 Back
BackSolution Properties, Raoult’s Law, and Phase Change Temperature Shifts
Study Guide - Smart Notes

Solution Formation and Intermolecular Forces
Types of Interactions in Solution Formation
When a solution forms, three main types of intermolecular forces (IMFs) must be considered: solute-solute, solvent-solvent, and solute-solvent interactions. The system tends to maximize strong attractive interactions, which minimizes the overall energy of the system.
Solute-solute interactions: Forces between solute particles must be overcome for dissolution.
Solvent-solvent interactions: Forces between solvent molecules must also be overcome.
Solute-solvent interactions: New interactions form between solute and solvent molecules, stabilizing the solution.
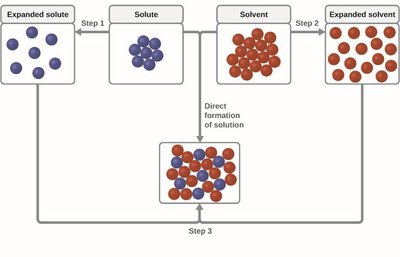
Example: Dissolving salt in water involves breaking ionic bonds in the salt and hydrogen bonds in water, then forming ion-dipole interactions.
Ion-Dipole Forces
When ionic compounds dissolve in polar solvents like water, a new type of IMF called the ion-dipole force is formed. Water molecules orient themselves around ions, with the oxygen atom (negative) facing cations and the hydrogen atoms (positive) facing anions. This structured arrangement is called a solvent shell and strongly favors exothermic dissolution reactions for ionic compounds.
Solvent shell: Layer of solvent molecules surrounding a solute particle.
Importance: Solvent shells are crucial in biological systems, such as protein folding.
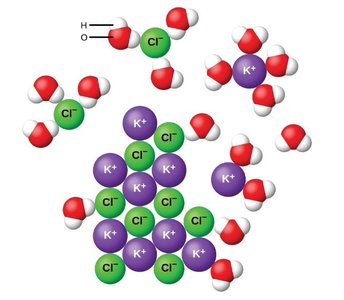
Example: Dissolving KCl in water forms K+ and Cl- ions surrounded by water molecules.
Solubility and Solution Types
Solubility Concepts
Solubility is the maximum amount of solute that can dissolve in a solvent at equilibrium under specific conditions. Solutions can be classified as:
Unsaturated: Contains less solute than the solubility limit.
Saturated: Contains solute at the solubility limit.
Supersaturated: Contains more solute than the solubility limit (unstable).
Dilute vs. Concentrated: Refers to the relative amount of solute present.
Miscibility describes the ability of two liquids to mix. "Like dissolves like"—polar solvents dissolve polar solutes, and nonpolar solvents dissolve nonpolar solutes.
Temperature and Pressure Effects:
Solids dissolve better at higher temperatures.
Gases dissolve worse at higher temperatures.
Gas solubility increases with pressure (Henry’s Law):
Electrolytes and Conductivity
Electrolytes are substances that produce ions in solution and conduct electricity. They are classified as:
Nonelectrolytes: Do not produce ions (e.g., covalent compounds like ethanol).
Strong electrolytes: Completely dissociate into ions (e.g., soluble ionic compounds, strong acids/bases).
Weak electrolytes: Partially dissociate (e.g., weak acids/bases).
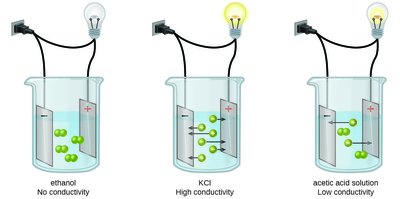
Example: KCl solution conducts electricity well, while ethanol does not.
Concentration Units and Conversions
Mole Fraction and Molality
Concentration can be expressed in several ways:
Mole fraction (χ): (between 0 and 1)
Molality (m):
Molarity (M):
Conversions between these units often require density and careful attention to whether the denominator refers to solution or solvent.
Raoult’s Law and Colligative Properties
Vapor Pressure Lowering (Raoult’s Law)
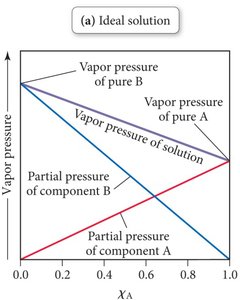
Adding a nonvolatile solute to a solvent lowers the solvent’s vapor pressure. Raoult’s Law for ideal solutions states:
, where is the vapor pressure of pure solvent A.
For mixtures of volatile liquids:
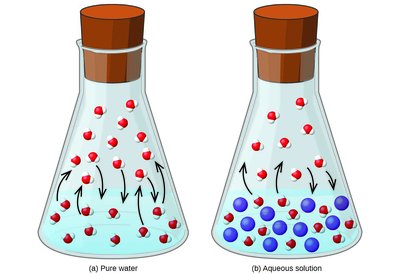
Example: Dissolving sugar in water reduces the vapor pressure compared to pure water.
Dynamic Equilibrium and Vapor Pressure
In a solution, fewer solvent molecules are present at the surface due to the presence of solute, resulting in fewer molecules escaping to the gas phase and thus a lower vapor pressure.
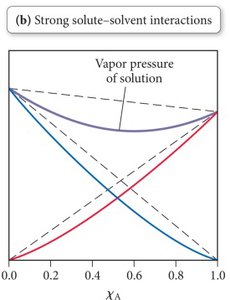
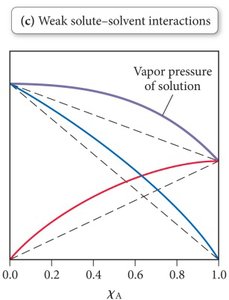
Non-Ideal Solutions
Non-ideal solutions occur when solute-solvent interactions differ from solvent-solvent or solute-solute interactions:
Stronger solute-solvent IMFs: Lower vapor pressure than predicted (e.g., acetone/chloroform).
Weaker solute-solvent IMFs: Higher vapor pressure than predicted (e.g., benzene/methanol).
Distillation and Fractional Distillation
Distillation Techniques
Distillation is a method of purifying liquids by exploiting differences in boiling points. The compound with the lowest boiling point vaporizes first and can be condensed and collected. Fractional distillation allows separation of mixtures into multiple components.
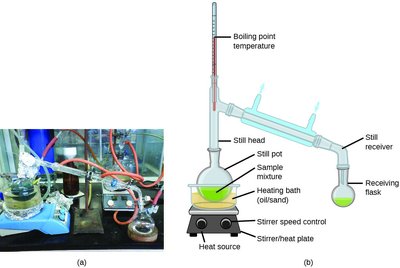
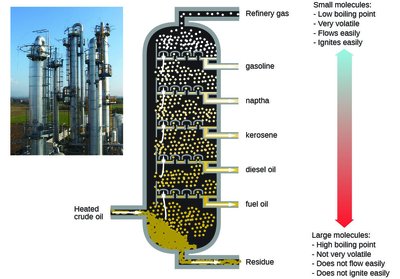
Example: Crude oil is separated into gasoline, kerosene, diesel, and other fractions by fractional distillation.
Colligative Properties: Boiling and Freezing Point Shifts
Phase Change Temperature Shifts
Adding a solute to a solvent affects its phase change temperatures:
Freezing point depression: Solute disrupts the solvent lattice, lowering the freezing point.
Boiling point elevation: Lower vapor pressure means a higher temperature is needed to boil.
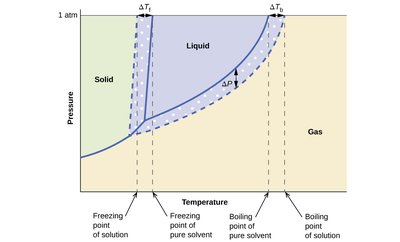
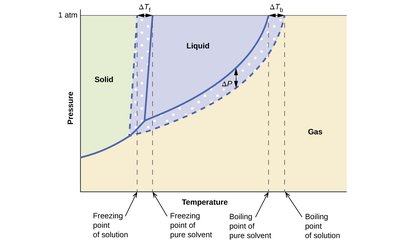
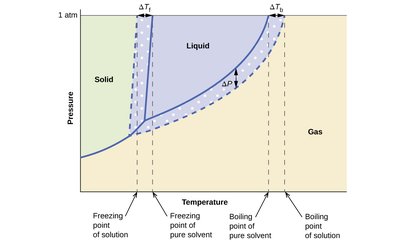
Key Equations:
Freezing point depression:
Boiling point elevation:
Example: Dissolving salt in water lowers its freezing point, which is why salt is used to melt ice on roads.
Additional info: These notes cover core concepts from Ch. 11 (Solutions and Colligative Properties) and connect to Ch. 10 (Liquids, Solids, and Intermolecular Forces) and Ch. 3 (Solutions and Chemical Quantities).
