 Back
BackSolution Concentration: Definitions, Calculations, and Applications
Study Guide - Smart Notes

Solution Concentration
Overview of Concentration Terms
Concentration is a fundamental concept in chemistry that describes the amount of solute present in a given quantity of solvent or solution. Several different units and methods are used to express concentration, each suited to specific types of solutions and applications.
Molarity (M): The number of moles of solute per liter of solution.
Molality (m): The number of moles of solute per kilogram of solvent.
Mass Percent (%): The mass of solute divided by the total mass of solution, multiplied by 100.
Parts per million (ppm) and parts per billion (ppb): Mass ratios scaled by 106 or 109 respectively.
Volume Percent (%): The volume of solute divided by the total volume of solution, multiplied by 100.
Mole Fraction (X): The ratio of moles of solute to the total moles in the solution.
Molarity (M)
Molarity is widely used for aqueous solutions and is defined as the number of moles of solute per liter of solution.
Formula:
Example: Calculating the molarity of 1.25 g of calcium chloride in 50.0 mL of solution.
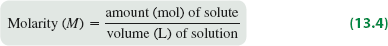
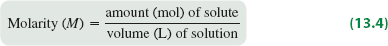
Molality (m)
Molality is useful when temperature changes are involved, as it depends only on the mass of the solvent.
Formula:
Example: Calculating the molality of 1.25 g of potassium nitrate in 25.0 mL of water (density = 1 g/mL).
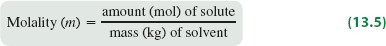
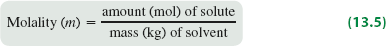
Mass Percent
Mass percent expresses concentration as the percentage of solute mass relative to the total solution mass.
Formula:
Example: Calculating the mass percent of sodium acetate in a solution of 5.00 g sodium acetate in 100.0 g water.
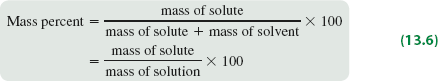
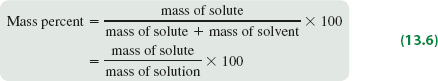
Parts per Million (ppm) and Parts per Billion (ppb)
These units are used for very dilute solutions, especially in environmental chemistry.
Formula for ppm:
Formula for ppb:
Example: Calculating ppm and ppb for 0.0025 mol of Hg in 10,000 L of water.
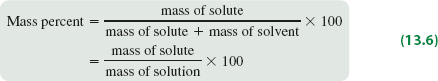
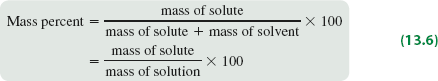
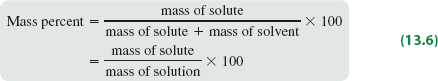
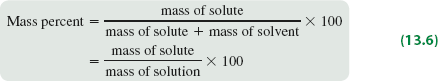
Volume Percent
Volume percent is commonly used for solutions of liquids, especially when both solute and solvent are liquids.
Formula:
Example: Calculating the volume percent of isopropyl alcohol in a solution with 80.0 g isopropyl alcohol (d = 0.786 g/mL) and 20.0 g water (d = 1 g/mL).


Mole Fraction (X)
Mole fraction is a unitless ratio that expresses the proportion of moles of a component relative to the total moles in the solution.
Formula:
Example: Calculating the mole fraction of iron(III) chloride when 10.5 g is dissolved in 250.0 g of water.
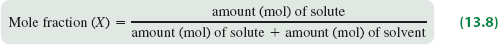
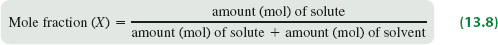
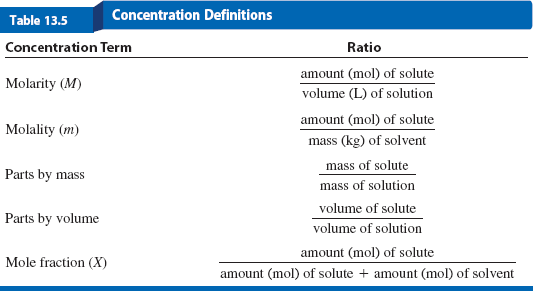
Summary Table: Concentration Definitions
The following table summarizes the main concentration terms and their mathematical definitions:
Concentration Term | Ratio |
|---|---|
Molarity (M) | amount (mol) of solute / volume (L) of solution |
Molality (m) | amount (mol) of solute / mass (kg) of solvent |
Parts by mass | mass of solute / mass of solution |
Parts by volume | volume of solute / volume of solution |
Mole fraction (X) | amount (mol) of solute / [amount (mol) of solute + amount (mol) of solvent] |
Key Points and Applications
Concentration units are chosen based on the nature of the solution and the required precision.
Molarity is temperature-dependent, while molality is not.
Mass percent, ppm, and ppb are useful for expressing concentrations in environmental and industrial contexts.
Mole fraction is important in thermodynamic calculations and colligative properties.
Additional info: The notes and examples provided cover all major concentration units relevant to general chemistry, including practical calculation steps and typical applications.
