 Back
BackChapter 4 Chemistry
Study Guide - Smart Notes

Reactions in Aqueous Solution
General Properties of Aqueous Solutions
Solutions are homogeneous mixtures composed of two or more pure substances. The component present in the greatest amount is called the solvent, while the other components are solutes. In aqueous solutions, water acts as the solvent.
Solvent: The substance in greatest abundance (e.g., water in sugar water).
Solute: The substance dissolved by the solvent (e.g., sugar in sugar water).
Electrolytic Properties
Substances in aqueous solution can be classified based on their ability to conduct electricity:
Electrolytes: Substances whose aqueous solutions contain ions and conduct electricity (e.g., NaCl).
Nonelectrolytes: Substances that do not form ions in solution and do not conduct electricity (e.g., C12H22O11).

Ionic Compounds in Water
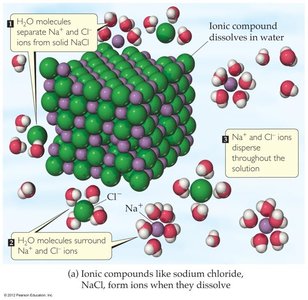
When ionic compounds dissolve in water, they dissociate into their constituent ions. This process is called dissociation:
Dissociation: Separation of ions from the solid structure into the solution.
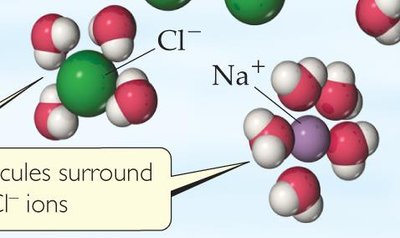
Each ion becomes surrounded by water molecules, a process called solvation or hydration, which prevents recombination of the ions.
Example equation:
Water as an Effective Solvent
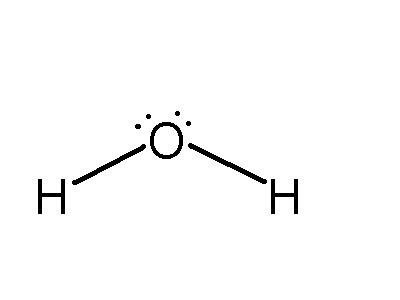
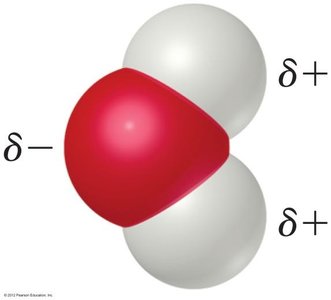
Water is a highly effective solvent for ionic compounds due to its polarity. Although electrically neutral, the water molecule has a partial negative charge near the oxygen atom and partial positive charges near the hydrogen atoms.
The bent molecular geometry and polar covalent bonds create a dipole moment.
This allows water to stabilize ions in solution by surrounding them with the oppositely charged end of the water molecule.
Molecular Compounds in Water
When molecular compounds dissolve in water, they usually remain as intact molecules and do not form ions. Thus, most molecular compounds are nonelectrolytes.
Example: Methanol (CH3OH) dissolves in water as intact molecules.
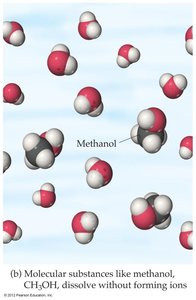
Ionic vs. Molecular Compounds
Ionic compounds are typically formed from metals and nonmetals, while molecular compounds are formed from nonmetals only. Some molecular substances, such as HCl, can ionize in water and behave as electrolytes.
Ionic compounds: NaCl, KCN, MgO, NH4Br
Molecular compounds: CH3OH, C12H22O11, HCl (ionizes in water)
Ammonium ion (NH4+) acts as a pseudo-alkali metal ion.
Strong and Weak Electrolytes
The strength of an electrolyte depends on its ability to exist as ions in solution:
Strong electrolytes: Completely (or nearly completely) ionize in solution (e.g., NaCl).
Weak electrolytes: Partially ionize in solution, with most molecules remaining intact (e.g., acetic acid, CH3COOH).
Example equations:
(100% ionization)
(1% ionization)
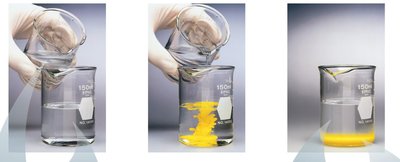
Precipitation Reactions
Precipitation reactions occur when two solutions are mixed and an insoluble product, called a precipitate, forms. This happens when pairs of oppositely charged ions attract each other strongly enough to form an insoluble ionic solid.
Example:
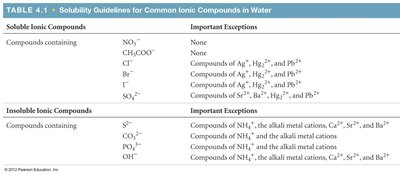
Solubility Guidelines for Ionic Compounds
The solubility of a substance is the amount that can be dissolved in a given quantity of solvent at a specific temperature. Substances with solubility less than 0.01 mol/L are considered insoluble.
Soluble Ionic Compounds | Important Exceptions |
|---|---|
NO3-, CH3COO- | None |
Cl-, Br-, I- | Compounds of Ag+, Hg22+, Pb2+ |
SO42- | Compounds of Sr2+, Ba2+, Hg22+, Pb2+ |
Insoluble Ionic Compounds | Important Exceptions |
S2-, CO32-, PO43-, OH- | Compounds of NH4+, alkali metal cations, Ca2+, Sr2+, Ba2+ |
Exchange (Metathesis) Reactions
Exchange (metathesis) reactions involve the exchange of cations and anions between two compounds. Precipitation and many neutralization reactions follow this pattern:
General form:
Example:
Ionic Equations
There are three types of equations used to describe reactions in aqueous solution:
Molecular equation: Shows all reactants and products as compounds.
Complete ionic equation: Shows all strong electrolytes as ions.
Net ionic equation: Includes only the species that actually participate in the reaction, omitting spectator ions.
Example (precipitation of PbI2):
Molecular:
Complete ionic:
Net ionic:
The sum of ionic charges must be the same on both sides of the balanced equation.
Acids, Bases, and Neutralization Reactions
Acids are substances that ionize in aqueous solution to form hydrogen ions (H+), acting as proton donors. Bases accept H+ ions and often produce hydroxide ions (OH-) in water.
Common acids: HCl, CH3COOH
Common bases: NaOH, KOH, NH3
Example of acid ionization:
Example of base reaction:
Strong and Weak Acids and Bases
Strong acids and bases are completely ionized in solution and are strong electrolytes. Weak acids and bases are only partially ionized and are weak electrolytes.
Strong acids: HCl, HBr, HI, HNO3, H2SO4, HClO3, HClO4
Strong bases: Group 1A metal hydroxides (e.g., NaOH), heavy Group 2A metal hydroxides (e.g., Ba(OH)2)
Neutralization Reactions and Salts
A neutralization reaction occurs when an acid and a base react to form water and a salt. The net ionic equation for a strong acid and strong base is:
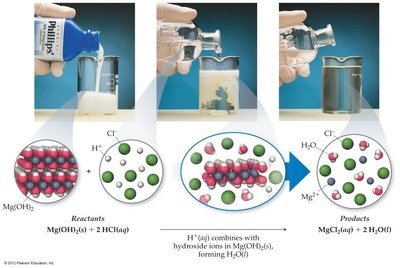
Concentrations of Solutions
Concentration expresses the amount of solute dissolved in a given quantity of solvent or solution. The most common unit is molarity (M):
Other units include parts per million (ppm):
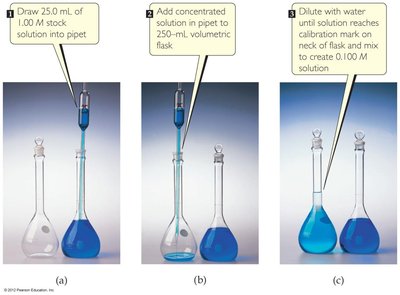
Dilution of Solutions
To prepare a solution of lower concentration from a more concentrated one, water is added. The relationship is:
Solution Stoichiometry
Stoichiometric calculations in solution involve the use of molarity and volume to determine the amount of reactants or products. The key relationships are:
Balanced chemical equations are essential for correct stoichiometric calculations.
Example: How many grams of Ca(OH)2 are needed to neutralize 25.0 mL of 0.100 M HNO3?
Stepwise calculation using molarity, volume, and molar mass.
