 Back
BackSolids and Modern Materials: Structure, Properties, and Applications
Study Guide - Smart Notes

Solids and Modern Materials
Graphene
Graphene is a revolutionary material in modern chemistry, consisting of a single layer of carbon atoms arranged in a hexagonal lattice. It is renowned for its exceptional properties, including strength, conductivity, and transparency.
Thinnest and strongest material: Only one atom thick, yet about 100 times stronger than steel.
Conductivity: Excellent conductor of heat and electricity; can be fully charged within seconds.
Transparency and impermeability: Transparent and impermeable to all substances.
Applications: Used in faster computers, foldable touchscreens, ultrathin light panels, and super-strong bulletproof vests.

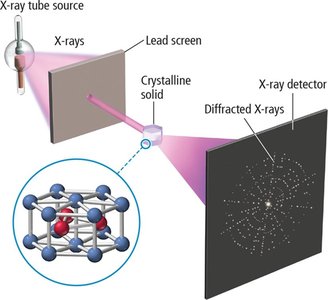
X-Ray Crystallography
Principles and Applications
X-ray crystallography is a technique used to determine the arrangement of atoms within a crystal. It relies on the diffraction of X-rays by the atomic planes in a crystalline solid.
X-ray diffraction: X-rays of a specific wavelength scatter from atomic planes, forming diffraction patterns.
Measurement: Diffraction angles are measured to determine atomic spacing and arrangement.
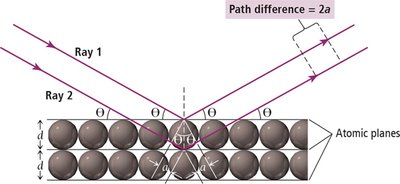
Bragg’s Law: The relationship between wavelength (\( \lambda \)), spacing (\( d \)), and diffraction angle (\( \theta \)) is given by: where
\( d \): spacing between planes
\( \theta \): angle of diffraction
\( \lambda \): wavelength
\( n \): order of diffraction
Unit Cells and Basic Structures
Types of Unit Cells
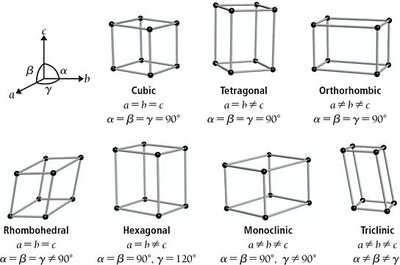
The arrangement of atoms in a crystal is described by unit cells, the smallest repeating units. Unit cells are classified by their symmetry and geometry.
Cubic: All sides and angles equal.
Tetragonal, Orthorhombic, Rhombohedral, Hexagonal, Monoclinic, Triclinic: Differ in side lengths and angles.
Cubic Crystal Lattices
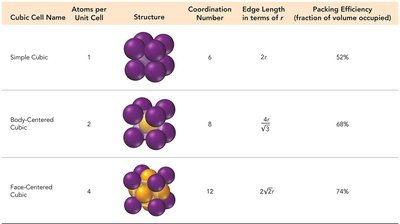
Cubic arrangements include simple cubic, body-centered cubic, and face-centered cubic lattices. Each type has a distinct structure, coordination number, and packing efficiency.
Cubic Cell Name | Atoms per Unit Cell | Structure | Coordination Number | Edge Length | Packing Efficiency |
|---|---|---|---|---|---|
Simple Cubic | 1 | Corner atoms | 6 | 52% | |
Body-Centered Cubic | 2 | Corner + center atom | 8 | 68% | |
Face-Centered Cubic | 4 | Corner + face atoms | 12 | 74% |
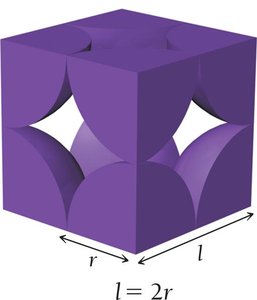
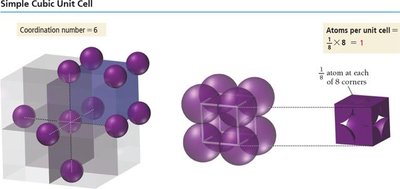
Simple Cubic Unit Cell
8 corners, each occupied by 1/8 of an atom.
Number of atoms per unit cell:
Edge length:
Coordination number: 6
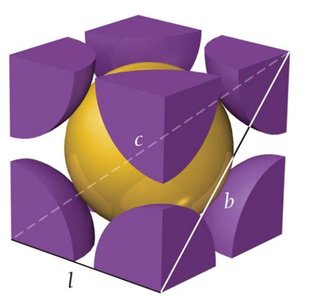
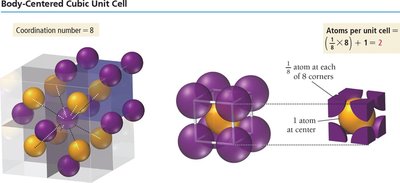
Body-Centered Cubic Unit Cell
1 atom at center, 8 corners each with 1/8 atom.
Number of atoms per unit cell:
Edge length:
Coordination number: 8
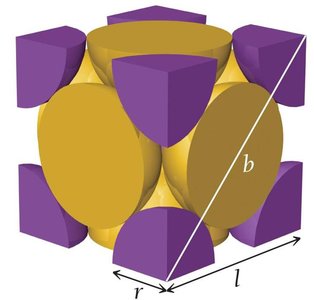
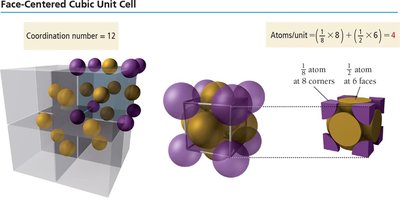
Face-Centered Cubic Unit Cell
8 corners (1/8 atom each), 6 faces (1/2 atom each).
Number of atoms per unit cell:
Edge length:
Coordination number: 12
Classifying Crystalline Solids
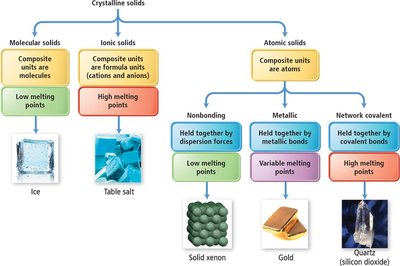
Crystalline solids are classified based on the nature of their composite units: molecular, ionic, or atomic.
Molecular solids: Composed of molecules held by intermolecular forces (dispersion, dipole-dipole, hydrogen bonding). Low melting points.
Ionic solids: Composed of ions held by strong electrostatic forces. High melting points.
Atomic solids: Composed of atoms. Subtypes include network covalent, nonbonding, and metallic solids.
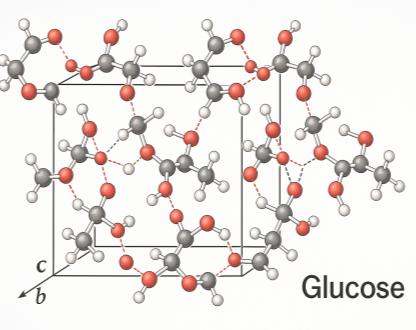
Molecular Solids
Examples: CO2, H2O, C6H12O6, C12H22O11
Held together by weak intermolecular forces.
Polymorphs: Different crystalline forms with distinct properties.
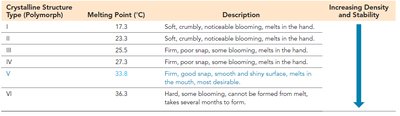
Ionic Solids
Composed of formula units (cations and anions).
Held by strong electrostatic attraction.
High melting points; coordination number depends on ion size and structure.
Atomic Solids
Network covalent: Covalent bonds, high melting points (e.g., graphite, diamond).
Nonbonding: Held by dispersion forces, very low melting points (e.g., solid noble gases).
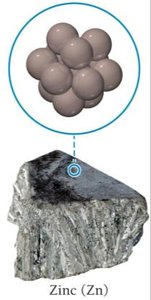
Metallic: Held by metallic bonds, variable melting points, closest-packed structures (e.g., gold, zinc).


The Structure of Ionic Solids
Ionic solids have structures closely related to unit cells, maintaining charge neutrality. Coordination number is determined by the number of cations surrounding an anion or vice versa, influenced by ion size ratio.
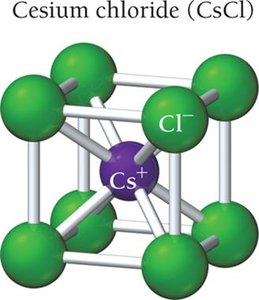
Cesium Chloride (CsCl)
Simple cubic structure.
Cs+ at center, Cl- at corners.
Coordination number: 8.
1:1 ratio in unit cell.
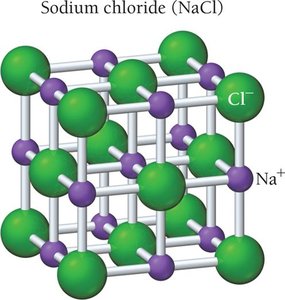
Sodium Chloride (NaCl)
Face-centered cubic structure (rock salt).
Na+ at center and edges, Cl- at corners and faces.
Coordination number: 6.
1:1 ratio in unit cell.
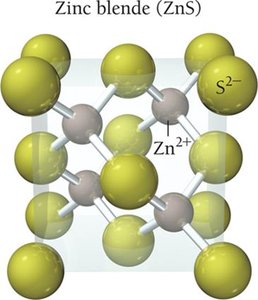
Zinc Sulfide (ZnS)
Face-centered cubic structure (zinc blende).
Zn2+ in tetrahedral holes, S2- at corners and faces.
Coordination number: 4.
1:1 ratio in unit cell.
Network Covalent Atomic Solids
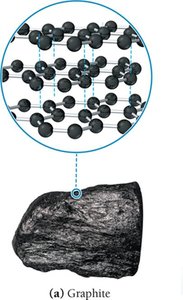
Carbon: Graphite
Flat sheets of carbon atoms in hexagonal rings.
SP2 hybridization; delocalized pi electrons enable conductivity.
Sheets held by dispersion forces; slippery nature.
Used as lubricant, electrical conductor, and for thermal management.
Carbon: Diamond
Tetrahedral rings of covalently bonded carbon atoms.
SP3 hybridization; all electrons form covalent bonds.
Rigid, strong, and dense; used for cutting tools.
Does not conduct electricity; chemically nonreactive.

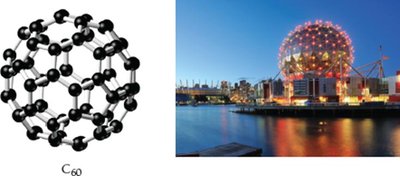
Carbon: Buckminsterfullerene (C60)
Soccer-ball-shaped clusters of 60 carbon atoms.
Known as fullerenes or buckyballs.
Used in drug delivery, antioxidants, solar cells, lubricants.

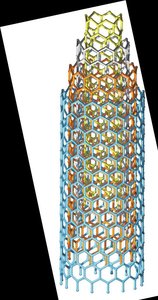
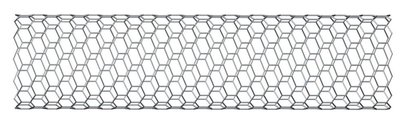
Carbon: Nanotubes
Sheets of interconnected C6 rings in cylindrical shape.
Single-walled or multiwalled.
Ultralight, extremely strong; used in electronics, sports equipment.
Silicates
Network covalent solids of Si and O atoms.
Si bonded tetrahedrally to four O atoms; O bonded to two Si atoms.
Quartz (SiO2) is the most common structure.
Constitute about 90% of Earth's crust; used in glass, ceramics, cement.
Ceramics, Cement, and Glass
Ceramics
Prepared from powders mixed with water.
Types: Silicate ceramics (clay), oxide ceramics (Al2O3, MgO), nonoxide ceramics (Si3N4, BN, SiC).
Used in plates, insulators, furnaces, cutting tools, engine parts.

Cement
Powdered mixture of limestone (CaCO3) and silica (SiO2).
Hardens by reacting with water, forming Si-O-Si bridges.
Used in construction; combined with sand and pebbles to make concrete.

Glass
Amorphous solid made by rapid cooling of molten silica.
Types: Soda-lime glass (windows), borosilicate glass (labware), leaded glass (decorative items).
Properties vary with composition; used in windows, cookware, decorative items.

Semiconductors and Band Theory
Band Theory
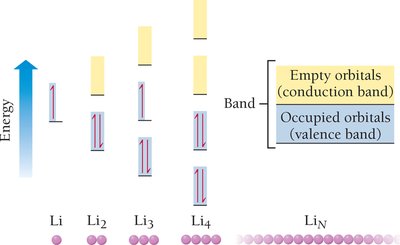
Band theory explains the electronic structure of solids, based on molecular orbital theory. Atomic orbitals combine to form bands of energy levels.
Valence band: Bonding orbitals.
Conduction band: Antibonding orbitals.
Band gap: Energy difference between bands; determines conductivity.
Band Gap and Conductivity
Conductors: Infinitesimal band gap; electrons move freely.
Semiconductors: Moderate band gap; limited electron movement.
Insulators: Large band gap; electrons cannot move, no conductivity.
Group 4A Elements and Band Gap
Element | Atomic Radius (pm) | Band Gap | Classification |
|---|---|---|---|
Carbon (diamond) | 77 | 5.5 eV | Insulator |
Silicon | 118 | 1.11 eV | Semiconductor |
Germanium | 122 | 0.67 eV | Semiconductor |
Tin | 140 | 0.08 eV | Metal |
Lead | 180 | none | Metal |
Doping: Controlling Conductivity
Doping: Adding impurities to increase conductivity.
n-type: Doped with elements having more valence electrons (e.g., phosphorus).
p-type: Doped with elements having fewer valence electrons (e.g., gallium).
p–n junction: Combination of p-type and n-type semiconductors; used in diodes and amplifiers.
Polymers and Plastics
Polymers
Large molecules made by linking monomers.
Natural (starches, proteins, DNA) or synthetic (plastic bottles, PVC pipes).
Polymerization
Addition polymerization: Monomers link without eliminating atoms.
Condensation polymerization: Elimination of atoms or small groups during reaction.
Copolymers: Formed from two different monomers.
Polymers of Commercial Importance
Polymer | Structure | Uses |
|---|---|---|
Polyethylene | Repeating CH2 units | Films, packaging, bottles |
Polypropylene | Repeating CH2-CH(CH3) units | Kitchenware, fibers, appliances |
Polystyrene | Repeating CH2-CH(phenyl) units | Packaging, disposable containers, insulation |
Polyvinyl chloride | Repeating CH2-CH(Cl) units | Pipe fittings, clear film |
Polyurethane | Repeating units with NH, C=O, O, R, R' | Foam, insulation, automotive parts |
Polyethylene terephthalate | Repeating units with O, CH2, C=O, aromatic ring | Tire cord, apparel, bottles |
Nylon 6,6 | Repeating units with NH, (CH2)6, C=O, (CH2)4, C=O | Furnishings, apparel, carpet fibers |
Additional info: The notes expand on brief points with academic context, definitions, and examples to ensure completeness and clarity for exam preparation.
