 Back
BackSolids, Liquids, and Intermolecular Forces: Dipole-Dipole, Hydrogen Bonding, and Ion-Dipole Interactions
Study Guide - Smart Notes

Solids, Liquids, and Intermolecular Forces
Dipole-Dipole Intermolecular Forces
Dipole-dipole forces are a type of intermolecular force that occurs between polar molecules. These forces arise due to the presence of permanent dipoles within molecules, which are regions of partial positive and negative charge resulting from differences in electronegativity between atoms.
Definition: Dipole-dipole forces exist between molecules with permanent dipoles (polar molecules).
Dipole Moment: The dipole moment (\( \mu \)) is a quantitative measure of the polarity of a molecule, given by \( \mu = q \times r \), where \( q \) is the charge and \( r \) is the distance between charges. Dipole moments are reported in Debyes (D), where 1 D = 3.34 × 10-30 C·m.
Vector Nature: The overall dipole moment of a molecule is the vector sum of individual bond dipoles.
Example: Hydrogen fluoride (HF) is a polar molecule because fluorine is highly electronegative, pulling electron density toward itself and creating a partial negative charge on F and a partial positive charge on H.
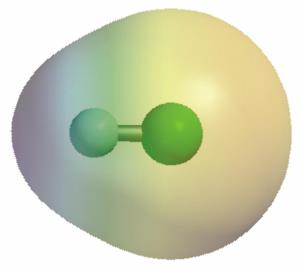
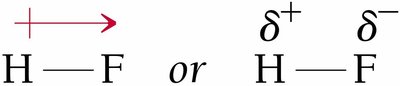
Polarity and Molecular Geometry
The presence of polar bonds does not guarantee that a molecule is polar. The molecular geometry, determined by VSEPR theory, must be considered to determine the net dipole moment.
Linear Molecule Example: CO2 has polar bonds, but the dipoles cancel due to its linear geometry, making it nonpolar.
Tetrahedral Molecule Example: CH4 is nonpolar because the dipoles cancel out in its tetrahedral geometry.
Example: CH2Cl2 is polar because the dipoles do not cancel.
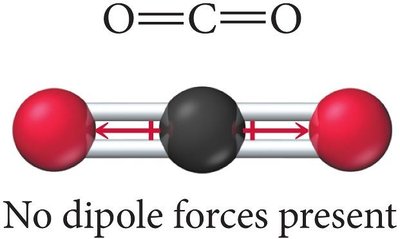
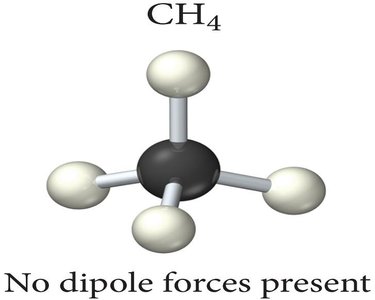
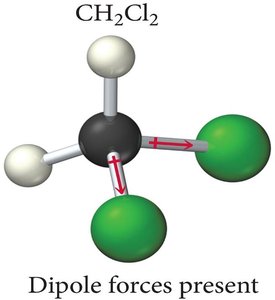
Identifying Dipole-Dipole Attraction
To determine if a molecule exhibits dipole-dipole attraction, assess both the presence of polar bonds and the molecular geometry.
Practice: HCl, CH3Cl, and NH3 are polar and exhibit dipole-dipole forces. BF3 and CCl4 are nonpolar due to their geometry.
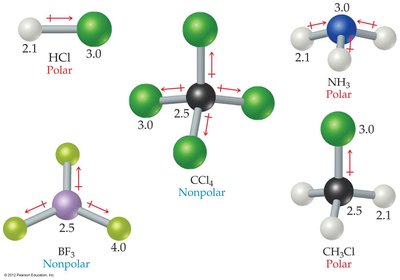
Physical Properties Influenced by Dipole-Dipole Forces
Polar molecules with similar shape and mass have higher melting and boiling points than nonpolar molecules due to stronger dipole-dipole attractions.
Example: Boiling points increase with increasing polarity and dipole moment.
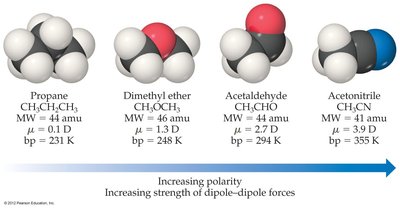
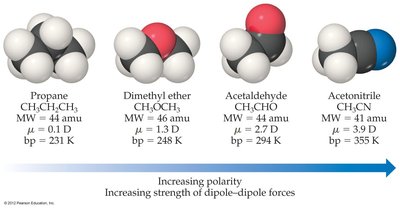
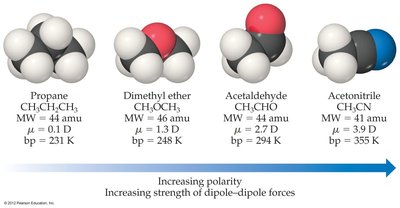
Miscibility and Polarity
The polarity of a molecule determines its miscibility with other substances. "Like dissolves like" is a guiding principle: polar liquids mix with polar liquids, and nonpolar liquids mix with nonpolar liquids.
Examples: Water (polar) is not miscible with n-hexane (nonpolar); methanol and water (both polar) are miscible; CCl4 and n-hexane (both nonpolar) are miscible.
Hydrogen Bonding
Hydrogen bonding is a special type of dipole-dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms (F, O, or N). This results in a strong attraction between the hydrogen atom of one molecule and the electronegative atom of another.
Definition: Hydrogen bonding is an attractive force between a hydrogen atom bonded to F, O, or N and the F, O, or N atom of another molecule.
Examples: HF, H2O, and NH3 exhibit hydrogen bonding.
Energy: The average energy of a hydrogen bond is about 40 kJ/mol.
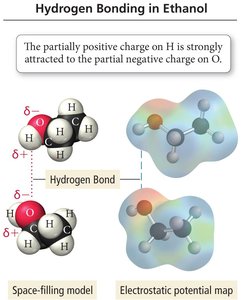
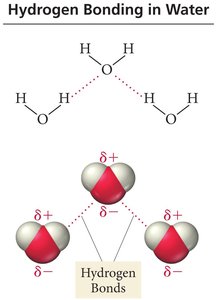
Unique Properties of Water
Hydrogen bonding gives water unique properties, such as expansion upon freezing and a high boiling point relative to its molar mass. Each water molecule can form four hydrogen bonds due to its two O-H bonds and two lone pairs on oxygen.
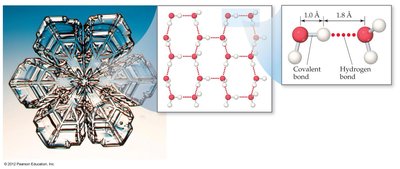
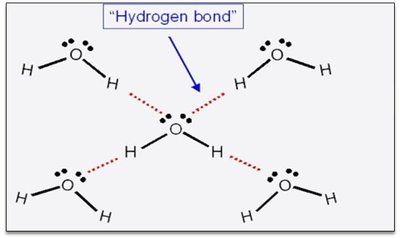
Other Molecules with Hydrogen Bonding
Examples: Ammonia (NH3), hydrogen fluoride (HF), hydrogen peroxide (H2O2), methanol (CH3OH), acetic acid (CH3COOH), methylamine (CH3NH2).
Effect of Hydrogen Bonding on Physical Properties
Hydrogen bonding is stronger than London dispersion and dipole-dipole forces. Substances capable of hydrogen bonding have higher melting and boiling points.
Example: Boiling point trends for hydrides of groups 4A and 6A show that H2O has a much higher boiling point due to hydrogen bonding.
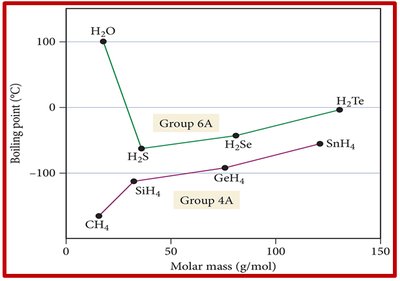
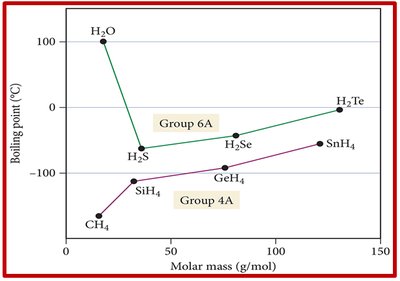
Hydrogen Bonding and Liquid State
Among compounds with similar molar masses and dipole moments, only those with hydrogen bonding are liquid at room temperature.
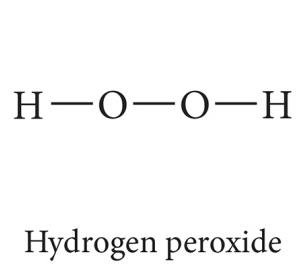
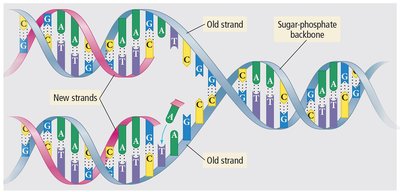
Hydrogen Bonding in DNA
Hydrogen bonds are crucial for the structure of DNA, holding the individual bases together.
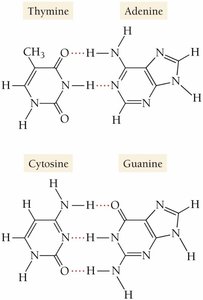
Distinguishing Hydrogen Bonding from Chemical Bonds
Hydrogen bonding is an intermolecular force, not a chemical bond. Chemical bonds occur within molecules, while hydrogen bonding occurs between molecules.
Ion-Dipole Intermolecular Forces
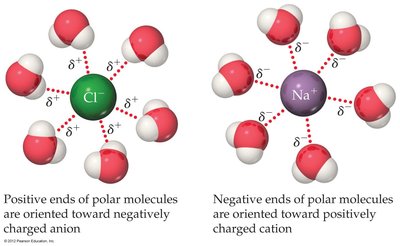
Ion-dipole forces are Coulombic attractions between ions and polar molecules, especially important in aqueous solutions.
Definition: Ion-dipole interactions occur between ions (cations or anions) and polar molecules.
Example: In aqueous sodium chloride, water molecules orient their dipoles toward Na+ and Cl- ions.
Strength: The strength of ion-dipole forces depends on the size and charge of the ion and the magnitude of the dipole moment. Cations interact more strongly due to their smaller size.
Summary Table: Types of Intermolecular Forces
Type | Occurs Between | Relative Strength | Example |
|---|---|---|---|
London Dispersion | All molecules | Weakest | CH4 |
Dipole-Dipole | Polar molecules | Intermediate | HCl, CH3Cl |
Hydrogen Bonding | H bonded to F, O, N | Strong | H2O, NH3 |
Ion-Dipole | Ions and polar molecules | Strongest | NaCl in water |
