 Back
BackReactivity Series, Displacement Reactions, and Redox Chemistry
Study Guide - Smart Notes

Reactivity Series of Metals
Definition and Arrangement
The reactivity series is a list of metals arranged in order of their reactivity, from most to least reactive. This series helps predict how metals will react with water, acids, and other compounds.
Most reactive: Potassium, sodium, lithium, calcium
Moderately reactive: Magnesium, aluminium, zinc, iron
Least reactive: Copper, silver, gold
Key Point: The position of a metal in the series determines its ability to displace other metals from compounds and its reaction with water and acids. 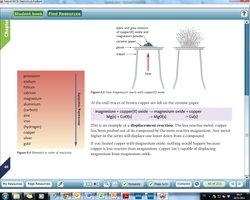
Deduction Using Water and Acids
Metals above hydrogen in the reactivity series react with water or steam to produce hydrogen gas.
Metals reacting with cold water: Potassium, sodium, lithium, calcium
Metals reacting with steam: Magnesium, zinc, iron
Metals below hydrogen: Do not react with water or steam (e.g., copper)
Example:
Potassium + water → potassium hydroxide + hydrogen
Magnesium + steam → magnesium oxide + hydrogen
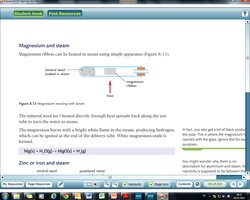
Displacement Reactions
Definition and Explanation
A displacement reaction occurs when a more reactive metal displaces a less reactive metal from its compound.
The more reactive metal replaces the less reactive metal in the compound.
Used to deduce the position of metals in the reactivity series.
Example: Magnesium displaces copper from copper(II) oxide: Additional info: Displacement reactions can also occur in aqueous solutions, where a metal displaces another from its salt. 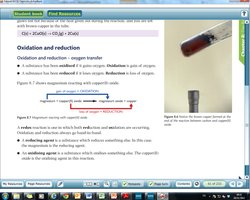
Oxidation and Reduction (Redox)
Definitions
Oxidation: Addition of oxygen or loss of electrons.
Reduction: Removal of oxygen or gain of electrons.
Redox reaction: A chemical reaction involving both oxidation and reduction.
Oxidising agent: Substance that causes oxidation by accepting electrons.
Reducing agent: Substance that causes reduction by donating electrons.
Mnemonic: OILRIG – Oxidation Is Loss (of electrons); Reduction Is Gain (of electrons). 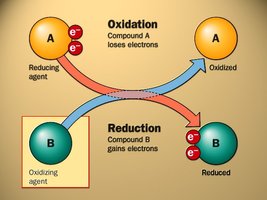
Electron Transfer and Ionic Equations
Reactions involving ions can be represented by ionic equations.
Spectator ions are ions that do not participate in the reaction.
Half equations show the electron transfer for individual substances.
Example:
Oxidation:
Reduction:
Reactions of Metals with Water and Acids
Reactions with Water
Metals above hydrogen react with water or steam to produce hydrogen.
Cold water: Metal hydroxide and hydrogen are formed.
Steam: Metal oxide and hydrogen are formed.
General equations:
Metal + cold water → metal hydroxide + hydrogen
Metal + steam → metal oxide + hydrogen
Reactions with Acids
Metals above hydrogen react with dilute acids to produce salt and hydrogen.
The higher the metal in the series, the more vigorous the reaction.
Metals below hydrogen do not react with simple dilute acids.
Example:
Magnesium + hydrochloric acid → magnesium chloride + hydrogen
Iron + sulfuric acid → iron(II) sulfate + hydrogen
Rusting of Iron and Its Prevention
Conditions for Rusting
Iron rusts in the presence of water and oxygen.
Rusting is accelerated by electrolytes (e.g., salt).
Rust is chemically represented as .
Prevention Methods
Barriers: Paint, oil, grease, plastic, or coating with less reactive metals (e.g., tin).
Alloying: Making stainless steel with chromium or nickel.
Galvanising: Coating iron with zinc.
Sacrificial Protection
Zinc (more reactive than iron) corrodes instead of iron when used as a coating.
Even if the zinc layer is scratched, zinc acts as a sacrificial anode and protects iron.
Electrons from zinc flow to iron, preventing iron from forming ions and rusting.
Example: Zinc blocks are attached to boat hulls to protect iron from rusting.
Summary Table: Reactivity Series and Metal Reactions
Metal | Reaction with Water | Reaction with Acid | Position in Series |
|---|---|---|---|
Potassium | Very vigorous | Too reactive | Top |
Sodium | Very vigorous | Too reactive | Top |
Lithium | Vigorous | Too reactive | Top |
Calcium | Gentle | Can react if acid is very dilute | High |
Magnesium | Almost no reaction (cold water), reacts with steam | Vigorous | Moderate |
Zinc | Reacts with steam | Slow (cold), faster (hot) | Moderate |
Iron | Reacts with steam | Slow (cold), faster (hot) | Moderate |
Copper | No reaction | No reaction | Low |
Silver | No reaction | No reaction | Bottom |
Gold | No reaction | No reaction | Bottom |
Additional info: The table summarizes the main reactions and positions of metals in the reactivity series, aiding prediction of their chemical behavior.
