 Back
BackSolubility Equilibria, Thermodynamics, and Entropy: Advanced Concepts in General Chemistry
Study Guide - Smart Notes

Solubility Equilibria and Factors Affecting Solubility
Common Ion Effect
The common ion effect describes the decrease in solubility of an ionic compound when a solution already contains one of the ions present in the compound. This is a direct application of Le Chatelier's Principle, which states that the addition of a common ion shifts the dissolution equilibrium to favor the formation of more solid, thus reducing solubility.
Example: The solubility of CaCO3 decreases in the presence of Ca2+ or CO32− ions.
Key Equation:
pH Effect on Solubility
The solubility of salts containing basic anions increases as the pH decreases (solution becomes more acidic). This is because the added H+ reacts with the basic anion, removing it from solution and shifting the equilibrium to dissolve more solid.
Salts with basic anions (e.g., F−, CO32−, OH−, SO32−, PO43−) are more soluble in acidic solutions.
Salts with anions from strong acids (e.g., Cl−, NO3−) are unaffected by pH changes.
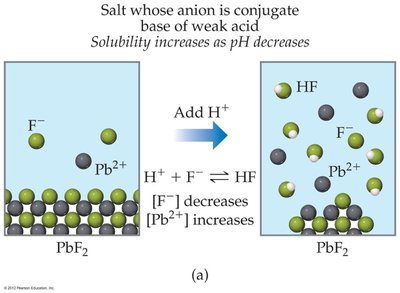
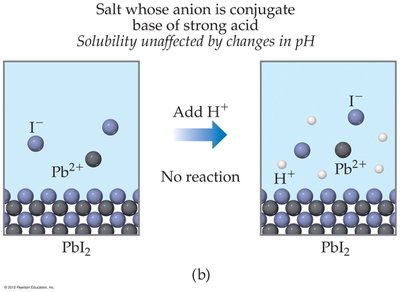
Example Calculation: To find the pH of a saturated Ca(OH)2 solution, use the Ksp expression and the relationship between [OH−] and pOH/pH.
Complex Ion Formation
Complex ions are formed when a metal cation (Lewis acid) binds to one or more ligands (Lewis bases) such as H2O or NH3. The formation of complex ions can greatly increase the solubility of otherwise insoluble salts by removing the metal ion from solution and shifting the equilibrium.
Example:
Formation constant (Kf): Indicates the stability of the complex ion.

Amphoterism
An amphoteric substance can react with both acids and bases. Many metal hydroxides (e.g., Al(OH)3) are amphoteric and dissolve in both strongly acidic and strongly basic solutions due to acid-base reactions or complex ion formation.
In acid:
In base:
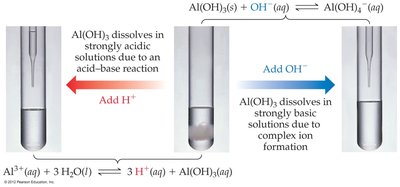
Thermodynamics: Spontaneity, Entropy, and the Laws of Thermodynamics
Spontaneous and Nonspontaneous Processes
A spontaneous process occurs without outside intervention. Spontaneity does not imply speed; some spontaneous processes are slow. The reverse of a spontaneous process is nonspontaneous. Temperature and pressure can affect spontaneity.
Example: Melting of ice is spontaneous above 0°C but nonspontaneous below 0°C.
Reversible process: Can be reversed by infinitesimal changes; maximizes work.
Irreversible process: Cannot be exactly reversed; all real spontaneous processes are irreversible.
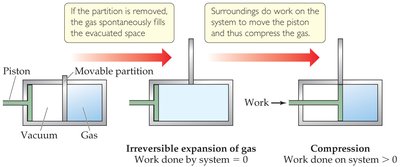

Entropy (S) and the Second Law of Thermodynamics
Entropy (S) is a measure of the disorder or randomness of a system. It is a state function, meaning its change depends only on the initial and final states, not the path taken. The second law of thermodynamics states that the entropy of the universe increases in any spontaneous process.
Mathematical definition:
For heat transfer at constant temperature:
Second Law: For a reversible process, ; for an irreversible (spontaneous) process, .

Molecular Interpretation of Entropy and the Third Law of Thermodynamics
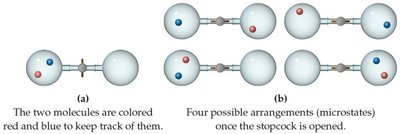
Statistical thermodynamics connects macroscopic properties with microscopic behavior. A microstate is a specific arrangement of molecules' positions and energies. The number of microstates (W) is related to entropy by the Boltzmann equation:
Where k is the Boltzmann constant ()
More microstates = higher entropy
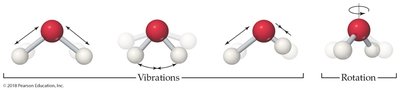
Molecules have different types of motion (degrees of freedom):
Translational: Movement from one place to another
Vibrational: Atoms oscillate within a molecule
Rotational: Molecule rotates about an axis
Entropy increases with:
Increasing volume (more positions for molecules)
Increasing temperature (greater energy distribution)
Increasing number of particles (more degrees of freedom)
Phase changes:
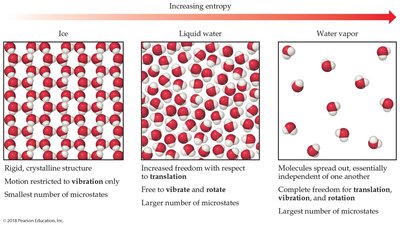
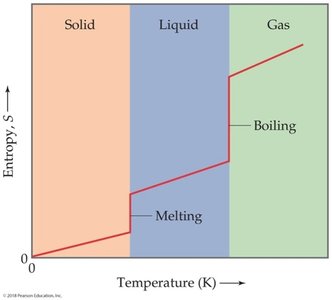
Third Law of Thermodynamics: The entropy of a perfect crystal at absolute zero (0 K) is zero. At this temperature, there is only one microstate (perfect order).
; at 0 K, , so
Summary Table: Factors Affecting Solubility
Factor | Effect on Solubility | Example |
|---|---|---|
Common Ion | Decreases | Adding Ca2+ to CaCO3 |
pH (for basic anion) | Increases as pH decreases | CaCO3 in acid |
Complex Ion Formation | Increases | AgCl in NH3 |
Amphoterism | Increases in strong acid or base | Al(OH)3 |
Practice Problems and Applications
Calculate the molar solubility of Ca(OH)2 in pure water and in buffered solutions using and pH relationships.
Predict which salts will be more soluble in acidic conditions (those with basic anions).
Classify processes as spontaneous/nonspontaneous, reversible/irreversible, and predict the sign of for chemical changes.
Apply the Boltzmann equation to relate entropy and microstates.
