Back
BackSolids and Modern Materials: Structure, Types, and Properties
Study Guide - Smart Notes

Solids and Modern Materials
Introduction to Solids
Solids are a fundamental state of matter characterized by closely packed particles arranged in a fixed, orderly pattern. The study of solids involves understanding their microscopic structure, types, and the forces that hold them together. Modern materials science explores these structures to develop new materials with desirable properties.
Unit Cells and Crystal Lattices
Crystal Lattice and Unit Cell
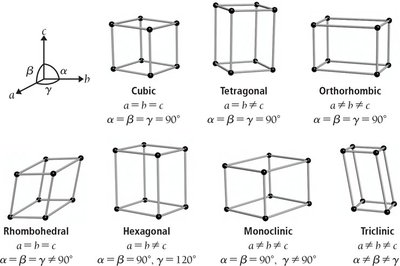
The crystal lattice is the regular, repeating arrangement of atoms, ions, or molecules in a crystalline solid. The unit cell is the smallest repeating unit that displays the full symmetry of the arrangement and, when repeated in three dimensions, creates the entire lattice. X-ray crystallography is a key technique for determining these arrangements.
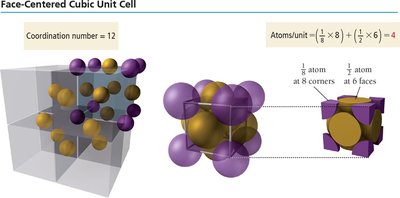
Coordination number: The number of nearest neighbors surrounding a particle in the lattice.
Packing efficiency: The percentage of space occupied by particles in the unit cell.
Types of Unit Cells
Unit cells are classified by their symmetry and the arrangement of particles. The most common types in cubic systems are:
Simple cubic (SC)
Body-centered cubic (BCC)
Face-centered cubic (FCC)
Cubic Unit Cells
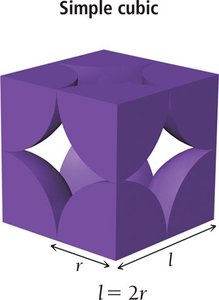
Simple Cubic Unit Cell
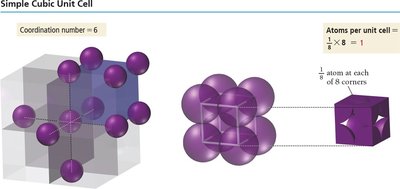
In the simple cubic structure, atoms are located at each corner of the cube. Each corner atom is shared among eight adjacent cubes, so only 1/8 of each atom belongs to one unit cell, resulting in one atom per unit cell. The edge length is twice the atomic radius, and the coordination number is 6.
Edge length:
Atoms per unit cell:
Coordination number: 6
Body-Centered Cubic (BCC) Unit Cell
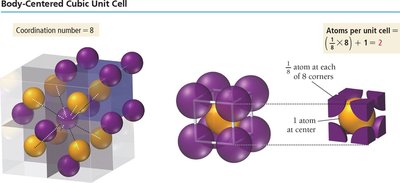
The BCC structure has atoms at each corner and one atom at the center of the cube. Each corner atom is shared among eight cubes, and the center atom belongs entirely to the unit cell, resulting in two atoms per unit cell. The coordination number is 8.
Edge length:
Atoms per unit cell:
Coordination number: 8
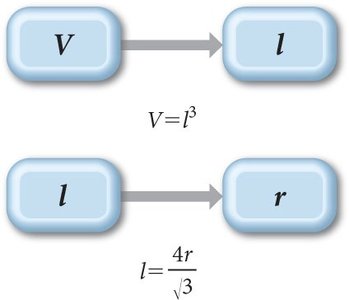
Relating Unit Cell Volume, Edge Length, and Atomic Radius
To find the atomic radius from the unit cell volume in a BCC structure:
Volume of cube:
Edge length:
Solve for given .
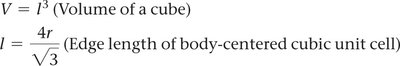

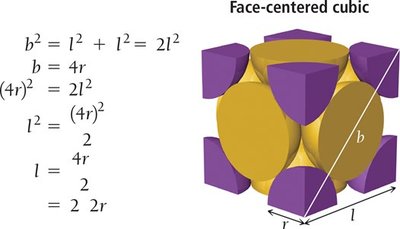
Face-Centered Cubic (FCC) Unit Cell
The FCC structure has atoms at each corner and at the center of each face. Each corner atom is shared among eight cubes, and each face atom is shared between two cubes, resulting in four atoms per unit cell. The coordination number is 12.
Edge length:
Atoms per unit cell:
Coordination number: 12
Density and Crystal Structure
The density of a crystalline solid can be calculated using the mass and volume of the unit cell:
Density:
Mass of unit cell: number of atoms per cell × mass of each atom
Volume:
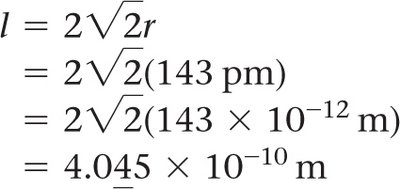
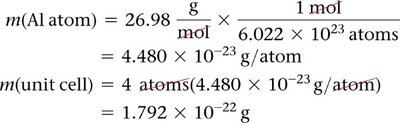
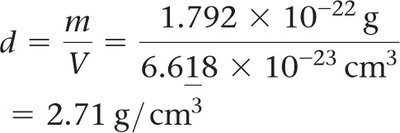
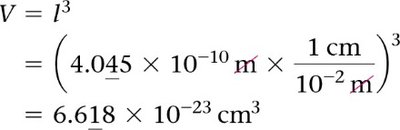
Comparison of Cubic Lattices
Cubic Cell Name | Atoms per Unit Cell | Coordination Number | Edge Length (in terms of r) | Packing Efficiency |
|---|---|---|---|---|
Simple Cubic | 1 | 6 | 2r | 52% |
Body-Centered Cubic | 2 | 8 | 4r/√3 | 68% |
Face-Centered Cubic | 4 | 12 | 2√2 r | 74% |
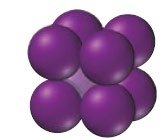
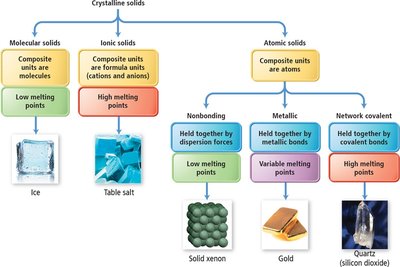
Classification of Crystalline Solids
Types of Crystalline Solids
Crystalline solids are classified based on the nature of their constituent particles and the forces holding them together:
Molecular solids: Composite units are molecules; held by intermolecular forces; low melting points.
Ionic solids: Composite units are ions; held by ionic bonds; high melting points.
Atomic solids: Composite units are atoms; can be nonbonding, metallic, or network covalent.
Molecular Solids
Molecular solids have molecules at their lattice points, held together by dispersion forces, dipole-dipole interactions, or hydrogen bonds. They generally have low melting points and may exist in different forms (polymorphs).
Examples: CO2, H2O, C12H22O11
Polymorphs: Different crystal structures with different properties
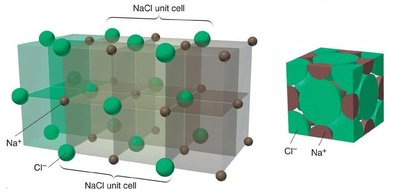
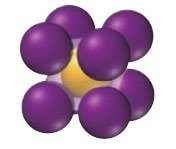
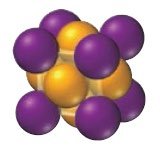
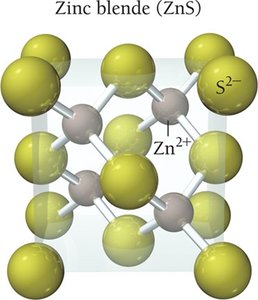
Ionic Solids
Ionic solids are composed of cations and anions held together by strong electrostatic (Coulombic) forces. They have high melting points and their structure must accommodate both types of ions. The coordination number depends on the relative sizes of the ions.
Example: NaCl
Coordination number: Number of close cation–anion interactions
Atomic Solids
Atomic solids are classified by the forces holding the atoms together:
Nonbonding atomic solids: Held by dispersion forces (e.g., solid noble gases); very low melting points.
Metallic atomic solids: Held by metallic bonds; variable melting points; high electrical conductivity.
Network covalent solids: Held by covalent bonds; very high melting points; rigid structures.

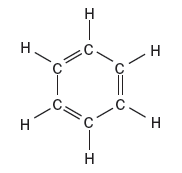
Network Covalent Solids: Carbon Allotropes
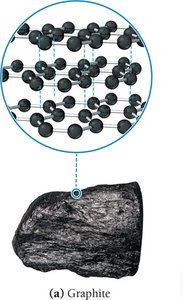
Graphite
Graphite consists of layers of carbon atoms arranged in hexagonal rings. Each carbon is sp2 hybridized, forming three sigma bonds and one pi bond, resulting in trigonal planar geometry. The layers are held together by dispersion forces, allowing them to slide past each other, making graphite a good lubricant and electrical conductor.
Bond length: 142 pm
Properties: High melting point, slippery, electrical conductor, thermal insulator, chemically nonreactive
Diamond
In diamond, each carbon atom is sp3 hybridized and forms four sigma bonds in a tetrahedral geometry, creating a rigid, three-dimensional network. This structure gives diamond its exceptional hardness, high melting point, and thermal conductivity, but it is an electrical insulator.
Properties: Very high melting point, very hard, electrical insulator, best known thermal conductor, chemically nonreactive

Polymers
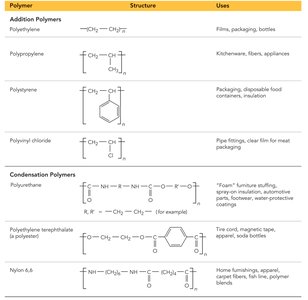
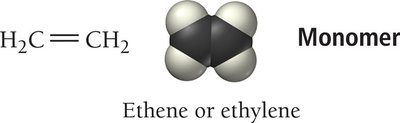
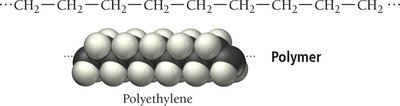
Introduction to Polymers
Polymers are large molecules formed by the repeated linking of smaller units called monomers. They can be natural (e.g., proteins, DNA) or synthetic (e.g., plastics, nylon).
Polymerization
Polymerization is the process of joining monomers to form a polymer. There are two main types:
Addition polymerization: Monomers link without eliminating atoms.
Condensation polymerization: Monomers link with the elimination of a small molecule (e.g., water).
Copolymer: Polymer made from two different monomers.
Dimer: Product of two monomers joined together.
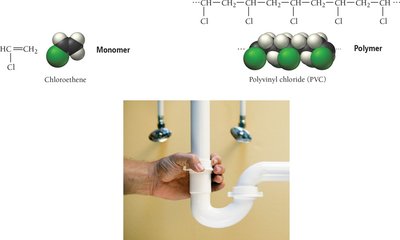
Polymers of Commercial Importance
Polymers are widely used in everyday life, from packaging materials to fibers and appliances. Their properties depend on the monomers used and the type of polymerization.