 Back
BackSolids and Modern Materials: Structure, Types, and Properties
Study Guide - Smart Notes

Solids and Modern Materials
Introduction to Solids and Crystal Structures
Solids are characterized by a fixed arrangement of particles, resulting in definite shape and volume. The study of solids involves understanding their microscopic arrangement, which determines their macroscopic properties. X-ray crystallography is a key technique used to determine the regular arrangements of atoms within a crystalline solid, known as the crystal lattice. The smallest repeating unit that displays the full symmetry of the arrangement is called the unit cell.
Crystal lattice: The three-dimensional arrangement of particles in a crystalline solid.
Unit cell: The smallest repeating unit in a crystal lattice that shows the pattern of arrangement for all the particles.
Unit cells are classified by their symmetry and are repeated to form the entire crystal.
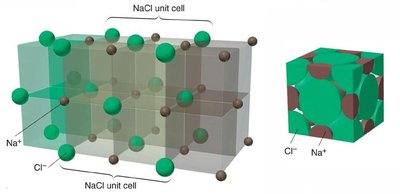

Types of Unit Cells and Their Properties
Unit cells are classified based on their geometry and the arrangement of particles. The most common types are cubic unit cells, which include simple cubic, body-centered cubic, and face-centered cubic structures.
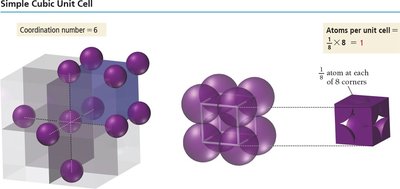
Coordination number: The number of other particles each particle is in contact with. For ions, it refers to the number of oppositely charged ions in contact.
Packing efficiency: The percentage of volume in the unit cell occupied by particles. Higher coordination numbers generally mean higher packing efficiency and stronger attractive forces.
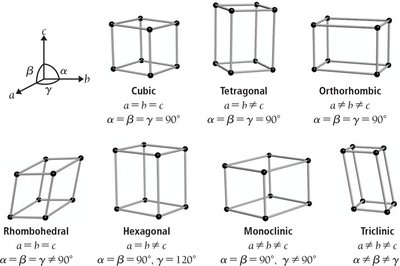
Cubic Unit Cells
Cubic unit cells have all edges of equal length and all angles at 90°. There are three main types:
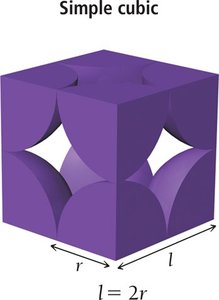
Simple cubic (SC): Particles at each corner of the cube. Coordination number = 6. Packing efficiency = 52%.
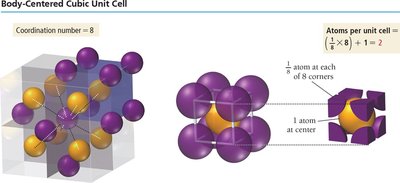
Body-centered cubic (BCC): Particles at each corner and one in the center. Coordination number = 8. Packing efficiency = 68%.
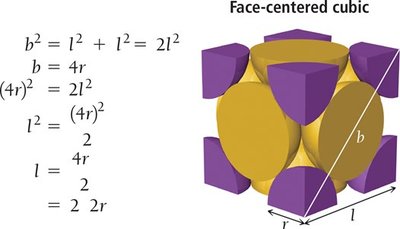
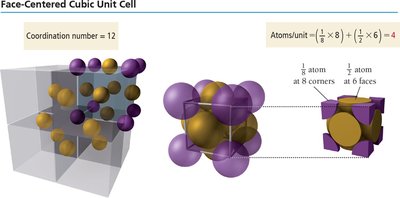
Face-centered cubic (FCC): Particles at each corner and one at the center of each face. Coordination number = 12. Packing efficiency = 74%.
Cubic Cell Name | Atoms per Unit Cell | Coordination Number | Edge Length (in terms of r) | Packing Efficiency |
|---|---|---|---|---|
Simple Cubic | 1 | 6 | 2r | 52% |
Body-Centered Cubic | 2 | 8 | \( \frac{4r}{\sqrt{3}} \) | 68% |
Face-Centered Cubic | 4 | 12 | \( 2\sqrt{2}r \) | 74% |
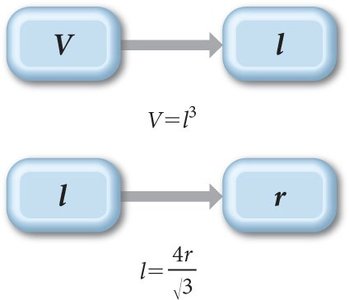
Mathematical Relationships in Unit Cells
Understanding the relationships between the volume, edge length, and atomic radius in unit cells is essential for solving problems related to crystal structures.
For a cube:
For body-centered cubic:
For face-centered cubic:

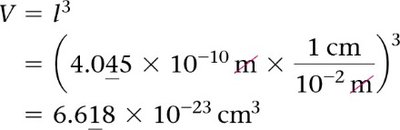
Classification of Crystalline Solids
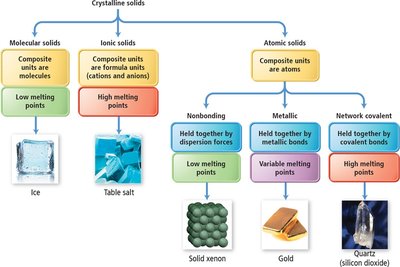
Crystalline solids are classified based on the nature of their constituent particles and the forces holding them together:
Molecular solids: Composite units are molecules, held together by intermolecular forces (dispersion, dipole-dipole, hydrogen bonds). Low melting points.
Ionic solids: Composite units are ions, held together by strong coulombic (ionic) forces. High melting points.
Atomic solids: Composite units are atoms. Subdivided into nonbonding (dispersion forces), metallic (metallic bonds), and network covalent (covalent bonds) solids.
Molecular Solids
Molecular solids have molecules at their lattice sites and are held together by relatively weak intermolecular forces, resulting in low melting points. Examples include CO2, H2O, and C12H22O11. Some molecular solids can exist in different crystalline forms, known as polymorphs, which have different properties and are important in pharmaceuticals.
Ionic Solids
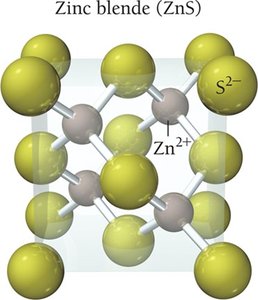
Ionic solids are composed of cations and anions arranged in a lattice and held together by strong ionic bonds. These solids have high melting points and are generally harder than molecular solids. The structure must accommodate both cations and anions, and the coordination number reflects the number of close cation–anion interactions, which increases stability.
Metallic Bonding and Atomic Solids
Metallic solids consist of metal atoms that release their valence electrons, forming a 'sea' of mobile electrons around fixed metal cations. This electron sea model explains properties such as electrical conductivity, malleability, and ductility. Atomic solids can also be nonbonding (noble gases), metallic, or network covalent (e.g., diamond, graphite).

Network Covalent Solids: Carbon Allotropes
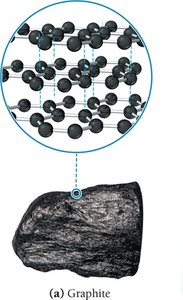
Network covalent solids are held together by covalent bonds throughout the structure, resulting in very high melting points and unique properties. Carbon exists as two important network covalent solids: graphite and diamond.
Graphite: Carbon atoms form flat sheets of fused six-membered rings (sp2 hybridization), with weak dispersion forces between sheets. Properties include high melting point, electrical conductivity, and lubricity.
Diamond: Each carbon atom forms four covalent bonds (sp3 hybridization) in a tetrahedral geometry, making diamond extremely hard, a thermal conductor, and an electrical insulator.

Polymers: Structure and Types
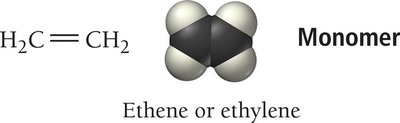
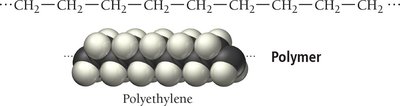
Polymers are very large molecules formed by linking together many small units called monomers. They can be natural (e.g., starch, proteins, DNA) or synthetic (e.g., plastics, nylon). Polymerization can occur via addition (no atoms lost) or condensation (small molecules eliminated). Copolymers contain two different monomers, and dimers are formed from two monomers.
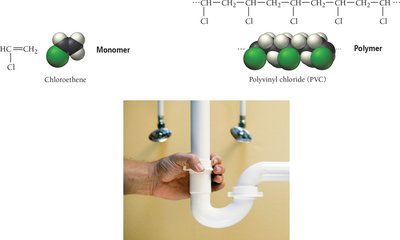
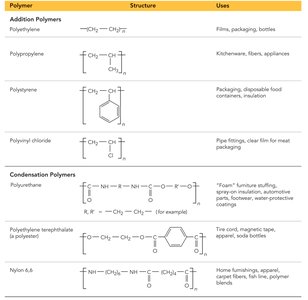
Additional info: The classification and properties of solids are foundational for understanding materials science, nanotechnology, and the development of new materials for industrial and technological applications.
