Back
BackSolutions and Their Properties: Electrolytes, Solubility, and Colligative Effects
Study Guide - Smart Notes

Electrolytes and Nonelectrolytes
Definitions and Conductivity
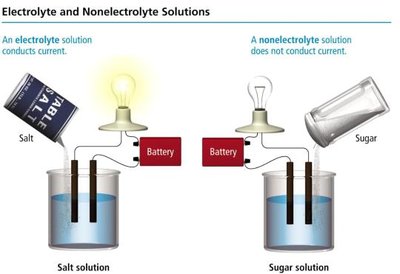
Electrolytes and nonelectrolytes are distinguished by their ability to conduct electricity when dissolved in water. This property depends on the presence of ions in solution.
Electrolyte: A substance that dissolves in water and produces ions, allowing the solution to conduct electricity.
Nonelectrolyte: A substance that dissolves in water but does not produce ions, so the solution does not conduct electricity.
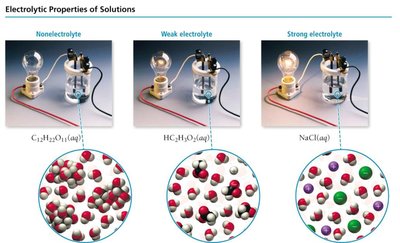
Strong Electrolyte: Dissolves and completely dissociates into ions (e.g., NaCl, HCl).
Weak Electrolyte: Dissolves and partially dissociates into ions (e.g., acetic acid).
Nonelectrolyte: Dissolves but does not dissociate (e.g., sugar, alcohol).
Energy Changes in Solution Formation
Enthalpy of Solution
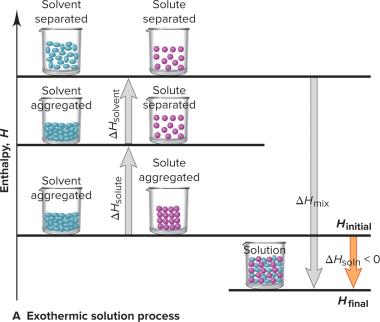
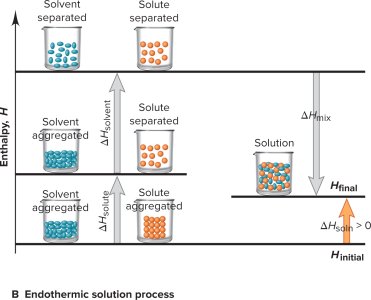
The formation of a solution involves energy changes associated with separating solute and solvent particles and mixing them together.
Step 1: Separation of solute particles (endothermic, requires energy).
Step 2: Separation of solvent particles (endothermic, requires energy).
Step 3: Mixing of solute and solvent particles (exothermic, releases energy).
Heat of Solution (ΔHsoln): The total enthalpy change, which can be exothermic (ΔHsoln < 0) or endothermic (ΔHsoln > 0).
Solubility and Equilibrium
Saturated, Unsaturated, and Supersaturated Solutions
Solubility describes the maximum amount of solute that can dissolve in a solvent at a given temperature.
Saturated Solution: Contains the maximum amount of dissolved solute in equilibrium with undissolved solute.
Unsaturated Solution: Contains less than the equilibrium concentration of dissolved solute; more solute can dissolve.
Supersaturated Solution: Contains more than the equilibrium concentration of solute; unstable and excess solute may crystallize.
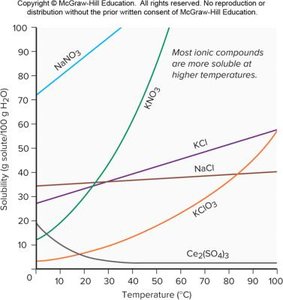
Temperature and Solubility
For most ionic salts, solubility increases with temperature.
Solubility of Gases and Henry's Law
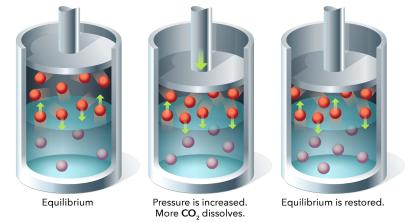
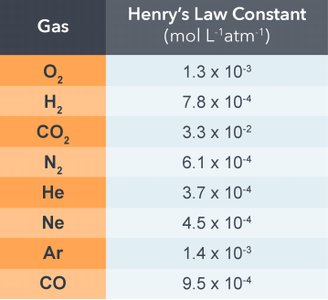
Gas Solubility and Pressure
The solubility of gases in liquids is governed by Henry's Law:
Henry's Law:
Where is the solubility, is the Henry's law constant, and is the partial pressure of the gas.
Gas solubility decreases with increasing temperature.
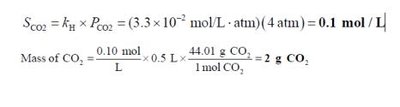
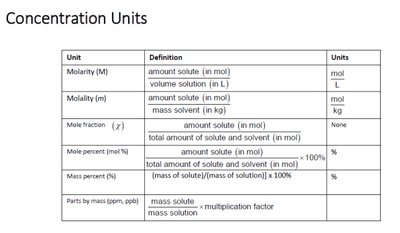
Concentration Units
Common Units of Solution Concentration
Concentration units are essential for quantifying the amount of solute in a solution.
Molarity (M): Moles of solute per liter of solution.
Molality (m): Moles of solute per kilogram of solvent.
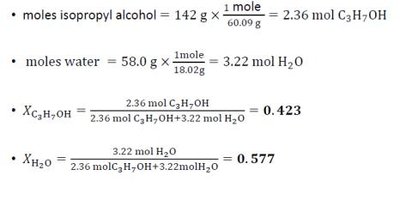
Mole Fraction (χ): Ratio of moles of one component to total moles in solution.
Mass Percent (%): Mass of solute divided by mass of solution, multiplied by 100.
Colligative Properties
Types and Definitions
Colligative properties depend only on the number of solute particles, not their identity. The four main colligative properties are:
Freezing Point Depression
Boiling Point Elevation
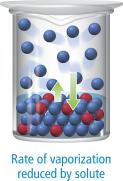
Vapor Pressure Lowering
Osmotic Pressure
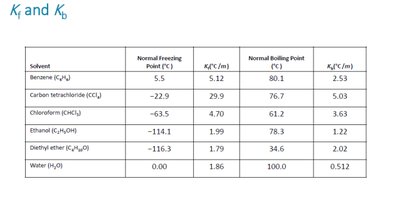
Freezing Point Depression
The freezing point of a solution is lower than that of the pure solvent.
is the freezing point depression constant, is molality.

Boiling Point Elevation
The boiling point of a solution is higher than that of the pure solvent.
is the boiling point elevation constant, is molality.


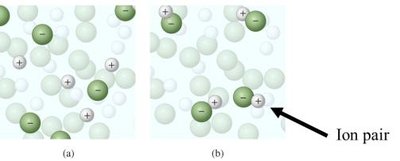
Van't Hoff Factor (i)
The Van't Hoff factor accounts for the number of particles produced by dissociation of electrolytes.
For nonelectrolytes, ; for strong electrolytes, equals the number of ions formed.
Colligative property equations: ,
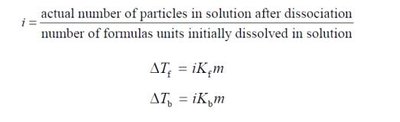
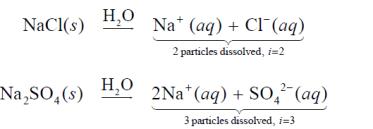
Vapor Pressure Lowering
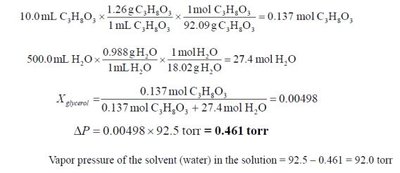
Raoult's Law
The vapor pressure of a solvent above a solution is lower than that of the pure solvent due to the presence of solute particles.
Raoult's Law:
Vapor pressure lowering:


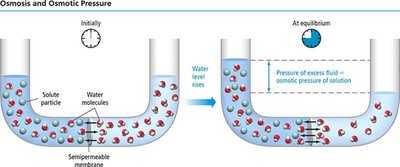
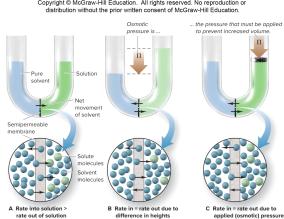
Osmosis and Osmotic Pressure
Osmosis and Semipermeable Membranes
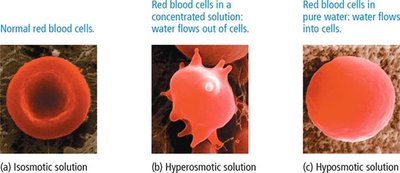
Osmosis is the movement of solvent molecules through a semipermeable membrane from a region of low solute concentration to high solute concentration.
Osmotic Pressure (π): The pressure required to prevent the net flow of solvent.
Formula:
= molarity, = gas constant , = temperature in Kelvin.


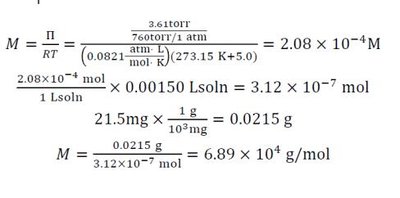
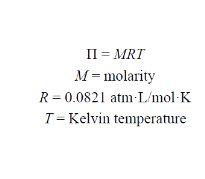
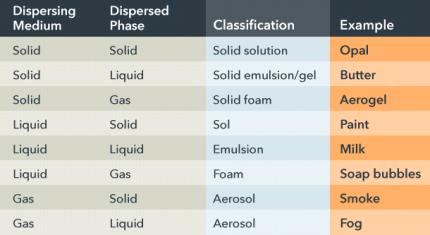
Colloids and the Tyndall Effect
Classification and Properties
Colloids are heterogeneous mixtures where the dispersed particles do not settle out and are intermediate in size between solutions and suspensions.
Colloid: Mixture with dispersed particles larger than those in a solution but smaller than those in a suspension.
Tyndall Effect: Scattering of light by colloidal particles.
Dispersing Medium | Dispersed Phase | Classification | Example |
|---|---|---|---|
Solid | Solid | Solid solution | Opal |
Solid | Liquid | Solid emulsion/gel | Butter |
Solid | Gas | Solid foam | Aerogel |
Liquid | Solid | Sol | Paint |
Liquid | Liquid | Emulsion | Milk |
Liquid | Gas | Foam | Soap bubbles |
Gas | Solid | Aerosol | Smoke |
Gas | Liquid | Aerosol | Fog |