 Back
BackSolutions and Phase Behavior in General Chemistry
Study Guide - Smart Notes

Solutions and Their Properties
Definition and Types of Solutions
A solution is a homogeneous mixture composed of two or more substances, where the solute is uniformly distributed within the solvent. Solutions can exist in all three phases: solid (e.g., brass), liquid (e.g., salt water), and gas (e.g., air). The solute is the minor component, and the solvent is the major component.
Solubility: The maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature.
Unsaturated solution: Contains less solute than the maximum amount; more solute can dissolve.
Saturated solution: Contains the maximum amount of solute; no more solute dissolves.
Supersaturated solution: Contains more solute than is stable; excess solute will precipitate out.

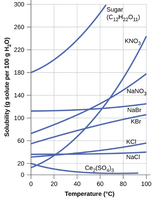
Solubility and Temperature
Solubility often depends on temperature. For most solids, solubility increases with temperature, while for gases, solubility decreases as temperature increases.
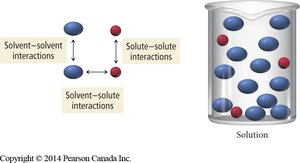
Relative Interactions and Solution Formation
Solution formation depends on the relative strengths of solute-solute, solvent-solvent, and solute-solvent interactions. The process is energetically favorable if the new interactions formed are comparable to or stronger than those broken.
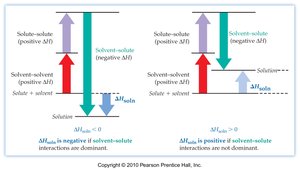
Thermodynamics of Mixing
The spontaneity of solution formation is governed by the Gibbs free energy change:
ΔmixH: Enthalpy change of mixing (can be positive or negative).
ΔmixS: Entropy change of mixing (usually positive due to increased disorder).
"Like Dissolves Like" Principle
Substances with similar types of intermolecular forces are likely to be soluble in each other:
Non-polar solutes dissolve in non-polar solvents (e.g., CCl4 in C6H6).
Polar solutes dissolve in polar solvents (e.g., ethanol in water).
Ionic compounds dissolve in polar solvents (e.g., NaCl in water).
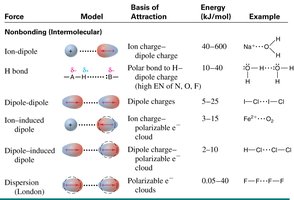
Intermolecular Forces in Solutions
Different types of intermolecular forces influence solubility and solution properties:
Ion-dipole: Between ions and polar molecules.
Hydrogen bonding: Between molecules with H bonded to N, O, or F.
Dipole-dipole: Between polar molecules.
London dispersion: Present in all molecules, dominant in non-polar substances.
Common Laboratory Solvents
Solvents are classified as polar or non-polar based on their molecular structure and ability to dissolve different solutes.
Common Polar Solvents | Common Nonpolar Solvents |
|---|---|
Water (H2O) | Hexane (C6H14) |
Acetone (CH3COCH3) | Diethyl ether (CH3CH2OCH2CH3) |
Methanol (CH3OH) | Methylbenzene (toluene, C7H8) |
Ethanol (CH3CH2OH) | Carbon tetrachloride (CCl4) |

Gas-Liquid Solutions and Henry's Law
Solubility of Gases in Liquids
Gases have weak intermolecular forces and are generally less soluble in polar solvents like water. However, small amounts of gases dissolve and are essential for life (e.g., O2 for aquatic organisms).
Solubility of gases decreases with increasing temperature.
CO2 in water forms carbonic acid, relevant to ocean acidification.

Henry's Law
The solubility of a gas in a liquid is proportional to the partial pressure of the gas above the liquid:
Sgas: Solubility of the gas
kgas: Henry's law constant (specific for each gas-solvent pair and temperature)
Pgas: Partial pressure of the gas

Liquid-Liquid Phase Behavior
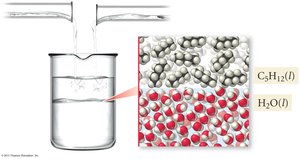
Miscibility and Immiscibility
Miscibility is the ability of two liquids to mix in all proportions, forming a homogeneous solution. If two liquids do not mix, they are immiscible. The entropy of mixing is generally positive, favoring mixing, but enthalpy changes can oppose or enhance this effect.
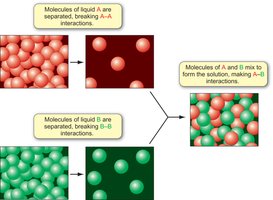
Enthalpy and Entropy in Mixing
The enthalpy change of mixing depends on the relative strengths of intermolecular forces. If A-B interactions are stronger than A-A and B-B, mixing is favored (ΔHmix negative).
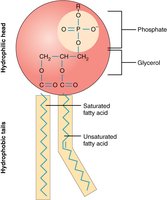
Dual Polarity and Surfactants
Some molecules, such as fatty acids, phospholipids, and soaps, have both polar and non-polar regions. These molecules can interact with both polar and non-polar substances, making them effective as surfactants.


Ideal and Non-Ideal Solutions
Ideal Solutions
Ideal solutions are those in which the enthalpy of mixing is zero and the components are similar in size, shape, and intermolecular forces. The Gibbs energy of mixing is determined solely by entropy.
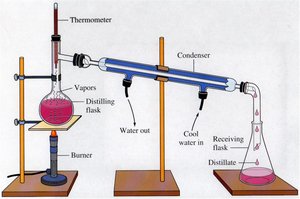
Distillation and Fractional Distillation
Distillation separates components of a liquid mixture based on differences in volatility. Fractional distillation is used when components have similar boiling points and involves repeated vaporization-condensation cycles.
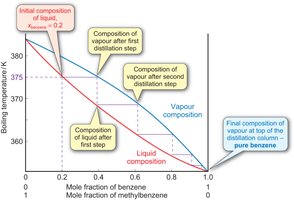
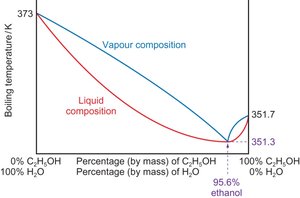
Azeotropes
Some mixtures form azeotropes, where the liquid and vapor compositions are the same at a certain ratio, preventing further separation by distillation.
Colligative Properties
Definition and Types
Colligative properties depend only on the number of solute particles in solution, not their identity. These include:
Vapor pressure lowering
Boiling point elevation
Freezing point depression
Osmotic pressure
Vapor Pressure Lowering and Raoult's Law
Adding a non-volatile solute to a solvent lowers the solvent's vapor pressure. Raoult's Law quantifies this effect:
pA: Vapor pressure of solvent in solution
XA: Mole fraction of solvent
pA∘: Vapor pressure of pure solvent
Boiling Point Elevation
The boiling point of a solution is higher than that of the pure solvent. The increase is given by:
Kb: Boiling point elevation constant
m: Molality of the solution
i: van't Hoff factor (number of particles per formula unit)
Freezing Point Depression
The freezing point of a solution is lower than that of the pure solvent:
Kf: Freezing point depression constant
Osmosis and Osmotic Pressure
Osmosis is the movement of solvent through a semi-permeable membrane from a region of lower solute concentration to higher concentration. The osmotic pressure (π) is given by:
C: Molar concentration of solute
R: Gas constant
T: Temperature in Kelvin
Phases and Phase Diagrams
Phases and Phase Boundaries
A phase is a physically and chemically uniform region of a system. Phase boundaries exist between immiscible liquids or between different states of matter (solid, liquid, gas).
Phase Diagrams
Phase diagrams plot pressure versus temperature and show the conditions under which different phases are stable. Key features include the triple point (where all three phases coexist) and the critical point (above which a supercritical fluid exists).
Supercritical Fluids
Above the critical temperature and pressure, substances exist as supercritical fluids, which have properties of both liquids and gases. Supercritical CO2 is used for decaffeination and other extraction processes.
Osmosis and Reverse Osmosis
Osmosis is crucial in biological systems (e.g., red blood cells). Reverse osmosis, where pressure is applied to force solvent from a concentrated solution to a dilute one, is used in water purification and desalination.
