 Back
BackSolubility Equilibria: Ksp, Precipitation, and Factors Affecting Solubility
Study Guide - Smart Notes

Solubility Equilibria and the Solubility Product Constant (Ksp)
Introduction to Solubility Equilibria
Solubility equilibria describe the dynamic balance between a solid ionic compound and its dissolved ions in a saturated solution. This equilibrium is characterized by the solubility product constant, Ksp, which quantifies the extent to which a compound can dissolve in water at a given temperature.
Saturated Solution: A solution in which the maximum amount of solute has dissolved, and any additional solid will not increase the concentration of dissolved ions.
Dynamic Equilibrium: The rate of dissolution equals the rate of precipitation, so the concentrations of ions remain constant.
Ksp Expression: For a generic salt AB that dissociates as AB(s) ↔ A+(aq) + B-(aq), the solubility product is given by:
The value of Ksp is specific to each compound and temperature.
Magnitude of Ksp
The magnitude of Ksp indicates the relative solubility of a compound:
Small Ksp: Indicates low solubility (e.g., tooth enamel: for Ca5(PO4)3OH).
Large Ksp: Indicates higher solubility.
Interconversion: Mass Solubility, Molar Solubility, and Ksp
Solubility can be expressed as mass solubility (g/L), molar solubility (mol/L), or as Ksp. These quantities are interrelated:
Mass Solubility: The maximum mass of solute that dissolves in a given volume of solvent.
Molar Solubility: The number of moles of solute that dissolve per liter of solution.
Ksp: Calculated from the equilibrium concentrations of ions.
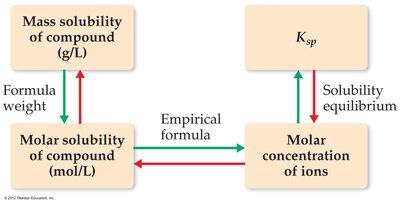
Calculating Solubility and Ksp
Example: Molar Solubility of Mg(OH)2
For Mg(OH)2(s) ↔ Mg2+(aq) + 2 OH-(aq), .
Let the molar solubility be s:
Solve for s to find the molar solubility.
Ranking Solubility
To compare the solubility of different salts, consider both the Ksp value and the stoichiometry of dissolution.
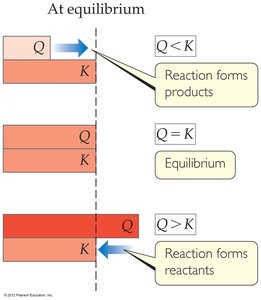
Precipitation and the Reaction Quotient (Q)
Predicting Precipitation
Whether a precipitate forms depends on the reaction quotient, Q, compared to Ksp:
If : No precipitate forms (solution is unsaturated).
If : Solution is saturated (at equilibrium).
If : Precipitate forms (solution is supersaturated).
Order of Precipitation and Separation of Ions
When multiple ions are present, the ion with the lowest product of ion concentration and Ksp will precipitate first. This principle is used in fractional (selective) precipitation to separate ions.
Factors Affecting Solubility
The Common Ion Effect
The presence of a common ion decreases the solubility of an ionic compound due to Le Châtelier’s principle. For example, adding Ca2+ to a solution of CaF2 will shift the equilibrium to the left, decreasing solubility.
Ksp Expression for CaF2:
Adding Ca2+ or F- reduces the solubility of CaF2.
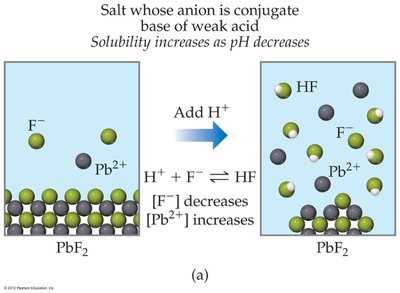
pH Effect on Solubility
The solubility of salts containing basic anions increases as pH decreases (solution becomes more acidic), because the added H+ reacts with the anion, shifting equilibrium to dissolve more solid.
For salts with anions that are conjugate bases of weak acids (e.g., F- in PbF2), solubility increases as pH decreases.
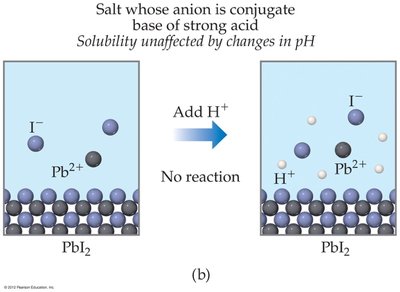
For salts with anions that are conjugate bases of strong acids (e.g., I- in PbI2), solubility is unaffected by pH.
Complex Ion Formation
Complex ion formation increases the solubility of certain ionic compounds. A complex ion is formed when a metal ion binds to one or more ligands (Lewis bases) such as NH3 or H2O.
Example:
The formation constant, Kf, quantifies the stability of the complex ion.
Complex Ion | Kf | Equilibrium Equation |
|---|---|---|
Ag(NH3)2+ | 1.7 × 107 | Ag+(aq) + 2 NH3(aq) ↔ Ag(NH3)2+(aq) |
Fe(CN)63- | 1 × 1031 | Fe3+(aq) + 6 CN-(aq) ↔ Fe(CN)63-(aq) |
Cu(NH3)42+ | 5.6 × 1011 | Cu2+(aq) + 4 NH3(aq) ↔ Cu(NH3)42+(aq) |
Additional info: See full table in textbook for more examples. |

Amphoterism
Some metal hydroxides are amphoteric, meaning they can dissolve in both strongly acidic and strongly basic solutions. This is due to their ability to react with both H+ (acid) and OH- (base) to form soluble species or complex ions.

Summary Table: Key Relationships in Solubility Equilibria
Concept | Description | Key Equation |
|---|---|---|
Ksp | Solubility product constant | |
Q | Reaction quotient (predicts precipitation) | Same as Ksp, but with initial concentrations |
Common Ion Effect | Decreases solubility | Le Châtelier’s principle |
pH Effect | Increases solubility for salts with basic anions | H+ reacts with anion |
Complex Ion Formation | Increases solubility | Formation constant Kf |
Amphoterism | Dissolves in acid and base | Acid-base and complex ion reactions |
Practice and Application
Write Ksp expressions for various salts (e.g., Ca3(PO4)2: ).
Calculate molar and mass solubility from Ksp and vice versa.
Predict precipitation using Q and Ksp.
Analyze the effect of common ions, pH, and complex ion formation on solubility.
