 Back
BackSolubility Rules and Classification of Ionic Compounds
Study Guide - Smart Notes

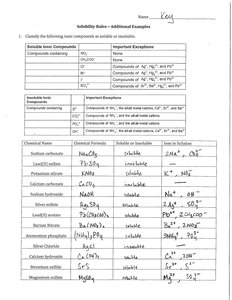
Q1. Classify the following ionic compounds as soluble or insoluble.
Background
Topic: Solubility Rules in Aqueous Solutions
This question tests your understanding of solubility rules for ionic compounds in water, including recognizing exceptions to general rules. It is a fundamental topic in general chemistry, especially for predicting precipitation reactions.
Key Terms and Formulas
Soluble: A compound that dissolves in water to form a homogeneous solution.
Insoluble: A compound that does not dissolve appreciably in water.
Solubility Rules: Guidelines for predicting whether an ionic compound will dissolve in water. For example:
All compounds containing Na+, K+, NH4+, NO3-, and CH3COO- are generally soluble.
Compounds containing Cl-, Br-, I- are soluble except with Ag+, Pb2+, and Hg22+.
Sulfates (SO42-) are soluble except with Ba2+, Pb2+, Ca2+, Sr2+.
Carbonates (CO32-), phosphates (PO43-), and hydroxides (OH-) are generally insoluble except with alkali metals and NH4+.
Step-by-Step Guidance
Identify the cation and anion in each compound. For example, in Na2CO3, the cation is Na+ and the anion is CO32-.
Refer to the solubility rules table to determine if the compound is generally soluble or insoluble. Start with the cation: alkali metals (like Na+, K+) and NH4+ are always soluble.
Check for any important exceptions listed in the solubility rules. For example, AgCl is insoluble due to the exception for Ag+ with Cl-.
For compounds with polyatomic ions (like SO42- or CO32-), check both the general rule and the exceptions for the specific cation present.
Write down your classification (soluble or insoluble) for each compound, but do not proceed to the final answer yet.
Try solving on your own before revealing the answer!
