 Back
BackSolubility Product Constant (Ksp) and Factors Affecting Solubility
Study Guide - Smart Notes

Solubility Product Constant (Ksp)
Equilibrium in Saturated Solutions
A saturated solution is at equilibrium between the dissolved ions and the undissolved solid. The solubility product constant, Ksp, is used to describe this equilibrium for slightly soluble salts.
Equilibrium Expression: For a salt AB2 that dissociates as AB2 (s) → A2+ (aq) + 2B- (aq), the equilibrium expression is:
Ksp Magnitude: The value of Ksp indicates the solubility of the solid; higher Ksp means greater solubility.
Solubility and Ksp
Solubility is the amount of substance needed to make a saturated solution, expressed in g/L or mol/L. Ksp is a unitless equilibrium constant, constant at a given temperature.
Solubility: Amount of solute required for saturation.
Ksp: Equilibrium constant for the dissolution process.
Influencing Factors: Solubility is affected by the presence of other ions (e.g., H+, common ions).
Example Calculations
Calculating Ksp: If a saturated solution of Ag2SO4 at 25°C has [Ag+] = 1.47 × 10-4 M, and Ag2SO4 dissociates completely, Ksp can be calculated using the stoichiometry and equilibrium concentrations.
Calculating Solubility from Ksp: For BaF2 with Ksp = 1.7 × 10-6, solubility in g/L and mol/L can be determined by setting up the equilibrium expression and solving for the concentration of Ba2+ and F-.
Determining Ion Concentrations: For calcium phosphate (Ksp = 2.0 × 10-29), use the stoichiometry to find [Ca2+] and [PO43-].
Factors Affecting Solubility
Common-Ion Effect
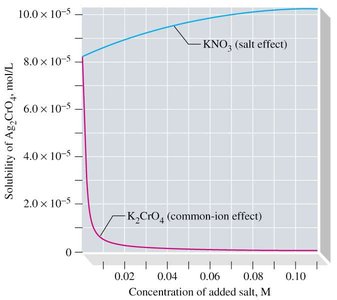
The solubility of a slightly soluble salt decreases in the presence of a common ion from a second solute. This is due to Le Chatelier's principle, which shifts the equilibrium toward the undissolved solid.
Example: Adding K2CrO4 to Ag2CrO4 decreases its solubility due to the common CrO42- ion.
pH Effect
The solubility of salts containing basic anions increases as [H+] increases (lower pH), because the basic anion reacts with H+ to form a weak acid, reducing its concentration and shifting equilibrium toward dissolution.
Example: Salts like CaF2, Pb3(PO4)2, and Cd(OH)2 have solubility affected by pH.
Complex Ion Effect
The solubility of a metal ion increases if it can form a complex ion with a Lewis base. Complex formation removes the metal ion from solution, shifting equilibrium toward dissolution.
Example: Ag+ forms [Ag(NH3)2]+ in the presence of NH3, increasing Ag+ solubility.
Diffuse Ion Effect (Salt Effect)
Adding a second salt that does not share a common ion creates ion clouds that "hide" the ions of the slightly soluble salt from each other, increasing solubility.
Example: Adding KNO3 to Ag2CrO4 increases its solubility due to the salt effect.
Precipitation and Solubility Rules
Precipitation Criteria
Precipitation occurs when the product of the initial ion concentrations (Q) exceeds the Ksp for the salt.
If Q > Ksp: Precipitation occurs until Q = Ksp.
If Q = Ksp: The system is at equilibrium.
If Q < Ksp: The solid dissolves until Q = Ksp.
Example Calculations
AgBr Precipitation: For AgBr (Ksp = 5.0 × 10-13), determine if precipitation occurs by calculating Q for given [Ag+] and [Br-].
CaF2 Precipitation: Mixing NaF and Ca(NO3)2 and comparing Q to Ksp determines if CaF2 precipitates.
Selective Precipitation
Separation of Metals by Precipitation
Selective precipitation is used to recover and separate metals from solution. The concentration of a precipitating ion required to cause precipitation depends on the Ksp values of the salts involved.
Example: In a solution with 0.030 M Ag+ and 0.0010 M Pb2+, calculate the concentration of sulfate needed to precipitate each metal and determine which metal precipitates first based on their Ksp values.
Summary Table: Factors Affecting Solubility
Factor | Effect on Solubility | Example |
|---|---|---|
Common-Ion Effect | Decreases | K2CrO4 added to Ag2CrO4 |
pH Effect | Increases (for basic anions) | CaF2, Cd(OH)2 |
Complex Ion Effect | Increases | Ag+ with NH3 |
Salt Effect (Diffuse Ion) | Increases | KNO3 added to Ag2CrO4 |
Additional info: Academic context and examples were expanded for clarity and completeness. The included image directly illustrates the common-ion and salt effects on solubility, matching the explanation above.
