 Back
BackChap 12 lecture 3 study guide chem 136
Study Guide - Smart Notes

Solids, Liquids, and Intermolecular Forces
Difference Between Intermolecular and Intramolecular Forces
Understanding the distinction between intermolecular and intramolecular forces is fundamental in chemistry. Intramolecular forces are the chemical bonds (such as covalent, ionic, or metallic bonds) that hold atoms together within a molecule. Intermolecular forces are the forces of attraction or repulsion between neighboring molecules, affecting physical properties like boiling and melting points.
Types of Intermolecular Forces
Intermolecular forces vary in strength and origin. The main types are:
London Dispersion Forces (LDFs): Present in all molecules and atoms, increasing with molecular weight.
Dipole-Dipole Forces: Occur between polar molecules; strength increases with dipole moment.
Hydrogen Bonding: Strongest among the three, present when H is bonded to F, O, or N.
Ion-Dipole Forces: Occur in solutions of ionic compounds and polar solvents; very strong and important in aqueous solutions.
Hydrogen Bonding
Hydrogen bonding is an attractive force between a hydrogen atom bonded to a highly electronegative atom (F, O, or N) and the F, O, or N atom of another molecule. This interaction is much stronger than typical dipole-dipole or dispersion forces.
Atoms involved: H, F, O, N only
Examples: HF, H2O, NH3, ethanol, hydrogen peroxide, acetic acid, methylamine
Average energy: 40 kJ/mol
Hydrogen bonding arises because highly electronegative atoms pull bonding electrons away from hydrogen, leaving its nucleus unshielded and highly positive, which is attracted to lone pairs on neighboring molecules.
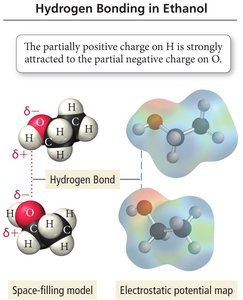
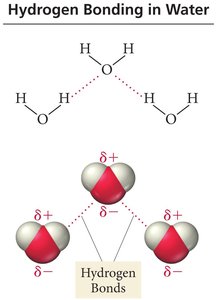
Unique Properties of Water
Water exhibits several unique properties due to hydrogen bonding:
Water expands upon freezing, making ice less dense than liquid water.
Each water molecule can form four hydrogen bonds, resulting in a high boiling point for its low molar mass.
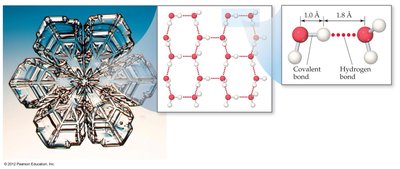
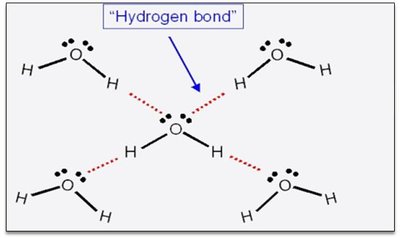
Other Examples of Hydrogen Bonding
Ammonia (NH3)
Hydrogen fluoride (HF)
Hydrogen peroxide (H2O2)
Methanol (CH3OH)
Acetic acid (CH3COOH)
Methylamine (CH3NH2)
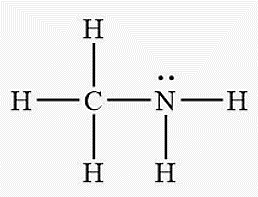
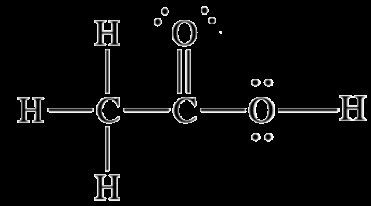
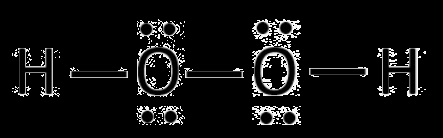
Effect of Hydrogen Bonding on Physical Properties
Hydrogen bonding significantly increases melting and boiling points compared to substances with only LDFs or dipole-dipole forces. For example, hydrides of Group 6A (like H2O) have much higher boiling points than Group 4A hydrides due to hydrogen bonding.
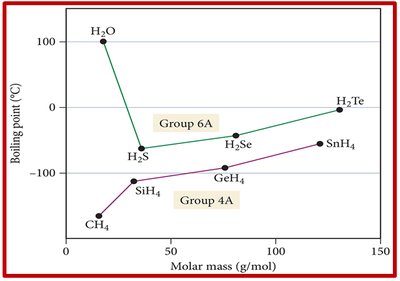
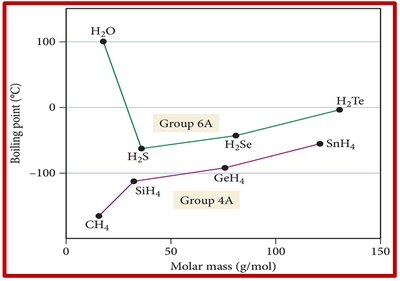
Comparing Compounds with Similar Molar Mass
Compounds with similar molar masses and dipole moments may differ in physical state due to hydrogen bonding. For example, hydrogen peroxide (H-O-O-H) is liquid at room temperature because it exhibits hydrogen bonding, while similar compounds without hydrogen bonding are not.
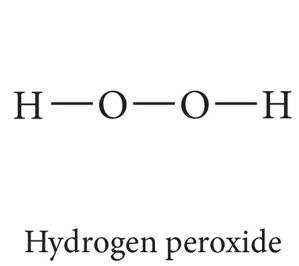
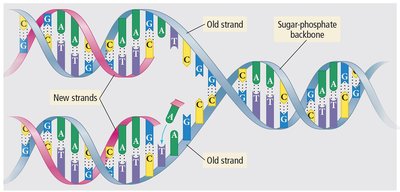
Hydrogen Bonding in DNA
Hydrogen bonds are crucial in biological systems, such as the structure of DNA, where they hold the base pairs together.
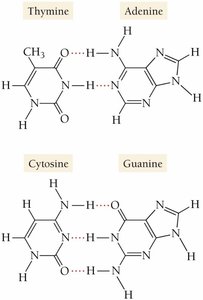
Important Distinction
Hydrogen bonding is an intermolecular force, not a chemical bond. Chemical bonds occur within molecules, while hydrogen bonds occur between molecules.
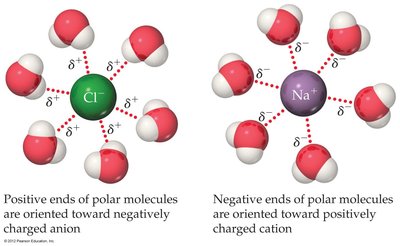
Ion-Dipole Forces
Ion-dipole interactions are Coulombic attractions between ions and polar molecules, especially important in aqueous solutions. The strength depends on ion size, charge, and dipole moment. Cations interact more strongly than anions due to their smaller size.
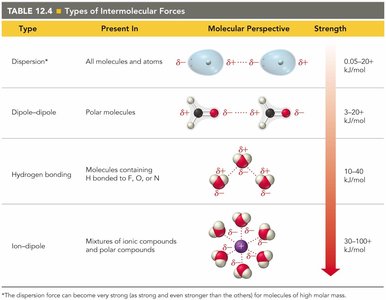
Summary Table: Types of Intermolecular Forces
Type | Present In | Molecular Perspective | Strength |
|---|---|---|---|
Dispersion | All molecules and atoms | Temporary dipoles | 0.05–20 kJ/mol |
Dipole-dipole | Polar molecules | Permanent dipoles | 3–20 kJ/mol |
Hydrogen bonding | H bonded to F, O, or N | Special strong dipole | 10–40 kJ/mol |
Ion-dipole | Ions and polar molecules | Ion and dipole | 30–100+ kJ/mol |
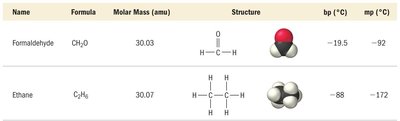
Comparison of Intermolecular Forces
For substances with similar molar mass and shape, LDFs are similar, but differences in dipole-dipole or hydrogen bonding determine overall strength. For substances with very different molar mass, LDFs dominate if hydrogen bonding is absent.
Practice: Identifying Intermolecular Forces
Compound | LDF | D-D | HB |
|---|---|---|---|
PCl3 | yes | yes | no |
CH3F | yes | yes | no |
I2 | yes | no | no |
HF | yes | yes | yes |
HCl | yes | yes | no |
CH2O | yes | yes | no |
CH3OH | yes | yes | yes |
BF3 | yes | no | no |
Intermolecular Forces and Physical Properties
Surface Tension, Viscosity, and Capillary Action
Surface Tension
Surface tension is the net inward pull experienced by molecules at the surface of a liquid, causing the surface to tighten like an elastic film. It is the energy required to increase the surface area of a liquid by a unit amount.
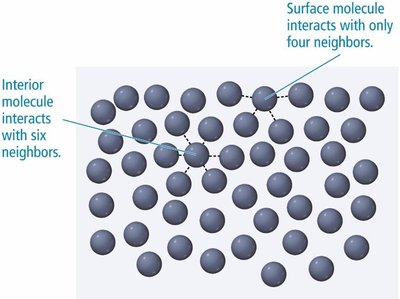
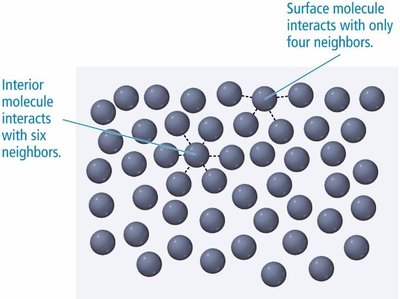
Water has a much higher surface tension than ethanol due to extensive hydrogen bonding.
Substance | Surface Tension (J/m2) |
|---|---|
Water, H2O | 7.3×10‒2 |
Ethanol, C2H5OH | 2.3×10‒2 |
Factors Affecting Surface Tension
Magnitude of IMFs: Stronger IMFs yield higher surface tension.
Temperature: Surface tension decreases as temperature increases due to increased molecular motion.
Viscosity
Viscosity is the resistance to flow in liquids and gases. Higher viscosity means slower flow. It is measured in kg/m·s or poise (P).
Strong IMFs increase viscosity.
Viscosity increases with molar mass.
Viscosity decreases with increasing temperature.
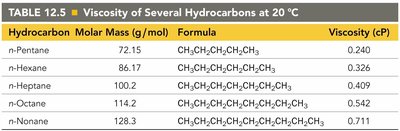
Hydrocarbon | Molar Mass (g/mol) | Formula | Viscosity (cP) |
|---|---|---|---|
n-Pentane | 72.15 | CH3CH2CH2CH2CH3 | 0.240 |
n-Hexane | 86.17 | CH3CH2CH2CH2CH2CH3 | 0.326 |
n-Heptane | 100.2 | CH3CH2CH2CH2CH2CH2CH3 | 0.409 |
n-Octane | 114.2 | CH3CH2CH2CH2CH2CH2CH2CH3 | 0.542 |
n-Nonane | 128.3 | CH3CH2CH2CH2CH2CH2CH2CH2CH3 | 0.711 |
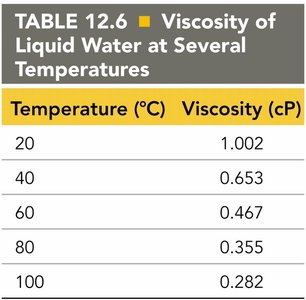
Temperature (°C) | Viscosity (cP) |
|---|---|
20 | 1.002 |
40 | 0.653 |
60 | 0.467 |
80 | 0.355 |
100 | 0.282 |
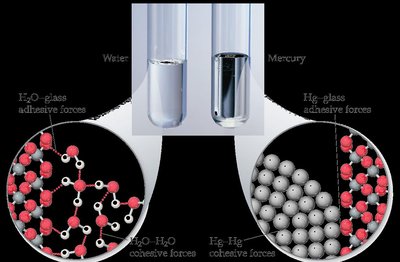
Capillary Action
Capillary action is the rise of a liquid through a narrow tube against gravity, resulting from the competition between cohesive (IMFs within the liquid) and adhesive (IMFs between liquid and surface) forces.
If adhesive forces > cohesive forces: liquid rises, concave meniscus (e.g., water).
If cohesive forces > adhesive forces: liquid does not rise, convex meniscus (e.g., mercury).
Additional info:
These notes cover the essential concepts of intermolecular forces, their types, and their effects on physical properties, as outlined in Chapter 12 of a general chemistry course. The included tables and images reinforce key points and provide visual context for understanding molecular interactions.
