 Back
BackSolids, Liquids, and Intermolecular Forces: Study Guide
Study Guide - Smart Notes

Solids, Liquids, and Intermolecular Forces
Introduction
This chapter explores the nature of solids, liquids, and the forces that hold molecules together. Understanding intermolecular forces is essential for predicting physical properties such as boiling point, melting point, and state of matter.
Difference Between Intermolecular and Intramolecular Forces
Definitions and Comparisons
Intermolecular forces are the attractions between molecules, while intramolecular forces are the chemical bonds within a molecule. These two types of forces differ in strength and their impact on physical and chemical properties.
Intermolecular Forces: Occur between neighboring molecules; affect physical properties (e.g., boiling point, melting point).
Intramolecular Forces: Present within a molecule; are the chemical bonds (e.g., covalent, ionic) joining atoms; influence chemical properties.
Relative Strength: Intermolecular forces are generally much weaker than intramolecular forces.
Example: Vaporization breaks intermolecular forces, while breaking covalent bonds requires much more energy.
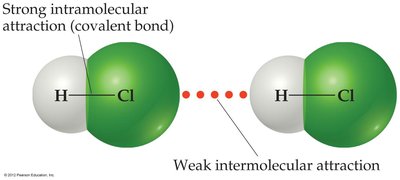
Importance of Intermolecular Forces
Physical Properties and States of Matter
The strength of intermolecular forces determines whether a substance is a solid, liquid, or gas at room temperature. Stronger intermolecular forces result in higher boiling and melting points.
Physical Properties Affected: Vapor pressure, boiling point, melting point, viscosity, surface tension.
State of Matter: Substances with strong intermolecular forces are solids or liquids; those with weak forces are gases.
Example: Chlorine (Cl2) is a gas, bromine (Br2) is a liquid, and iodine (I2) is a solid at room temperature due to increasing intermolecular attractions.
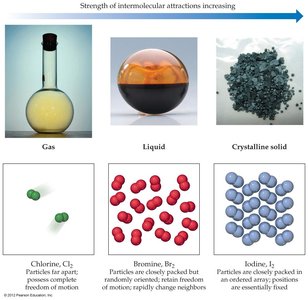
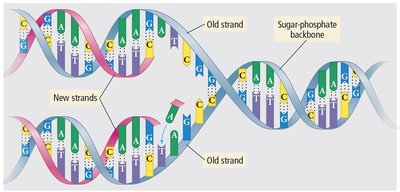
Biological Relevance
Intermolecular forces, such as hydrogen bonding, are crucial for the structure and function of biological molecules like DNA.
Example: Hydrogen bonds between DNA bases stabilize the double helix structure.
Types of Intermolecular Forces
Overview
There are several types of intermolecular forces, each with distinct origins and effects:
London Dispersion Forces (LDFs): Present in all molecules, caused by temporary fluctuations in electron distribution.
Dipole-Dipole Forces: Occur between polar molecules.
Hydrogen Bonding: A special type of dipole-dipole interaction involving H bonded to N, O, or F.
Ion-Dipole Forces: Occur between ions and polar molecules, especially in solutions.
London Dispersion Forces (LDFs)
LDFs arise from momentary uneven electron distributions, creating instantaneous and induced dipoles. These forces are present in all atoms and molecules, regardless of polarity.
Origin: Fluctuations in electron distribution create temporary dipoles.
Induced Dipole: A temporary dipole in one molecule induces a dipole in a neighboring molecule.
Strength: Generally weak, but can be significant in large atoms/molecules.
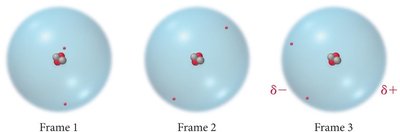


Factors Affecting London Dispersion Forces
The strength of LDFs depends on several factors:
Polarizability: The ease with which the electron cloud can be distorted. Larger atoms/molecules with more electrons are more polarizable.
Molar Mass: Higher molar mass means more electrons and stronger LDFs.
Molecular Shape: Long, chain-like molecules have more surface area for interaction, leading to stronger LDFs than compact, spherical molecules.
Polarizability Example: F2 vs. Br2
Br2 has a larger, more easily polarized electron cloud than F2, resulting in stronger LDFs and a higher boiling point.
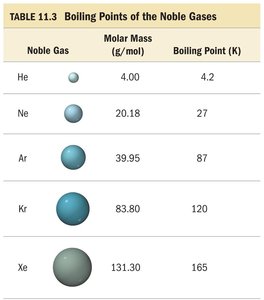
Molar Mass Example: Noble Gases
As molar mass increases among noble gases, boiling points rise due to stronger LDFs.
Noble Gas | Molar Mass (g/mol) | Boiling Point (K) |
|---|---|---|
He | 4.00 | 4.2 |
Ne | 20.18 | 27 |
Ar | 39.95 | 87 |
Kr | 83.80 | 120 |
Xe | 131.30 | 165 |
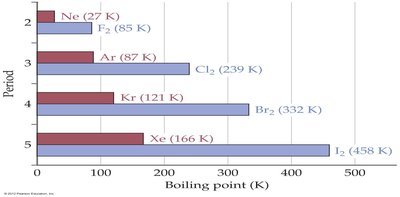
Molar Mass Example: Halogens and Noble Gases
Boiling points of halogens and noble gases increase with period due to increasing molar mass and stronger LDFs.
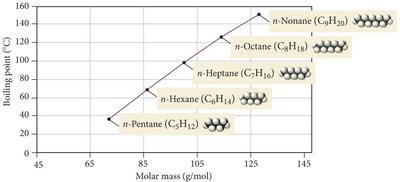
Molecular Shape Example: n-Alkanes
Boiling points of n-alkanes increase with molar mass, reflecting stronger LDFs.
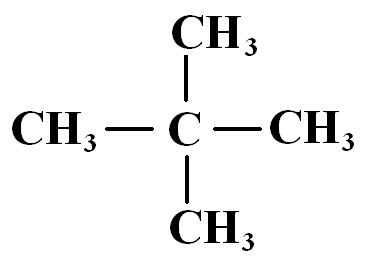
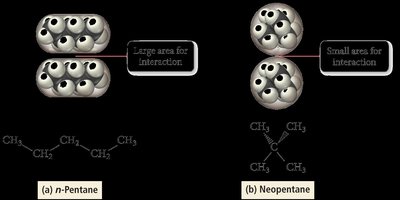
Molecular Shape Example: n-Pentane vs. Neopentane
n-Pentane (long chain) has a higher boiling point than neopentane (compact), due to greater surface area for intermolecular interaction.
Summary: London Dispersion Forces
LDFs operate between all molecules and atoms, regardless of polarity.
They are caused by fluctuations in electron distribution, not by electronegativity differences.
LDFs are weak in small molecules (e.g., H2, He) but significant in large molecules (e.g., Xe, I2, CCl4).
The more electrons a molecule has, the stronger its LDFs.
Example: Ranking London Dispersion Forces
Molecule | Melting Point (K) |
|---|---|
CH4 | 90.7 |
CF4 | 123 |
CCl4 | 250 |
CBr4 | 363 |
CI4 | 444 |
The trend shows that as the number of electrons increases, the strength of London dispersion forces increases, resulting in higher melting points.
