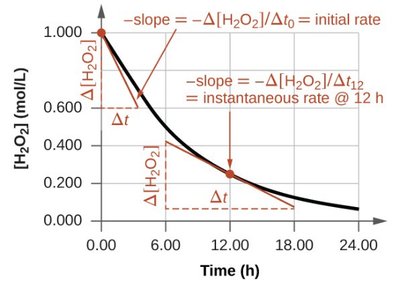
Average and Instantaneous Rates
Average rate is calculated over a time interval; instantaneous rate is the slope of the concentration vs. time curve at a specific moment.
Average rate:
Instantaneous rate: (tangent to curve)
Factors Affecting Reaction Rate
Several factors influence the rate of a chemical reaction:
Nature of reactants: Each reaction has its own intrinsic rate.
Preparation of reactants: Physical state and mixing affect contact and rate.
Temperature: Higher temperature increases rate.
Concentration: Higher concentration increases rate.
Catalysts: Catalysts speed up reactions by providing alternative pathways.
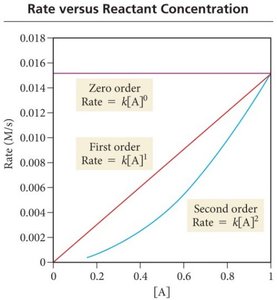
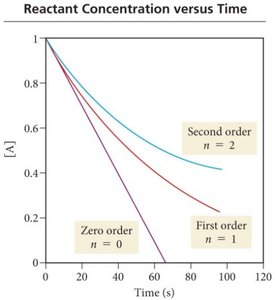
Rate Laws and Reaction Order
A rate law expresses the relationship between reaction rate and reactant concentrations. For a reaction products:
Rate =
n is the reaction order; k is the rate constant.
n can be 0 (zero order), 1 (first order), 2 (second order), or fractional.
Determining Reaction Order: Method of Initial Rates
The method of initial rates involves measuring the initial rate for different reactant concentrations. By comparing how the rate changes when concentration is doubled, the order can be determined:
No change: zeroth order
Rate doubles: first order
Rate quadruples: second order
Mathematically, for two experiments:
Take logarithms to solve for n:
Rate Laws with Multiple Reactants
For reactions involving multiple reactants, the rate law may be:
Rate =
The overall order is .
Orders can be determined by varying one reactant at a time and comparing rates.
Example Table: Determining reaction order for BrO3-, Br-, and H+:
Expt. | [BrO3-] (M) | [Br-] (M) | [H+] (M) | Initial Rate (M/s) |
|---|---|---|---|---|
1 | 0.10 | 0.10 | 0.10 | 8.0 x 10-4 |
2 | 0.20 | 0.10 | 0.10 | 1.6 x 10-3 |
3 | 0.20 | 0.20 | 0.10 | 3.2 x 10-3 |
4 | 0.10 | 0.10 | 0.20 | 3.2 x 10-3 |
Order with respect to BrO3-: 1
Order with respect to Br-: 1
Order with respect to H+: 2
Overall order: 4
Rate law: Rate =
Additional info: Reaction orders can be fractional and may include products or catalysts in advanced cases.
