 Back
BackChap 12 lecture 1 study guide chem 136
Study Guide - Smart Notes

Solids, Liquids, and Intermolecular Forces
Introduction
This chapter explores the nature of solids and liquids, focusing on the forces that hold molecules together and influence their physical properties. Understanding these forces is essential for predicting the behavior of substances under various conditions.
Difference between Intermolecular and Intramolecular Forces
Definitions and Key Differences
Intramolecular Forces: These are the chemical bonds (such as covalent, ionic, or metallic bonds) that hold atoms together within a molecule. They are responsible for the chemical identity and properties of the molecule.
Intermolecular Forces (IMFs): These are the forces of attraction or repulsion between neighboring molecules. They are much weaker than intramolecular forces and primarily influence the physical properties of substances, such as boiling point, melting point, and state of matter.
Example: In hydrogen chloride (HCl), the covalent bond between H and Cl is an intramolecular force, while the attraction between two HCl molecules is an intermolecular force.
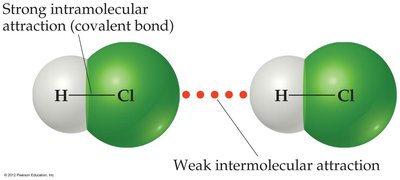
Additional info: Intermolecular forces involve smaller charges at greater distances, while intramolecular forces involve larger charges at very close distances. This explains why breaking a chemical bond requires much more energy than overcoming intermolecular attractions.
Importance of Intermolecular Forces
Influence on Physical Properties
The strength of IMFs determines whether a substance is a solid, liquid, or gas at room temperature.
Stronger IMFs lead to higher boiling and melting points, greater viscosity, and higher surface tension.
Substances with weak IMFs are typically gases at room temperature, while those with moderate to strong IMFs are liquids or solids.
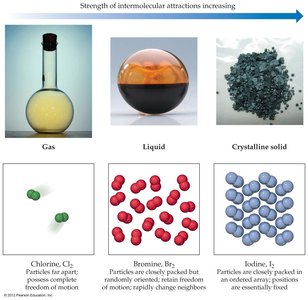
Example: Chlorine (Cl2) is a gas, bromine (Br2) is a liquid, and iodine (I2) is a solid at room temperature due to increasing strength of IMFs.
Application: Hydrogen bonding, a type of IMF, is crucial for the structure of biological molecules such as DNA, where it holds the two strands together.
Types of Intermolecular Forces
Overview
London Dispersion Forces (LDFs): Present in all molecules and atoms, caused by temporary fluctuations in electron distribution.
Dipole-Dipole Forces: Occur between polar molecules due to permanent dipoles.
Hydrogen Bonding: A special, strong type of dipole-dipole interaction involving H bonded to N, O, or F.
Ion-Dipole Forces: Occur between ions and polar molecules, especially important in solutions.
London Dispersion Forces (LDFs)
Origin and Mechanism
London dispersion forces arise from momentary fluctuations in the electron distribution within atoms or molecules, creating instantaneous dipoles that induce dipoles in neighboring particles. These are the only IMFs present in nonpolar molecules and noble gases.
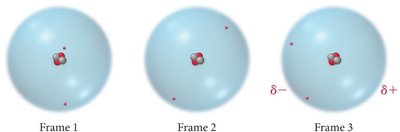
When an instantaneous dipole forms in one atom, it can induce a dipole in a neighboring atom, resulting in a weak attraction between them.

Factors Affecting the Strength of London Dispersion Forces
Polarizability: The ease with which the electron cloud can be distorted. Larger atoms/molecules with more electrons are more polarizable, leading to stronger LDFs.
Molar Mass: Higher molar mass generally means more electrons and a larger, more easily distorted electron cloud, increasing LDF strength.
Molecular Shape: Molecules with greater surface area (e.g., long chains) have stronger LDFs than compact, spherical molecules of the same molar mass.
Examples and Applications
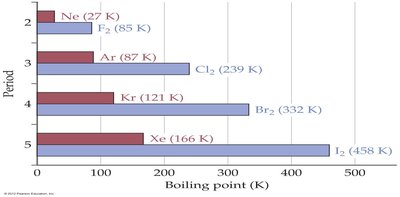
Halogens: F2 (gas) has weaker LDFs than Br2 (liquid) due to fewer electrons and lower polarizability.
Noble Gases: Boiling points increase with molar mass due to stronger LDFs.
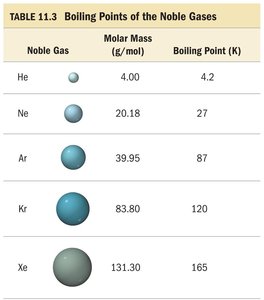
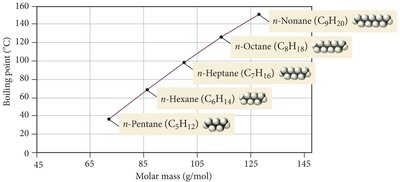
n-Alkanes: As the molar mass of n-alkanes increases, so does the boiling point, reflecting stronger LDFs.
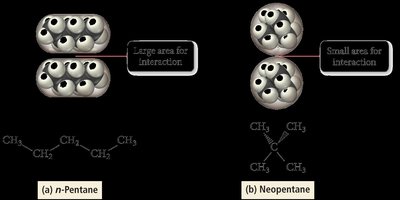
Molecular Shape and LDFs
For molecules with the same molar mass, shape affects the strength of LDFs. Straight-chain isomers (e.g., n-pentane) have higher boiling points than branched isomers (e.g., neopentane) due to greater surface area for intermolecular contact.
Summary of London Dispersion Forces
LDFs are present in all molecules and atoms, regardless of polarity.
They result from temporary fluctuations in electron distribution, not from permanent dipoles or electronegativity differences.
LDFs are weak in small molecules but can be significant in large, heavy atoms/molecules.
The strength of LDFs increases with the number of electrons, molar mass, and molecular surface area.
Ranking London Dispersion Forces
Among a series of molecules, the one with the most electrons (highest molar mass) will have the strongest LDFs and, typically, the highest melting and boiling points.
