 Back
BackReactions in Aqueous Solutions: Electrolytes, Precipitation, Acids & Bases, and Redox
Study Guide - Smart Notes

Reactions in Aqueous Solutions
General Properties of Aqueous Solutions
Aqueous solutions are mixtures where substances are dissolved in water, forming a homogeneous phase. Understanding the components and classifications of these solutions is fundamental in general chemistry.
Solution: A homogeneous mixture of two or more substances, where one or more solutes are dissolved in a solvent.
Solute: The substance present in a lesser amount, dissolved in the solvent.
Solvent: The substance present in the greater amount; in aqueous solutions, the solvent is water.
Example: In a solution of salt water, NaCl is the solute and water is the solvent.
Classification of Solutions: Electrolytes and Nonelectrolytes
Solutions can be classified based on their ability to conduct electricity, which depends on the presence of ions.
Electrolyte: A substance that dissolves in water to produce a solution that conducts electricity due to the presence of ions.
Nonelectrolyte: A substance that dissolves in water but does not produce ions, so the solution does not conduct electricity.
Strong and Weak Electrolytes
Strong Electrolyte: Completely dissociates into ions in solution. Includes soluble ionic compounds, strong acids, and strong bases.
Weak Electrolyte: Partially dissociates into ions in solution. Includes weak acids, weak bases, and some insoluble ionic compounds.
Example (Strong Electrolyte Dissociation):
Example (Weak Electrolyte Dissociation):
Chemical Equilibrium
Chemical equilibrium occurs when the rate of the forward reaction equals the rate of the reverse reaction, resulting in constant concentrations of reactants and products.
Precipitation Reactions and Solubility Guidelines
Precipitation reactions result in the formation of an insoluble product, called a precipitate, when two solutions are mixed. Solubility guidelines help predict whether a precipitate will form.
Precipitate: An insoluble solid formed in a chemical reaction in solution.
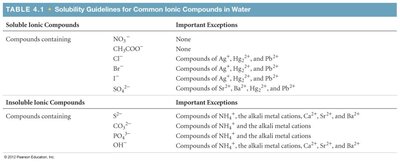
Solubility Guidelines for Common Ionic Compounds in Water | |
Soluble Ionic Compounds | Important Exceptions |
|---|---|
NO3-, CH3COO- | None |
Cl-, Br-, I- | Compounds of Ag+, Hg22+, and Pb2+ |
SO42- | Compounds of Sr2+, Ba2+, Hg22+, and Pb2+ |
Insoluble Ionic Compounds | Important Exceptions |
S2-, CO32-, PO43-, OH- | Compounds of NH4+, alkali metal cations, Ca2+, Sr2+, Ba2+ |
Exchange (Metathesis) Reactions
In exchange (metathesis) reactions, cations and anions exchange partners, often resulting in the formation of a precipitate, gas, or weak electrolyte.
Molecular, Complete Ionic, and Net Ionic Equations
Molecular Equation: Shows the complete chemical formulas of reactants and products.
Complete Ionic Equation: Shows all strong electrolytes as ions.
Net Ionic Equation: Includes only the ions and molecules directly involved in the reaction.
Spectator Ions: Ions that do not participate directly in the reaction.
Acids, Bases, and Neutralization Reactions
Acids and bases are important classes of electrolytes. Their reactions often result in the formation of water and a salt (neutralization).
Acid: A substance that produces H+ ions in aqueous solution.
Base: A substance that produces OH- ions in aqueous solution.
Naming Acids:
If the anion ends in -ide: hydro- prefix, -ic ending (e.g., HCl: hydrochloric acid).
If the anion ends in -ate: -ic ending (e.g., H2SO4: sulfuric acid).
If the anion ends in -ite: -ous ending (e.g., HNO2: nitrous acid).
Strong and Weak Acids and Bases
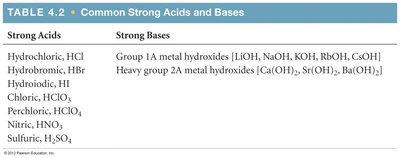
Common Strong Acids and Bases | |
Strong Acids | Strong Bases |
|---|---|
Hydrochloric (HCl) Hydrobromic (HBr) Hydroiodic (HI) Chloric (HClO3) Perchloric (HClO4) Nitric (HNO3) Sulfuric (H2SO4) | Group 1A metal hydroxides (LiOH, NaOH, KOH, RbOH, CsOH) Heavy group 2A metal hydroxides (Ca(OH)2, Sr(OH)2, Ba(OH)2) |
Strong Acid/Base: Completely ionizes in solution.
Weak Acid/Base: Partially ionizes in solution.
Neutralization Reactions
When an acid reacts with a base, water and a salt are formed:
Concentration of Solutions
Concentration expresses the amount of solute in a given quantity of solvent or solution. The most common unit is molarity (M).
Molarity (M):
Solution Stoichiometry and Dilution
Stoichiometry in solutions involves using molarity and volume to calculate the amount of reactants or products. Dilution equations relate concentrations and volumes before and after dilution:
Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between substances. Key terms include:
Oxidation: Loss of electrons by a substance.
Reduction: Gain of electrons by a substance.
Oxidizing Agent: Substance that causes oxidation (is reduced).
Reducing Agent: Substance that causes reduction (is oxidized).
Oxidation Numbers (Oxidation States)
Oxidation numbers help track electron transfer in redox reactions. Key rules include:
Elemental form: 0
Monoatomic ion: equals the ion charge
Sum in a neutral compound: 0; in a polyatomic ion: equals the ion charge
Nonmetals usually negative; exceptions exist
Activity Series of Metals
The activity series ranks metals by their ease of oxidation. A metal higher in the series can reduce the ions of metals below it.
Activity Series of Metals in Aqueous Solution | |
Metal | Oxidation Reaction |
|---|---|
Lithium | Li(s) → Li+(aq) + e- |
Potassium | K(s) → K+(aq) + e- |
Barium | Ba(s) → Ba2+(aq) + 2e- |
Calcium | Ca(s) → Ca2+(aq) + 2e- |
Sodium | Na(s) → Na+(aq) + e- |
Magnesium | Mg(s) → Mg2+(aq) + 2e- |
Aluminum | Al(s) → Al3+(aq) + 3e- |
... | ... |
Gold | Au(s) → Au3+(aq) + 3e- |
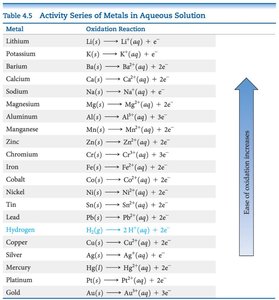
Example: Zinc can displace copper from solution because Zn is above Cu in the activity series.
Additional info: The above notes include expanded definitions, examples, and tables for clarity and completeness, as well as the most relevant images from the provided materials to reinforce key concepts.
